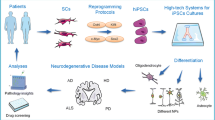
Abstract
Three-dimensional (3D) in vitro culture systems using human induced pluripotent stem cells (hiPSCs) are useful tools to model neurodegenerative disease biology in physiologically relevant microenvironments. Though many successful biomaterials-based 3D model systems have been established for other neurogenerative diseases, such as Alzheimer’s disease, relatively few exist for Parkinson’s disease (PD) research. We employed tissue engineering approaches to construct a 3D silk scaffold-based platform for the culture of hiPSC-dopaminergic (DA) neurons derived from healthy individuals and PD patients harboring LRRK2 G2019S or GBA N370S mutations. We then compared results from protein, gene expression, and metabolic analyses obtained from two-dimensional (2D) and 3D culture systems. The 3D platform enabled the formation of dense dopamine neuronal network architectures and developed biological profiles both similar and distinct from 2D culture systems in healthy and PD disease lines. PD cultures developed in 3D platforms showed elevated levels of α-synuclein and alterations in purine metabolite profiles. Furthermore, computational network analysis of transcriptomic networks nominated several novel molecular interactions occurring in neurons from patients with mutations in LRRK2 and GBA. We conclude that the brain-like 3D system presented here is a realistic platform to interrogate molecular mechanisms underlying PD biology.
Similar content being viewed by others
Availability of data and material
The datasets used and analyzed for this study can be obtained from corresponding authors upon reasonable request. Raw sequencing data (consented hiPSC donors only) and normalized read counts can be freely accessed through the Gene Expression Omnibus (GSE172409).
Abbreviations
- 2D:
-
Two dimensional
- 3D:
-
Three dimensional
- ANOVA:
-
Analysis of variance
- BP:
-
Biological processes
- CCK:
-
Cholecystokinin
- CCKBR:
-
Cholecystokinin B receptor
- CDG:
-
Candidate disease gene
- CM:
-
Conditioned media
- Cyclic AMP:
-
Cyclic 3´,5´-cyclic adenosine monophosphate
- DA:
-
Dopaminergic
- DAPI:
-
4′, 6-Diamidino-2-phenylindole
- DEG:
-
Differentially expressed gene
- DGKG:
-
Diacylglycerol Kinase Gamma
- DPBS:
-
Dulbecco’s phosphate buffered saline
- DRD1:
-
Dopamine receptor D1
- dsDNA:
-
Double-stranded Deoxyribonucleic acid
- ENPEP:
-
Glutamyl Aminopeptidase
- EXTL1:
-
Exostosin Like Glycosyltransferase 1
- FPPs:
-
Floor plate progenitors
- GBA:
-
Glucocerebrosidase
- GLIDE:
-
Global and local integrated diffusion embedding
- GO:
-
Gene ontology
- GRK5:
-
G Protein-Coupled Receptor Kinase 5
- HES1:
-
Hes Family BHLH Transcription Factor 1
- hiPSCs:
-
Human induced pluripotent stem cells
- IDA:
-
Information-dependent acquisition
- IGFBP5:
-
Insulin Like Growth Factor Binding Protein 5
- KLK6:
-
Kallikrein Related Peptidase 6
- LATS2:
-
Large Tumor Suppressor Kinase 2
- LC–MS:
-
Liquid chromatography tandem mass spectrometry
- LRRK2:
-
Leucine-rich repeat kinase 2
- LYN:
-
LYN Proto-Oncogene, Src Family Tyrosine Kinase
- MAOB:
-
Monoamine oxidase B
- MAPT:
-
Microtubule associated protein tau
- miRNA:
-
Micro ribonucleic acid
- mRNA:
-
Messenger ribonucleic acid
- MS2:
-
Fragmentation spectra
- PCA:
-
Principle component analysis
- PD:
-
Parkinson’s Disease
- PDK4:
-
Pyruvate Dehydrogenase Kinase 4
- PITX3:
-
Paired Like Homeodomain 3
- PLCB2:
-
Phospholipase C Beta 2
- PLD1:
-
Phospholipase D1
- PLK1:
-
Polo like kinase 1
- PSMD4:
-
Proteasome 26S Subunit, Non-ATPase 4
- PTER:
-
Phosphotriesterase Related
- SNCA:
-
α-Synuclein (gene)
- SNCAIP:
-
α-Synuclein interacting protein
- SPON1:
-
Spondin 1
- TCF12:
-
Transcription Factor 12
- TEAD1:
-
TEA domain transcription factor 1
- TEAD2:
-
TEA domain transcription factor 2
- TEAD3:
-
TEA domain transcription factor 3
- TH:
-
Tyrosine hydroxylase
- TUJ1:
-
β3-Tubulin
- USP9X:
-
Ubiquitin specific peptidase 9 X-linked
- YAS1:
-
Yes1 associated transcriptional regulator
References
Dorsey ER, Constantinescu R, Thompson JP et al (2007) Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology 68:384–386. https://doi.org/10.1212/01.wnl.0000247740.47667.03
Marras C, Beck JC, Bower JH et al (2018) Prevalence of Parkinson’s disease across North America. npj Parkinson’s Dis 4:21. https://doi.org/10.1038/s41531-018-0058-0
Cacabelos R (2017) Parkinson’s disease: from pathogenesis to pharmacogenomics. Int J Mol Sci 18:551. https://doi.org/10.3390/ijms18030551
Obeso JA, Stamelou M, Goetz CG et al (2017) Past, present, and future of Parkinson’s disease: a special essay on the 200th Anniversary of the Shaking Palsy. Mov Disord 32:1264–1310. https://doi.org/10.1002/mds.27115
Katzenschlager R, Lees AJ (2002) Treatment of Parkinson’s disease: levodopa as the first choice. J Neurol 249:1–1. https://doi.org/10.1007/s00415-002-1204-4
Ekstrand MI, Terzioglu M, Galter D et al (2007) Progressive Parkinsonism in mice with respiratory-chain-deficient dopamine neurons. Proc Natl Acad Sci U S A 104:1325–1330. https://doi.org/10.1073/pnas.0605208103
