Abstract
Although brain tumours have been documented and recorded since the nineteenth century, 2016 marked 90 years since Percival Bailey and Harvey Cushing coined the term “glioblastoma multiforme”. Since that time, although extensive developments in diagnosis and treatment have been made, relatively little improvement on prognosis has been achieved. The resilience of GBM thus makes treating this tumour one of the biggest challenges currently faced by neuro-oncology. Aggressive and robust development, coupled with difficulties of complete resection, drug delivery and therapeutic resistance to treatment are some of the main issues that this nemesis presents today. Current treatments are far from satisfactory with poor prognosis, and focus on palliative management rather than curative intervention. However, therapeutic research leading to developments in novel treatment stratagems show promise in combating this disease. Here we present a review on GBM, looking at the history and advances which have shaped neurosurgery over the last century that cumulate to the present day management of GBM, while also exploring future perspectives in treatment options that could lead to new treatments on the road to a cure.

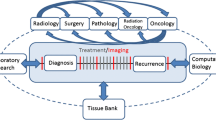
Adapted from Ref. [30]

Similar content being viewed by others
Abbreviations
- BBB:
-
Blood brain barrier
- BCNU:
-
Carmustine
- CCNU:
-
Lomustine
- CED:
-
Convection enhanced delivery
- CNS:
-
Central nervous system
- CSCs:
-
Cancer Stem Cells
- DNA:
-
Deoxyribonucleic acid
- EGFR:
-
Epidermal Growth Factor Receptor
- FDA:
-
US Food and Drug Administration
- GBM:
-
Glioblastoma
- MGMT:
-
O 6-methylguanine-DNA-methyltransferase
- MRI:
-
Magnetic resonance imaging
- PARP:
-
Poly ADP ribose polymerase
- PARPis:
-
Poly ADP ribose polymerase inhibitors
- p53:
-
Tumour suppressor protein 53
- RT:
-
Radiotherapy
- siRNA:
-
Small interfering Ribonucleic Acid
- TMZ:
-
Temozolomide
References
Jones E (1999) Colgi, Cajal and the neuron doctrine. J Hist Neurosci 8(2):170–178
Bock O (2013) Cajal, Golgi, Nansen, Schäfer and the neuron doctrine. Endeavour 37(4):228–234
DeAngelis L, Mellinghoff I (2011) Virchow 2011 or how to ID(H) human glioblastoma. J Clin Oncol 29(34):4473–4474
Scherer H (1940) A critical review: the pathology of cerebral gliomas. J Neurol. Psychiatry 3(2):147–177
Baker Fn (1993) The Massachusetts General Hospital. Early history and neurosurgery to 1939. J Neurosurg 79(6):948–959
Lister J (1867) On the antiseptic principle in the practice of surgery. The British Medical Journal 2:246–248
Kerr P, Caputy A, Horwitz N (2005) A history of cerebral localization. Neurosurg Focus 18 (4):e1
Greenblatt S (1997) The crucial decade: modern neurosurgery’s definitive development in Harvey Cushing’s early research and practice, 1900 to 1910. J Neurosurg 87(6):964–971
Macewen W (1881) Intra-cranial lesions: illustrating some points in connexion with the localisation of cerebral affections and the advantages of antiseptic trephining. Lancet 118(3031):581–583
Greenblatt S (2007) A surgeon for the brain. Brain 130(1):303–306
Macewen W (1879) Tumour of the dura mater; convulsions; removal of tumour by trephining; recovery. Glasgow Med J 12:208–213
Bennet A, Godlee R (1885) Case of a cerebral tumour. Med Chir Trans 68:243–275
Kirkpatrick D (1984) The first primary brain-tumor operation. J Neurosurg 61(5):809–813
Greenblatt S (2003) Harvey Cushing’s paradigmatic contribution to neurosurgery and the evolution of his thoughts about specialization. Bull Hist Med 77(4):789–822
Horsley V (1887) Remarks on ten consecutive cases of operations upon the brain and cranial cavity to illustrate the details and safety of the method employed. Br Med J 1(1373):863–865
Horsley V (1886) Brain surgery. Br Med J 2(1345):670–675
Tan T, Black P (2002) Sir Victor Horsley (1857–1916): pioneer of neurological surgery. Neurosurgery 50(3):607–611
Toledo-Pereyra L (2009) X-rays surgical revolution. J Invest Surg 22(5):327–332
Voorhees J, Cohen-Gadol A, Spencer D (2005) Early evolution of neurological surgery: conquering increased intracranial pressure, infection, and blood loss. Neurosurg Focus 18(4):e2
Dandy W (1918) Ventriculography following the injection of air into the cerebral ventricles. Ann Surg 68(1):5–11
Ferguson S, Lesniak M (2005) Percival Bailey and the classification of brain tumors. Neurosurg Focus 18(4):e7
Bailey O (1985) Genesis of the Percival Bailey-Cushing classification of gliomas. Pediatr Neurosci 12(4–5):261–265
Globus J, Strauss I (1925) Spongioblastoma multiforme. A primary malignant from of brain neoplasm: its clinical and anatomic features. Arch Neurol Psychiatr 14 (2):139–191
Bailey P, Cushing H (1925) Medulloblastoma Cerebelli. A common type of midcerebellar glioma of childhood. Arch Neurol Psychiatr 14 (2):192–224
Bailey P, Cushing H (1926) A classification of the tumors of the Glioma group on histogenetic basis with correlated study of prognosis. JB Lipponcott, Philadelphia
Scheithauer B (2008) Development of the WHO classification of tumors of the central nervous system: a historical perspective. Brain Pathol 19(4):551–564
Canale D, Longo L (1990) Harvey Cushing and pediatric neurosurgery. Neurosurgery 27(4):602–610
Bailey P (1927) Further remarks concerning tumors of the glioma group. Bull Johns Hopkins Hosp 40:354–389
Bradač G, Büll U, Fahlbusch R, Grumme T, Kazner E, Kretzschmar K, Lanksch W, Meese W, Schramm J, Steinhoff H, Stochdorph O, Wende S (1982) Computed tomography in intracranial tumors: differential diagnosis and clinical aspects. Springer, Berlin
Bailey P (1933) Intracranial tumors. C.C. Thomas, Springfield
Penfield W (1931) The classification of gliomas and neuroglia cell types. Arch Neurol Psychiatr 26 (4):745–753
Zülch K (1981) Historical development of the classification of brain tumours and the new proposal of the World Health Organization (WHO). Neurosurg Rev 4(3):123–127
Ohgaki H, Kleihues P (2013) The definition of primary and secondary glioblastoma. Clin Cancer Res 19(4):764–772
Peiffer J, Kleihues P (1999) Hans-Joachim Scherer (1906–1945), pioneer in glioma research. Brain Pathol 9(2):241–245
Scherer H (1940) Cerebral astrocytomas and their derivatives. Am J Cancer 40(2):159–198
Lee K, Choe G, Nam K, Seo A, Yun S, Kim K, Cho H, Park S (2013) Immunohistochemical classification of primary and secondary glioblastomas. Korean J Pathol 47(6):541–548
Spiegel E, Wycis H, Marks M, Lee A (1947) Stereotaxic apparatus for operations on the human brain. Science 106(2754):349–350
Uluç K, Kujoth G, Başkaya M (2009) Operating microscopes: past, present, and future. Neurosurg Focus 27(3):e4