Konnova EA, Swanberg M (2018) Animal models of Parkinson’s disease. In: Stoker TB, Greenland JC (eds) Parkinson’s disease: pathogenesis and clinical aspects, 1st edn. Codon Publications, Brisbane, AU, pp 83–106
Kin K, Yasuhara T, Kameda M, Date I (2019) Animal models for Parkinson’s disease research: Trends in the 2000s. Int J Mol Sci 20:5402–5402. https://doi.org/10.3390/ijms20215402
Buckner RL, Krienen FM (2013) The evolution of distributed association networks in the human brain. Trends Cogn Sci 17:648–665. https://doi.org/10.1016/j.tics.2013.09.017
Lovett ML, Nieland TJF, Dingle YTL, Kaplan DL (2020) Innovations in 3D tissue models of human brain physiology and diseases. Adv Func Mater. https://doi.org/10.1002/adfm.201909146
Defelipe J (2011) The evolution of the brain, the human nature of cortical circuits, and intellectual creativity. Front Neuroanat 5:29. https://doi.org/10.3389/fnana.2011.00029
Nestler EJ, Hyman SE (2010) Animal models of neuropsychiatric disorders. Nat Neurosci 13:1161–1169. https://doi.org/10.1038/nn.2647
Laubach M, Amarante LM, Swanson K, White SR (2018) What, if anything, is rodent prefrontal cortex? eNeuro. https://doi.org/10.1523/ENEURO.0315-18.2018
Bayes A, Collins MO, Croning MD et al (2012) Comparative study of human and mouse postsynaptic proteomes finds high compositional conservation and abundance differences for key synaptic proteins. PLoS ONE 7:e46683. https://doi.org/10.1371/journal.pone.0046683
Bozek K, Wei Y, Yan Z et al (2015) Organization and evolution of brain lipidome revealed by large-scale analysis of human, chimpanzee, macaque, and mouse tissues. Neuron 85:695–702. https://doi.org/10.1016/j.neuron.2015.01.003
Pinson A, Namba T, Huttner WB (2019) Malformations of human neocortex in development—their progenitor cell basis and experimental model systems. Front Cell Neurosci 13:305. https://doi.org/10.3389/fncel.2019.00305
Ransohoff RM (2018) All (animal) models (of neurodegeneration) are wrong. Are they also useful? J Exp Med 215:2955–2958. https://doi.org/10.1084/jem.20182042
Simmnacher K, Lanfer J, Rizo T et al (2019) Modeling cell-cell interactions in Parkinson’s disease using human stem cell-based models. Front Cell Neurosci 13:571. https://doi.org/10.3389/fncel.2019.00571
Lang C, Campbell KR, Ryan BJ et al (2019) Single-cell sequencing of iPSC-dopamine neurons reconstructs disease progression and identifies HDAC4 as a regulator of parkinson cell phenotypes. Cell Stem Cell 24(93–106):e6. https://doi.org/10.1016/j.stem.2018.10.023
Teixeira AI, Ilkhanizadeh S, Wigenius JA et al (2009) The promotion of neuronal maturation on soft substrates. Biomaterials 30:4567–4572. https://doi.org/10.1016/j.biomaterials.2009.05.013
Baker BM, Chen CS (2012) Deconstructing the third dimension: how 3D culture microenvironments alter cellular cues. J Cell Sci 125:3015–3024. https://doi.org/10.1242/jcs.079509
Centeno EGZ, Cimarosti H, Bithell A (2018) 2D versus 3D human induced pluripotent stem cell-derived cultures for neurodegenerative disease modelling. Mol Neurodegener 13:27. https://doi.org/10.1186/s13024-018-0258-4
Birgersdotter A, Sandberg R, Ernberg I (2005) Gene expression perturbation in vitro–a growing case for three-dimensional (3D) culture systems. Semin Cancer Biol 15:405–412. https://doi.org/10.1016/j.semcancer.2005.06.009
Tekin H, Simmons S, Cummings B et al (2018) Effects of 3D culturing conditions on the transcriptomic profile of stem-cell-derived neurons. Nat Biomed Eng 2:540–554. https://doi.org/10.1038/s41551-018-0219-9
D’Avanzo C, Aronson J, Kim YH et al (2015) Alzheimer’s in 3D culture: challenges and perspectives. BioEssays 37:1139–1148. https://doi.org/10.1002/bies.201500063
Watson PMD, Kavanagh E, Allenby G, Vassey M (2017) Bioengineered 3D glial cell culture systems and applications for neurodegeneration and neuroinflammation. SLAS Discov 22:583–601. https://doi.org/10.1177/2472555217691450
Choi SH, Kim YH, Hebisch M et al (2014) A three-dimensional human neural cell culture model of Alzheimer’s disease. Nature 515:274–278. https://doi.org/10.1038/nature13800
Zhang D, Pekkanen-Mattila M, Shahsavani M et al (2014) A 3D Alzheimer’s disease culture model and the induction of P21-activated kinase mediated sensing in iPSC derived neurons. Biomaterials 35:1420–1428. https://doi.org/10.1016/j.biomaterials.2013.11.028
Jo J, Xiao Y, Sun AX et al (2016) Midbrain-like organoids from human pluripotent stem cells contain functional dopaminergic and neuromelanin-producing neurons. Cell Stem Cell 19:248–257. https://doi.org/10.1016/j.stem.2016.07.005
Bolognin S, Fossepre M, Qing X et al (2018) 3D Cultures of Parkinson’s disease-specific dopaminergic neurons for high content phenotyping and drug testing. Adv Sci (Weinh) 6:1800927. https://doi.org/10.1002/advs.201800927
Kane KIW, Moreno EL, Hachi S et al (2019) Automated microfluidic cell culture of stem cell derived dopaminergic neurons. Sci Rep 9:1796. https://doi.org/10.1038/s41598-018-34828-3
Son MY, Sim H, Son YS et al (2017) Distinctive genomic signature of neural and intestinal organoids from familial Parkinson’s disease patient-derived induced pluripotent stem cells. Neuropathol Appl Neurobiol 43:584–603. https://doi.org/10.1111/nan.12396