Sachs S (1950) The problem of glioblastomas. J Neurosurg 7(3):185–189
Zülch K (1986) Brain tumours. Their biology and pathology. 3rd edn. Springer, Berlin
Netsky M, August B, Fowler W (1950) The longevity of patients with glioblastoma multiforme. J Neurosurg 7(3):261–269
Delgado-López P, Corrales-García E (2016) Survival in glioblastoma: a review on the impact of treatment modalities. Clin Transl Oncol 18(11):1062–1072
Mut M, Lopes M, Shaffrey M (2005) Lucien J. Rubinstein: enduring contributions to neuro-oncology. Neurosurg Focus 18(4):e8
Roth J, Elvidge A (1960) Glioblastoma multiforme: a clinical survey. J Neurosurg 17(4):736–750
Lindquist C (1995) Gamma knife radiosurgery. Semin Radiat Oncol 5(3):197–202
Yanagihara T, Saadatmand H, Wang T (2016) Reevaluating stereotactic radiosurgery for glioblastoma: new potential for targeted dose-escalation. J Neurooncol 130:397–411. doi:10.1007/s11060-016-2270-2
Hounsfield G (1973) Computerized transverse axial scanning (tomography). 1. Description of system. Br J Radiol 46(552):1016–1022
Lauterbur P (1973) Image formation by induced local interactions: examples of employing nuclear magnetic resonance. Nature 242:190–191
Daumas-Duport C, Scheithauer B, O’Fallon J, Kelly P (1988) Grading of astrocytomas. A simple and reproducible method. Cancer 62(10):2152–2165
Zülch K (1980) Principles of the new World Health Organization (WHO) classification of brain tumors. Neuroradiology 19(2):59–66
Buckner J, Brown P, O’Neill B, Meyer F, Wetmore C, Uhm J (2007) Central nervous system tumors. Mayo Clin Proc 82(10):1271–1286
Agnihotri S, Burrell K, Wolf A, Jalali S, Hawkins C, Rutka J, Zadeh G (2013) Glioblastoma, a brief review of history, molecular genetics, animal models and novel therapeutic strategies. Arch Immunol Ther Exp (Warsz) 61(1):25–41
Dandy W (1928) Removal of right cerebral hemisphere for certain tumors with hemiplegia: preliminary report. J Am Med Assoc 90(11):823–825
Wright J, Chugh J, Wright C, Alonso F, Hdeib A, Gittleman H, Barnholtz-Sloan J, Sloan A (2016) Laser interstitial thermal therapy followed by minimal-access transsulcal resection for the treatment of large and difficult to access brain tumors. Neurosurg Focus 41(4):e14
Hervey-Jumper S, Berger M (2016) Maximizing safe resection of low- and high-grade glioma. J Neurooncol 130:269–282
Yong R, Lonser R (2011) Surgery for glioblastoma multiforme: striking a balance. World Neurosurg 76 (6):528–530
Young R, Jamshidi A, Davis G, Sherman J (2015) Current trends in the surgical management and treatment of adult glioblastoma. Ann Transl Med 3(9):121
Smets T, Lawson T, Grandin C, Jankovski A, Raftopoulos C (2013) Immediate post-operative MRI suggestive of the site and timing of glioblastoma recurrence after gross total resection: a retrospective longitudinal preliminary study. Eur Radiol 23(6):1467–1477
Mirimanoff R, Gorlia T, Mason W, van den Bent M, Kortmann R, Fisher B, Reni M, Brandes A, Curschmann J, Villa S, Cairncross G, Allgeier A, Lacombe D, Stupp R (2006) Radiotherapy and temozolomide for newly diagnosed glioblastoma: recursive partitioning analysis of the EORTC 26981/22981-NCIC CE3 phase III randomized trial. J Clin Oncol 24(16):2563–2569
Patel D, Agarwal N, Tomei K, Hansberry D, Goldstein I (2015) Optimal timing of whole-brain radiation therapy following craniotomy for cerebral malignancies. World Neurosurg 84 (2):412–419
Stupp R, Brada M, van den Bent M, Tonn J, Pentheroudakis G (2014) High-grade glioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 25(3):93–101
Corso C, Bindra R (2016) Success and failures of combined modalities in glioblastoma multiforme: old problems and new directions. Semin Radiat Oncol 26(4):281–298
Ehrlich P (1913) Address in pathology on chemotherapeutics: scientific principles, methods, and results. Lancet 2(5694):445–451
Krumbhaar E, Krunbhaar H (1919) The blood and bone barrow in yellow cross gas (mustard gas) poisoning. J Med Res 40(3):497–508
Krumbhaar E (1919) Rôle of the blood and bone marrow in certain forms of gas poisoning: I. Peripheral blood changes and their significance. J Am Med Assoc 72(1):39–41
Fenn J, Udelsman R (2011) First use of intravenous chemotherapy cancer treatment: rectifying the record. J Am Coll Surg 212(3):413–417
Thomas R, Recht L, Nagpal S (2013) Advances in the management of glioblastoma: the role of temozolomide and MGMT testing. Clin Pharmacol 5:1–9
Connors T (1996) Anticancer drug development: the way forward. Oncologist 1(3):180–181
Deeken J, Löscher W (2007) The blood–brain barrier and cancer: transporters, treatment, and Trojan horses. Clin Cancer Res 13(6):1663–1674
Rubin P, Gash D, Hansen J, Nelson D, Williams J (1994) Disruption of the blood–brain barrier as the primary effect of CNS irradiation. Radiother Oncol 31(1):51–60
Muldoon L, Soussain C, Jahnke K, Johanson C, Siegal T, Smith Q, Hall W, Hynynen K, Senter P, Peereboom D, Neuwelt E (2007) Chemotherapy delivery issues in central nervous system malignancy: a reality check. J Clin Oncol 25(16):2295–2305
Cohen M, Shen Y, Keegan P, Pazdur R (2009) FDA drug approval summary: bevacizumab (Avastin) as treatment of recurrent glioblastoma multiforme. Oncologist 14(11):1131–1138
Ashby L, Smith K, Stea B (2016) Gliadel wafer implantation combined with standard radiotherapy and concurrent followed by adjuvant temozolomide for treatment of newly diagnosed high-grade glioma: a systematic literature review. World J Surg Oncol 14(1):225
Westphal M, Hilt D, Bortey E, Delavault P, Olivares R, Warnke P, Whittle I, Jääskeläinen J, Ram Z (2003) A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro-Oncology 5 (2):79–88
Bock H, Puchner M, Lohmann F, Schütze M, Koll S, Ketter R, Buchalla R, Rainov N, Kantelhardt S, Rohde V, Giese A (2010) First-line treatment of malignant glioma with carmustine implants followed by concomitant radiochemotherapy: a multicenter experience. Neurosurg Rev 33(4):441–449
Brem H, Piantadosi S, Burger P, Walker M, Selker R, Vick N, Black K, Sisti M, Brem S, Mohr G, Muller P, Morawetz R, Clifford Schold S, Group P-BTT (1995) Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet 345(8956):1008–1012
Borner M, Scheithauer W, Twelves C, Maroun J, Wilke H (2001) Answering patients’ needs: oral alternatives to intravenous therapy. Oncologist 6(Suppl 4):12–16
Liu G, Franssen E, Fitch M, Warner E (1997) Patient preferences for oral versus intravenous palliative chemotherapy. J Clin Oncol 15(1):110–115
Brada M, Stenning S, Gabe R, Thompson L, Levy D, Rampling R, Erridge S, Saran F, Gattamaneni R, Hopkins K, Beall S, Collins V, Lee S (2010) Temozolomide versus procarbazine, lomustine, and vincristine in recurrent high-grade glioma. J Clin Oncol 28(30):4601–4608