Monzel AS, Smits LM, Hemmer K et al (2017) Derivation of human midbrain-specific organoids from neuroepithelial stem cells. Stem Cell Rep 8:1144–1154. https://doi.org/10.1016/j.stemcr.2017.03.010
Caiazza MC, Lang C, Wade-Martins R (2020) What we can learn from iPSC-derived cellular models of Parkinson’s disease. In: Björklund A, Cenci MA (eds) Recent advances in Parkinson’s disease, 1st edn. Elsevier, Amsterdam, NL, pp 3–25
Bouyer C, Chen P, Güven S et al (2016) A Bio-acoustic levitational (BAL) assembly method for engineering of multilayered, 3D brain-like constructs, using human embryonic stem cell derived neuro-progenitors. Adv Mater 28:161–167. https://doi.org/10.1002/adma.201503916
Tang-Schomer MD, White JD, Tien LW et al (2014) Bioengineered functional brain-like cortical tissue. Proc Natl Acad Sci 111:13811–13816. https://doi.org/10.1073/pnas.1324214111
Liaudanskaya V, Chung JY, Mizzoni C et al (2020) Modeling controlled cortical impact injury in 3D brain-like tissue cultures. Adv Healthc Mater 9:e2000122. https://doi.org/10.1002/adhm.202000122
Rouleau N, Cantley WL, Liaudanskaya V et al (2020) A long-living bioengineered neural tissue platform to study neurodegeneration. Macromol Biosci 20:e2000004. https://doi.org/10.1002/mabi.202000004
Sood D, Cairns DM, Dabbi JM et al (2019) Functional maturation of human neural stem cells in a 3D bioengineered brain model enriched with fetal brain-derived matrix. Sci Rep 9:17874. https://doi.org/10.1038/s41598-019-54248-1
Cairns DM, Rouleau N, Parker RN et al (2020) A 3D human brain-like tissue model of herpes-induced Alzheimer’s disease. Sci Adv 6:eaay8828. https://doi.org/10.1126/sciadv.aay8828
Zhuang P, Sun AX, An J et al (2018) 3D neural tissue models: from spheroids to bioprinting. Biomaterials 154:113–133. https://doi.org/10.1016/j.biomaterials.2017.10.002
Naahidi S, Jafari M, Logan M et al (2017) Biocompatibility of hydrogel-based scaffolds for tissue engineering applications. Biotechnol Adv 35:530–544. https://doi.org/10.1016/j.biotechadv.2017.05.006
Watanabe K, Nakamura M, Okano H, Toyama Y (2007) Establishment of three-dimensional culture of neural stem/progenitor cells in collagen Type-1 Gel. Restor Neurol Neurosci 25:109–117
Wang X, He J, Wang Y, Cui F-Z (2012) Hyaluronic acid-based scaffold for central neural tissue engineering. Interface Focus 2:278–291. https://doi.org/10.1098/rsfs.2012.0016
Sood D, Chwalek K, Stuntz E et al (2016) Fetal brain extracellular matrix boosts neuronal network formation in 3D bioengineered model of cortical brain tissue. ACS Biomater Sci Eng 2:131–140. https://doi.org/10.1021/acsbiomaterials.5b00446
Moxon SR, Corbett NJ, Fisher K et al (2019) Blended alginate/collagen hydrogels promote neurogenesis and neuronal maturation. Mater Sci Eng, C 104:109904. https://doi.org/10.1016/j.msec.2019.109904
Klein C, Westenberger A (2012) Genetics of Parkinson’s disease. Cold Spring Harb Perspect Med 2:a008888–a008888. https://doi.org/10.1101/cshperspect.a008888
Kumari U, Tan EK (2009) LRRK2 in Parkinson’s disease: genetic and clinical studies from patients. FEBS J 276:6455–6463. https://doi.org/10.1111/j.1742-4658.2009.07344.x
Harvey K, Outeiro TF (2019) The role of LRRK2 in cell signalling. Biochem Soc Trans 47:197–207. https://doi.org/10.1042/bst20180464
Martin I, Kim JW, Dawson VL, Dawson TM (2014) LRRK2 pathobiology in Parkinson’s disease. J Neurochem 131:554–565. https://doi.org/10.1111/jnc.12949
O’Hara DM, Pawar G, Kalia SK, Kalia LV (2020) LRRK2 and α-synuclein: distinct or synergistic players in Parkinson’s disease? Front Neurosci. https://doi.org/10.3389/fnins.2020.00577
Bieri G, Brahic M, Bousset L et al (2019) LRRK2 modifies α-syn pathology and spread in mouse models and human neurons. Acta Neuropathol 137:961–980. https://doi.org/10.1007/s00401-019-01995-0
Lu J, Wu M, Yue Z (2020) Autophagy and Parkinson’s disease. In: Le W (ed) Autophagy: biology and diseases: clinical science. Springer Singapore, Singapore, pp 21–51
Schapira AHV (2006) The importance of LRRK2 mutations in Parkinson disease. Arch Neurol 63:1225–1228. https://doi.org/10.1001/archneur.63.9.1225
Macías-García D, Periñán MT, Muñoz-Delgado L et al (2021) Serum lipid profile among sporadic and familial forms of Parkinson’s disease. npj Parkinson’s Dis 7:59. https://doi.org/10.1038/s41531-021-00206-6
Dehay B, Martinez-Vicente M, Caldwell GA et al (2013) Lysosomal impairment in Parkinson’s disease. Mov Disord 28:725–732. https://doi.org/10.1002/mds.25462
Barkhuizen M, Anderson DG, Grobler AF (2016) Advances in GBA-associated Parkinson’s disease—pathology, presentation and therapies. Neurochem Int 93:6–25. https://doi.org/10.1016/j.neuint.2015.12.004
Rockwood DN, Preda RC, Yucel T et al (2011) Materials fabrication from Bombyx mori silk fibroin. Nat Protoc 6:1612–1631. https://doi.org/10.1038/nprot.2011.379
Kim UJ, Park J, Kim HJ et al (2005) Three-dimensional aqueous-derived biomaterial scaffolds from silk fibroin. Biomaterials 26:2775–2785. https://doi.org/10.1016/j.biomaterials.2004.07.044
Paull D, Sevilla A, Zhou H et al (2015) Automated, high-throughput derivation, characterization and differentiation of induced pluripotent stem cells. Nat Methods 12:885–892. https://doi.org/10.1038/nmeth.3507
Kahler DJ, Ahmad FS, Ritz A et al (2013) Improved methods for reprogramming human dermal fibroblasts using fluorescence activated cell sorting. PLoS ONE 8:e59867. https://doi.org/10.1371/journal.pone.0059867
Qi Y, Zhang XJ, Renier N et al (2017) Combined small-molecule inhibition accelerates the derivation of functional cortical neurons from human pluripotent stem cells. Nat Biotechnol 35:154–163. https://doi.org/10.1038/nbt.3777
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Bajad SU, Lu W, Kimball EH et al (2006) Separation and quantitation of water soluble cellular metabolites by hydrophilic interaction chromatography-tandem mass spectrometry. J Chromatogr A 1125:76–88. https://doi.org/10.1016/j.chroma.2006.05.019
Smith CA, Want EJ, O’Maille G et al (2006) XCMS: processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal Chem 78:779–787. https://doi.org/10.1021/ac051437y
Alden N, Krishnan S, Porokhin V et al (2017) Biologically consistent annotation of metabolomics data. Anal Chem 89:13097–13104. https://doi.org/10.1021/acs.analchem.7b02162
Smith CA, O’Maille G, Want EJ et al (2005) METLIN: a metabolite mass spectral database. Ther Drug Monit 27:747–751. https://doi.org/10.1097/01.ftd.0000179845.53213.39
Wishart DS, Feunang YD, Marcu A et al (2018) HMDB 4.0: the human metabolome database for 2018. Nucleic Acids Res 46:D608–D617. https://doi.org/10.1093/nar/gkx1089
Johnson SG (2018) NIST standard reference database 1A v17. National Institute of Standards and Technology, Gaithersburg
Afgan E, Baker D, Batut B et al (2018) The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2018 update. Nucleic Acids Res 46:W537–W544. https://doi.org/10.1093/nar/gky379
Dobin A, Davis CA, Schlesinger F et al (2013) STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29:15–21. https://doi.org/10.1093/bioinformatics/bts635
Liao Y, Smyth GK, Shi W (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30:923–930. https://doi.org/10.1093/bioinformatics/btt656
Love M, Anders S, Huber W (2014) Differential analysis of count data–the DESeq2 package. Genome Biol 15(10):1186
Ritchie ME, Phipson B, Wu D et al (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47–e47. https://doi.org/10.1093/nar/gkv007
Wickham H (2016) ggplot2: elegant graphics for data analysis. New York
Fox J, Weisberg S (2011) Multivariate linear models in R. Thousand Oaks, CA, USA
Stephens M (2017) False discovery rates: a new deal. Biostatistics 18:275–294. https://doi.org/10.1093/biostatistics/kxw041
Yu G, Wang LG, Han Y, He QY (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287. https://doi.org/10.1089/omi.2011.0118
Supek F, Bosnjak M, Skunca N, Smuc T (2011) REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS ONE 6:e21800. https://doi.org/10.1371/journal.pone.0021800
Heberle H, Meirelles GV, da Silva FR et al (2015) InteractiVenn: a web-based tool for the analysis of sets through Venn diagrams. BMC Bioinform 16:169. https://doi.org/10.1186/s12859-015-0611-3
Cv M, Huynen M, Jaeggi D et al (2003) STRING: a database of predicted functional associations between proteins. Nucleic Acids Res 31:258–261. https://doi.org/10.1093/nar/gkg034
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Turei D, Korcsmaros T, Saez-Rodriguez J (2016) OmniPath: guidelines and gateway for literature-curated signaling pathway resources. Nat Methods 13:966–967. https://doi.org/10.1038/nmeth.4077
Choobdar S, Ahsen ME, Crawford J et al (2019) Assessment of network module identification across complex diseases. Nat Methods 16:843–852. https://doi.org/10.1038/s41592-019-0509-5
Kohler S, Bauer S, Horn D, Robinson PN (2008) Walking the interactome for prioritization of candidate disease genes. Am J Hum Genet 82:949–958. https://doi.org/10.1016/j.ajhg.2008.02.013
Devkota K, Murphy JM, Cowen LJ (2020) GLIDE: combining local methods and diffusion state embeddings to predict missing interactions in biological networks. Bioinformatics 36:i464–i473. https://doi.org/10.1093/bioinformatics/btaa459
Chong J, Xia J (2018) MetaboAnalystR: an R package for flexible and reproducible analysis of metabolomics data. Bioinformatics 34:4313–4314. https://doi.org/10.1093/bioinformatics/bty528
Barnes S, Benton HP, Casazza K et al (2016) Training in metabolomics research. II. Processing and statistical analysis of metabolomics data, metabolite identification, pathway analysis, applications of metabolomics and its future. J Mass Spectrom 51:535–548. https://doi.org/10.1002/jms.3780
Koch JC, Bitow F, Haack J et al (2015) Alpha-Synuclein affects neurite morphology, autophagy, vesicle transport and axonal degeneration in CNS neurons. Cell Death Dis 6:e1811. https://doi.org/10.1038/cddis.2015.169
Cheng F, Vivacqua G, Yu S (2011) The role of alpha-synuclein in neurotransmission and synaptic plasticity. J Chem Neuroanat 42:242–248. https://doi.org/10.1016/j.jchemneu.2010.12.001
Nicholson JK, Connelly J, Lindon JC, Holmes E (2002) Metabonomics: a platform for studying drug toxicity and gene function. Nat Rev Drug Discov 1:153–161. https://doi.org/10.1038/nrd728