Silber J, Bobola M, Blank A, Chamberlain M (2012) O(6)-methylguanine-DNA methyltransferase in glioma therapy: promise and problems. Biochim Biophys Acta 1826(1):71–82
Prados M, Byron S, Tran N, Phillips J, Molinaro A, Ligon K, Wen P, Kuhn J, Mellinghoff I, de Groot J, Colman H, Cloughesy T, Chang S, Ryken T, Tembe W, Kiefer J, Berens M, Craig D, Carpten J, Trent J (2015) Toward precision medicine in glioblastoma: the promise and the challenges. Neuro-Oncology 17(8):1051–1063
Davies A, Weinberg U, Palti Y (2013) Tumor treating fields: a new frontier in cancer therapy. Ann N Y Acad Sci 1291:86–95
Taillibert S, Le Rhun E, Chamberlain M (2015) Tumor treating fields: a new standard treatment for glioblastoma? Curr Opin Neurol 28(6):659–664
Domingo-Musibay E, Galanis E (2015) What next for newly diagnosed glioblastoma? Future Oncol 11(24):3273–3283
Stupp R, Wong E, Kanner A, Steinberg D, Engelhard H, Heidecke V, Kirson E, Taillibert S, Liebermann F, Dbalý V, Ram Z, Villano J, Rainov N, Weinberg U, Schiff D, Kunschner L, Raizer J, Honnorat J, Sloan A, Malkin M, Landolfi J, Payer F, Mehdorn M, Weil R, Pannullo S, Westphal M, Smrcka M, Chin L, Kostron H, Hofer S, Bruce J, Cosgrove R, Paleologous N, Palti Y, Gutin P (2012) NovoTTF-100A versus physician’s choice chemotherapy in recurrent glioblastoma: a randomised phase III trial of a novel treatment modality. Eur J Cancer 48(14):2192–2202
Wick W, Weller M, van den Bent M, Stupp R (2010) Bevacizumab and recurrent malignant gliomas: a European perspective. J Clin Oncol 28(12):188–189
Desjardins A (2015) Neuro-oncology: what is the optimal use of bevacizumab in glioblastoma? Nat Rev Neurol 11(8):429–430
Gilbert M, Dignam J, Armstrong T, Wefel J, Blumenthal D, Vogelbaum M, Colman H, Chakravarti A, Pugh S, Won M, Jeraj R, Brown P, Jaeckle K, Schiff D, Stieber V, Brachman D, Werner-Wasik M, Tremont-Lukats I, Sulman E, Aldape K, Curran WJ, Mehta M (2014) A randomized trial of bevacizumab for newly diagnosed glioblastoma. N Engl J Med 370(8):699–708
Chinot O, Wick W, Mason W, Henriksson R, Saran F, Nishikawa R, Carpentier A, Hoang-Xuan K, Kavan F, Cernea D, Brandes A, Hilton M, Abrey L, Cloughesy T (2014) Bevacizumab plus radiotherapy-temozolomide for newly diagnosed glioblastoma. N Engl J Med 370(8):709–722
Wick W, Chinot O, Bendszus M, Mason W, Henriksson R, Saran F, Nishikawa R, Revil C, Kerloeguen Y, Cloughesy T (2016) Evaluation of pseudoprogression rates and tumor progression patterns in a phase III trial of bevacizumab plus radiotherapy/temozolomide for newly diagnosed glioblastoma. Neuro-Oncology 18 (10):1434–1441
Chinot O, Nishikawa R, Mason W, Henriksson R, Saran F, Cloughesy T, Garcia J, Revil C, Abrey L, Wick W (2016) Upfront bevacizumab may extend survival for glioblastoma patients who do not receive second-line therapy: an exploratory analysis of AVAglio. Neuro-Oncology 18(9):1313–1318
Rose S (2011) FDA pulls approval for Avastin in breast cancer. Cancer Discov 1(7):1–2
Kovic B, Xie F (2015) Economic evaluation of bevacizumab for the first-line treatment of newly diagnosed glioblastoma multiforme. J Clin Oncol 33(20):2296–2302
Campos B, Olsen L, Urup T, Poulsen H (2016) A comprehensive profile of recurrent glioblastoma. Oncogene 35:5819–5825