Shao Y, Le W (2019) Recent advances and perspectives of metabolomics-based investigations in Parkinson’s disease. Mol Neurodegener 14:3. https://doi.org/10.1186/s13024-018-0304-2
La Manno G, Gyllborg D, Codeluppi S et al (2016) Molecular diversity of midbrain development in mouse, human, and stem cells. Cell 167:566-580.e19. https://doi.org/10.1016/j.cell.2016.09.027
Abbott RD, Kimmerling EP, Cairns DM, Kaplan DL (2016) Silk as a biomaterial to support long-term three-dimensional tissue cultures. ACS Appl Mater Interfaces 8:21861–21868. https://doi.org/10.1021/acsami.5b12114
Jia C, Qi H, Cheng C et al (2020) α-Synuclein negatively regulates Nurr1 expression through NF-κB-related mechanism. Front Mol Neurosci. https://doi.org/10.3389/fnmol.2020.00064
Awad O, Panicker LM, Deranieh RM et al (2017) Altered differentiation potential of Gaucher’s disease iPSC neuronal progenitors due to Wnt/β-catenin downregulation. Stem Cell Reports 9:1853–1867. https://doi.org/10.1016/j.stemcr.2017.10.029
Woodard CM, Campos BA, Kuo SH et al (2014) iPSC-derived dopamine neurons reveal differences between monozygotic twins discordant for Parkinson’s disease. Cell Rep 9:1173–1182. https://doi.org/10.1016/j.celrep.2014.10.023
Elkouris M, Kouroupi G, Vourvoukelis A et al (2019) Long non-coding RNAs associated with neurodegeneration-linked genes are reduced in Parkinson’s disease patients. Front Cell Neurosci. https://doi.org/10.3389/fncel.2019.00058
Chiba-Falek O, Lopez GJ, Nussbaum RL (2006) Levels of alpha-synuclein mRNA in sporadic Parkinson disease patients. Mov Disord 21:1703–1708. https://doi.org/10.1002/mds.21007
Rocha EM, De Miranda B, Sanders LH (2018) Alpha-synuclein: pathology, mitochondrial dysfunction and neuroinflammation in Parkinson’s disease. Neurobiol Dis 109:249–257. https://doi.org/10.1016/j.nbd.2017.04.004
Dale N, Frenguelli B (2009) Release of adenosine and ATP during ischemia and epilepsy. Curr Neuropharmacol 7:160–179. https://doi.org/10.2174/157015909789152146
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30
Frolkis A, Knox C, Lim E et al (2010) SMPDB: the small molecule pathway database. Nucleic Acids Res 38:D480–D487. https://doi.org/10.1093/nar/gkp1002
Nazario LR, da Silva RS, Bonan CD (2017) Targeting adenosine signaling in Parkinson’s disease: from pharmacological to non-pharmacological approaches. Front Neurosci. https://doi.org/10.3389/fnins.2017.00658
Huang W, Xu Y, Zhang Y et al (2019) Metabolomics-driven identification of adenosine deaminase as therapeutic target in a mouse model of Parkinson’s disease. J Neurochem 150:282–295. https://doi.org/10.1111/jnc.14774
Johansen KK, Wang L, Aasly JO et al (2009) Metabolomic profiling in LRRK2-related Parkinson’s disease. PLoS ONE 4:e7551. https://doi.org/10.1371/journal.pone.0007551
Garcia-Esparcia P, Hernandez-Ortega K, Ansoleaga B et al (2015) Purine metabolism gene deregulation in Parkinson’s disease. Neuropathol Appl Neurobiol 41:926–940. https://doi.org/10.1111/nan.12221
Engelender S, Kaminsky Z, Guo X et al (1999) Synphilin-1 associates with α-synuclein and promotes the formation of cytosolic inclusions. Nat Genet 22:110–114. https://doi.org/10.1038/8820
Alvarez-Castelao B, Castaño JG (2011) Synphilin-1 inhibits alpha-synuclein degradation by the proteasome. Cell Mol Life Sci 68:2643–2654. https://doi.org/10.1007/s00018-010-0592-3
Rott R, Szargel R, Haskin J et al (2011) α-Synuclein fate is determined by USP9X-regulated monoubiquitination. Proc Natl Acad Sci 108:18666–18671. https://doi.org/10.1073/pnas.1105725108
Nakamura T, Yamashita H, Takahashi T, Nakamura S (2001) Activated fyn phosphorylates α-synuclein at tyrosine residue 125. Biochem Biophys Res Commun 280:1085–1092. https://doi.org/10.1006/bbrc.2000.4253
Pampalakis G, Sykioti V-S, Ximerakis M et al (2017) KLK6 proteolysis is implicated in the turnover and uptake of extracellular alpha-synuclein species. Oncotarget 8:14502–14515. https://doi.org/10.18632/oncotarget.13264
Pronin AN, Morris AJ, Surguchov A, Benovic JL (2000) Synucleins are a novel class of substrates for G protein-coupled receptor kinases. J Biol Chem 275:26515–26522. https://doi.org/10.1074/jbc.M003542200
Waxman EA, Giasson BI (2011) Characterization of kinases involved in the phosphorylation of aggregated α-synuclein. J Neurosci Res 89:231–247. https://doi.org/10.1002/jnr.22537
Liu Y, Su Y, Wang X (2013) Phosphatidic acid-mediated signaling. In: Capelluto D (ed) Lipid-mediated protein signaling, 1st edn. Springer, Dordrecht, pp 159–176
Ishisaka M, Hara H (2014) The roles of diacylglycerol kinases in the central nervous system: review of genetic studies in mice. J Pharmacol Sci 124:336–343. https://doi.org/10.1254/jphs.13R07CR
Wood PL, Tippireddy S, Feriante J, Woltjer RL (2018) Augmented frontal cortex diacylglycerol levels in Parkinson’s disease and Lewy Body Disease. PLoS ONE 13:e0191815. https://doi.org/10.1371/journal.pone.0191815
Raben DM, Barber CN (2017) Phosphatidic acid and neurotransmission. Adv Biol Regul 63:15–21. https://doi.org/10.1016/j.jbior.2016.09.004
Wang X, Devaiah SP, Zhang W, Welti R (2006) Signaling functions of phosphatidic acid. Prog Lipid Res 45:250–278. https://doi.org/10.1016/j.plipres.2006.01.005
Conde MA, Alza NP, Iglesias González PA et al (2018) Phospholipase D1 downregulation by α-synuclein: Implications for neurodegeneration in Parkinson’s disease. Biochim Biophys Acta 1863:639–650. https://doi.org/10.1016/j.bbalip.2018.03.006
Bae EJ, Lee HJ, Jang YH et al (2014) Phospholipase D1 regulates autophagic flux and clearance of α-synuclein aggregates. Cell Death Differ 21:1132–1141. https://doi.org/10.1038/cdd.2014.30
Javoy-Agid F, Hirsch EC, Dumas S et al (1990) Decreased tyrosine hydroxylase messenger RNA in the surviving dopamine neurons of the substantia nigra in Parkinson’s disease: An in situ hybridization study. Neuroscience 38:245–253. https://doi.org/10.1016/0306-4522(90)90389-l
Simunovic F, Yi M, Wang Y et al (2009) Gene expression profiling of substantia nigra dopamine neurons: further insights into Parkinson’s disease pathology. Brain 132:1795–1809. https://doi.org/10.1093/brain/awn323
Fuchs J, Mueller JC, Lichtner P et al (2009) The transcription factor PITX3 is associated with sporadic Parkinson’s disease. Neurobiol Aging 30:731–738. https://doi.org/10.1016/j.neurobiolaging.2007.08.014
Nguyen Ha N, Byers B, Cord B et al (2011) LRRK2 mutant iPSC-derived DA neurons demonstrate increased susceptibility to oxidative stress. Cell Stem Cell 8:267–280. https://doi.org/10.1016/j.stem.2011.01.013
Donega V, Burm SM, van Strien ME et al (2019) Transcriptome and proteome profiling of neural stem cells from the human subventricular zone in Parkinson’s disease. Acta Neuropathol Commun 7:84. https://doi.org/10.1186/s40478-019-0736-0
Riley BE, Gardai SJ, Emig-Agius D et al (2014) Systems-based analyses of brain regions functionally impacted in Parkinson’s disease reveals underlying causal mechanisms. PLoS ONE 9:e102909. https://doi.org/10.1371/journal.pone.0102909
Kang SS, Ahn EH, Zhang Z et al (2018) α-Synuclein stimulation of monoamine oxidase-B and legumain protease mediates the pathology of Parkinson’s disease. EMBO J 37:e98878. https://doi.org/10.15252/embj.201798878
Henderson MX, Sengupta M, Trojanowski JQ, Lee VMY (2019) Alzheimer’s disease tau is a prominent pathology in LRRK2 Parkinson’s disease. Acta Neuropathol Commun. https://doi.org/10.1186/s40478-019-0836-x
Fujii C, Harada S, Ohkoshi N et al (1999) Association between polymorphism of the cholecystokinin gene and idiopathic Parkinson’s disease. Clin Genet 56:395–400. https://doi.org/10.1034/j.1399-0004.1999.560508.x
Wang J, Si Y-M, Liu Z-L, Yu L (2003) Cholecystokinin, cholecystokinin-A receptor and cholecystokinin-B receptor gene polymorphisms in Parkinson’s disease. Pharmacogenet Genom. https://doi.org/10.1097/00008571-200306000-00008
Virreira Winter S, Karayel O, Strauss MT et al (2021) Urinary proteome profiling for stratifying patients with familial Parkinson’s disease. EMBO Mol Med. https://doi.org/10.15252/emmm.202013257
Duke DC, Moran LB, Kalaitzakis ME et al (2006) Transcriptome analysis reveals link between proteasomal and mitochondrial pathways in Parkinson’s disease. Neurogenetics 7:139–148. https://doi.org/10.1007/s10048-006-0033-5
Demishtein A, Fraiberg M, Berko D et al (2017) SQSTM1/p62-mediated autophagy compensates for loss of proteasome polyubiquitin recruiting capacity. Autophagy 13:1697–1708. https://doi.org/10.1080/15548627.2017.1356549
B’Chir W, Maurin A-C, Carraro V et al (2013) The eIF2α/ATF4 pathway is essential for stress-induced autophagy gene expression. Nucleic Acids Res 41:7683–7699. https://doi.org/10.1093/nar/gkt563
Husnjak K, Elsasser S, Zhang N et al (2008) Proteasome subunit Rpn13 is a novel ubiquitin receptor. Nature 453:481–488. https://doi.org/10.1038/nature06926
Morato Torres CA, Wassouf Z, Zafar F et al (2020) The Role of Alpha-synuclein and other Parkinson’s genes in neurodevelopmental and neurodegenerative disorders. Int J Mol Sci 21:5724. https://doi.org/10.3390/ijms21165724
Zhang J-W, Ma Y-M, Jing L et al (2020) Synaptic remodeling and reduced expression of the transcription factors, HES1 and HES5, in the cortex neurons of cognitively impaired hyperhomocysteinemic mice. Pathology Res Pract 216:152953. https://doi.org/10.1016/j.prp.2020.152953
Mesman S, Smidt MP (2017) Tcf12 is involved in early cell-fate determination and subset specification of midbrain dopamine neurons. Front Mol Neurosci. https://doi.org/10.3389/fnmol.2017.00353
Konishi H, Ogawa T, Nakagomi S et al (2010) Id1, Id2 and Id3 are induced in rat melanotrophs of the pituitary gland by dopamine suppression under continuous stress. Neuroscience 169:1527–1534. https://doi.org/10.1016/j.neuroscience.2010.06.030
Cohlberg JA, Li J, Uversky VN, Fink AL (2002) Heparin and Other Glycosaminoglycans Stimulate the Formation of Amyloid Fibrils from α-Synuclein in Vitro†. Biochemistry 41:1502–1511. https://doi.org/10.1021/bi011711s
Presto J, Thuveson M, Carlsson P et al (2008) Heparan sulfate biosynthesis enzymes EXT1 and EXT2 affect NDST1 expression and heparan sulfate sulfation. Proc Natl Acad Sci 105:4751–4756. https://doi.org/10.1073/pnas.0705807105
Wang D, He J, Huang B et al (2020) Emerging role of the Hippo pathway in autophagy. Cell Death Dis. https://doi.org/10.1038/s41419-020-03069-6
Sahu MR, Mondal AC (2020) The emerging role of Hippo signaling in neurodegeneration. J Neurosci Res 98:796–814. https://doi.org/10.1002/jnr.24551
Jankovic J (2008) Parkinson’s disease and movement disorders: moving forward. Lancet Neurol 7:9–11. https://doi.org/10.1016/S1474-4422(07)70302-2
Sulzer D (2007) Multiple hit hypotheses for dopamine neuron loss in Parkinson’s disease. Trends Neurosci 30:244–250. https://doi.org/10.1016/j.tins.2007.03.009
Amirikia M, Shariatzadeh SMA, Jorsaraei SGA, Mehranjani MS (2018) Auto-fluorescence of a silk fibroin-based scaffold and its interference with fluorophores in labeled cells. Eur Biophys J 47:573–581. https://doi.org/10.1007/s00249-018-1279-1
Acknowledgements
We thank the National Institutes of Health and the National Science Foundation for financial support of this project, and the New York Stem Cell Foundation’s Global Stem Cell Array® team for generating and providing the hiPSC lines. The authors acknowledge the Tufts University High Performance Compute Cluster (https://it.tufts.edu/high-performance-computing) and the confocal microscopy lab core (National Institutes of Health S10 OD021624) which were utilized for the research reported in this paper. Additional support from the National Science Foundation came through the Tufts T-Tripods Institute from the Harnessing the Data Revolution “Big Idea” effort. We also thank Dr. Volha Liaudanskaya and Dr. Mattia Bonzanni for experimental and intellectual feedback, as well as Sydney Peters and Emily Kim for technical support.
Funding
NIH (P41EB027062) to DLK, NSF (1934553) to LJC, NSF (1337760) to KL.
Author information
Authors and Affiliations
Contributions
Conceptualization: NJF, TJFN, DLK; Methodology: NJF, YMG, YG, LJC; Formal analysis: NJF, RB, KD, LJC; Investigation: NJF, TJFN, DLK; Resources: ML, KL, YMG, GC, SN, DLK; Data curation: NJF, KD, RB, ML, KL; Original manuscript writing: NJF, DLK, TJFN; Supervision: TJFN, DLK; Funding acquisition: DLK, LJC. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
All work was approved by the Tufts Institutional Biosafety Committee.
Consent to participate
The skin biopsies used for the generation of hiPSCs were donated from consenting participants under a protocol covering use in research, approved by the Western Institutional Review Board (WIRB).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
18_2021_4047_MOESM1_ESM.tiff
Supplemental File S1 Representative fragmentation spectra generated from untargeted liquid chromatography tandem mass spectrometry analysis. Mass/charge (m/z) peaks were compared between experimental samples (top panels, representative samples shown) and purified standards (bottom panels), which confirmed the identities of (A) Adenosine, (B) adenine, and (C) adenosine 3',5'-cyclic monophosphate metabolites. All results were acquired in positive ionization mode
18_2021_4047_MOESM2_ESM.tiff
Supplemental File S2 Principle component analysis of transcriptomic data of control, GBA and LRRK2 DA neurons. (A) 3D culture is consistently shifted along the PC1 and PC2 axes compared to 2D, indicating a culture condition dependent shift in global gene expression. Panel (B) highlights the results for the individual groups
18_2021_4047_MOESM3_ESM.xlsx
Supplemental File S3 Differential expression results using the DESeq2 R package, sorted by PD comparison in 2D and 3D culture format. A differential expression cutoff of abs(Log2(Fold change) > 0.585 in combination with q < 0.01 was used to determine significance
18_2021_4047_MOESM4_ESM.xlsx
Supplemental File S4 Over-representation analysis of biological processes when comparing heathy hiPSC-DA neurons cultured in 3D versus 2D conditions (3D CTRL versus 2D CTRL). Upregulated and downregulated GO terms were determined using differentially expressed genes (q < 0.01) with positive and negative fold change, respectively
18_2021_4047_MOESM5_ESM.xlsx
Supplemental File S5 Lists of the top 100 differentially expressed genes (HGNC symbols) from each Parkinson’s disease comparison that also existed in the DREAM3 molecular interaction network. Each list was utilized as the initial seed network for candidate disease gene prioritization and subsequent link prediction analysis
18_2021_4047_MOESM6_ESM.xlsx
Supplemental File S6 List of genes and molecular interactions for each link prediction network generated from PD genotype comparisons. The top 100 differentially expressed genes (DEGs), that also existed in the molecular interaction network DREAM3 (Supplemental file S5), were utilized to generate individual link prediction networks from all disease comparisons made in 2D and 3D culture formats: LRRK2 vs CTRL, GBA_PD vs CTRL, and GBA_PD vs GBA_CTRL. Application of Kohler’s random walk with restart nominated candidate disease genes (CDGs) within the neighborhood of the DEGs. Likely molecular interactions between DEGs and CDGs were computationally predicted by using global and local integrated diffusion embedding (GLIDE)
18_2021_4047_MOESM7_ESM.tiff
Supplemental File S7 α-synuclein protein measurements in 2D and 3D cultures (with reference to Fig. 3). α-synuclein protein levels (ELISA) in (A) 2D and (B) 3D culture conditions were determined from individual cell donor lines. (C) Fold change of α-synuclein protein levels was normalized to results from healthy individual cultures indicating increased expression in 3D cultures relative to the 2D culture. Statistical significance for (D) 2D and (E) 3D cultures were determined with one-way ANOVA followed by Tukey’s multiple comparison testing. (F) Fold change of α-synuclein protein levels (C) were compared across 2D and 3D culture systems by multiple t-tests with the two-stage step-up procedure of Benjamini, Krieger, and Yekutieli. Six or eight individual cultures were collected from two independent experiments, depending on the sample. A false discovery rate (q value or adjusted p value) of less than 0.05 was considered significant
18_2021_4047_MOESM8_ESM.tiff
Supplemental File S8 Neuronal network quantifications of TH+ and TUJ1+ signal ratios from immunofluorescent images suggested similar dopamine neuron content across individual donor lines and culture conditions. Statistical testing was performed using a two-way ANOVA with a significance cutoff of p = 0.05. Data displayed as the mean ± SEM from n = 4 replicate cultures per group
18_2021_4047_MOESM9_ESM.tiff
Supplemental File S9 Representative immunofluorescent images of 3D construct seeded with patient derived dopaminergic neurons. Wide-field images show homogeneously attached cells stained with DAPI (nuclei, blue) onto the scaffolding and neuronal processes (β3-tubulin, green) extending along the scaffolding and into the surrounding collagen hydrogel. To account for the autofluorescence of the silk scaffolding [147], digitally expanded images provide a clarified view of individual nuclei and neurites. Scale bars represent 500 μm
18_2021_4047_MOESM10_ESM.pdf
Supplemental File S10 Gene expression levels for neuronal, glial, and pluripotency markers. The low expression levels of markers for astrocytes, microglia, oligodendrocytes, and hiPSCs suggested that each groups’ culture composition were primarily neuronal
18_2021_4047_MOESM11_ESM.pdf
Supplemental File S11 3D CTRL versus 2D CTRL differential expression datasets for human dopamine neuron gene sets (hDA0, hDA1, and hDA2, characterized from developing human brains [93]). The 3D system upregulated a higher number of dopamine neuron genes, suggesting a more in vivo-like phenotype compared to 2D culture. Genes were highlighted blue if upregulated in 3D systems (log2(Fold Change) > 0.585 and p.adj < 0.05) or red if upregulated in 2D systems (log2(Fold Change) < 0.585 and p.adj < 0.05). Genes unaffected by culture condition were colored gray
18_2021_4047_MOESM12_ESM.xlsx
Supplemental File S12 Differentially expressed genes that are unique or shared between 2D and 3D cultures for each of the LRRK2 and GBA genotype comparisons (with reference to Fig. 6A, 6B). Differential expression analysis was performed with the DESeq2 R package and statistical significance was determined with a cutoff of abs(Log2(Fold change) > 0.585 in combination with q < 0.01
18_2021_4047_MOESM13_ESM.pdf
Supplemental File S13 Log2(Fold Change) of differentially expressed genes with a STRING association score to α-synuclein (with reference to Fig. 6C, Fig. 6D). Differential expression analysis was performed with the DESeq2 R package and statistical significance was determined with a cutoff of abs(Log2(Fold change) > 0.585 in combination with q < 0.01
18_2021_4047_MOESM14_ESM.pdf
Supplemental File S14 Log2(Fold Change) of differentially expressed genes in link prediction networks (with reference to Fig. 7). Differential expression analysis was performed with the DESeq2 R package and statistical significance was determined with a cutoff of abs(Log2(Fold change) > 0.585 in combination with q < 0.01
18_2021_4047_MOESM15_ESM.pdf
Supplemental File S15 Link prediction networks unique to 2D or 3D culture systems generated using differential expression data. The top 100 differentially expressed genes (DEGs), that also existed in the molecular interaction network DREAM3 (Supplemental file S5), were utilized to generate individual link prediction networks from all disease comparisons made in 2D and 3D culture formats: LRRK2 vs CTRL, GBA_PD vs CTRL, and GBA_PD vs GBA_CTRL. Application of Kohler’s random walk with restart nominated candidate disease genes (CDGs) within the neighborhood of the DEGs. Molecular interactions between DEGs and CDGs were computationally predicted using global and local integrated diffusion embedding (GLIDE). Networks unique to each culture condition were extracted for GBA or LRRK2 genotype comparisons. Genes with a green frame were commonly observed across GBA and LRRK2 genotype comparisons for the given culture condition
Rights and permissions
About this article
Cite this article
Fiore, N.J., Ganat, Y.M., Devkota, K. et al. Bioengineered models of Parkinson’s disease using patient-derived dopaminergic neurons exhibit distinct biological profiles in a 3D microenvironment. Cell. Mol. Life Sci. 79, 78 (2022). https://doi.org/10.1007/s00018-021-04047-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-021-04047-7