Sun H, Du S, Liao G, Xie X, Ren C, Yuan Y (2015) Do glioma patients derive any therapeutic benefit from taking a higher cumulative dose of temozolomide regimens? A meta-analysis. Medicine (Baltimore) 94(20):e827
Tosoni A, Franceschi E, Poggi R, Brandes A (2016) Relapsed glioblastoma: treatment strategies for initial and subsequent recurrences. Curr Treat Options Oncol 17(9):49
Yin A, Cheng J, Zhang X, Liu B (2013) The treatment of glioblastomas: a systematic update on clinical Phase III trials. Crit Rev Oncol Hematol 87(3):265–282
clinicaltrials.gov National Library of Medicine (US) Accessed 31 Oct 2016
Tavano L, Muzzalupo R (2016) Multi-functional vesicles for cancer therapy: the ultimate magic bullet. Colloids Surfaces B 147:161–171
Stupp R, Hegi M, Mason W, van den Bent M, Taphoorn M, Janzer R, Ludwin S, Allgeier A, Fisher B, Belanger K, Hau P, Brandes A, Gijtenbeek J, Marosi C, Vecht C, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross J, Mirimanoff R, Groups EOfRaToCBTaRO, Group NCIoCCT (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10(5):459–466
Scott J, Bauchet L, Fraum T, Nayak L, Cooper A, Chao S, Suh J, Vogelbaum M, Peereboom D, Zouaoui S, Mathieu-Daudé H, Fabbro-Peray P, Rigau V, Taillandier L, Abrey L, DeAngelis L, Shih J, Iwamoto F (2012) Recursive partitioning analysis of prognostic factors for glioblastoma patients aged 70 years or older. Cancer 118(22):5595–5600
Neagu M, Reardon D (2015) An update on the role of immunotherapy and vaccine strategies for primary brain tumors. Curr Treat Options Oncol 16(11):54
Sampson J, Mitchell D (2015) Vaccination strategies for neuro-oncology. Neuro-Oncology 17 (7):15–25
Ardon H, Van Gool S, Lopes I, Maes W, Sciot R, Wilms G, Demaerel P, Bijttebier P, Claes L, Goffin J, Van Calenbergh F, De Vleeschouwer S (2010) Integration of autologous dendritic cell-based immunotherapy in the primary treatment for patients with newly diagnosed glioblastoma multiforme: a pilot study. J Neurooncol 99(2):261–272
Schijns V, Pretto C, Devillers L, Pierre D, Hofman F, Chen T, Mespouille P, Hantos P, Glorieux P, Bota D, Stathopoulos A (2015) First clinical results of a personalized immunotherapeutic vaccine against recurrent, incompletely resected, treatment-resistant glioblastoma multiforme (GBM) tumors, based on combined allo- and auto-immune tumor reactivity. Vaccine 33(23):269–2696
Reijneveld J, Voest E, Taphoorn M (2000) Angiogenesis in malignant primary and metastatic brain tumors. J Neurol 247(8):597–608
Scaringi C, Enrici R, Minniti G (2013) Combining molecular targeted agents with radiation therapy for malignant gliomas. Onco Targets Ther 6:1079–1095
McGee M, Hamner J, Williams R, Rosati S, Sims T, Ng C, Gaber M, Calabrese C, Wu J, Nathwani A, Duntsch C, Merchant T, Davidoff A (2010) Improved intratumoral oxygenation through vascular normalization increases glioma sensitivity to ionizing radiation. Int J Radiat Oncol Biol Phys 76(5):1537–1545
Gupta V, Jaskowiak N, Beckett M, Mauceri H, Grunstein J, Johnson R, Calvin D, Nodzenski E, Pejovic M, Kufe D, Posner M, Weichselbaum R (2002) Vascular endothelial growth factor enhances endothelial cell survival and tumor radioresistance. Cancer J 8(1):47–54
Bolderson E, Richard D, Zhou B, Khanna K (2009) Recent advances in cancer therapy targeting proteins involved in DNA double-strand break repair. Clin Cancer Res 15(20):6314–6320
Rouleau M, Patel A, Hendzel M, Kaufmann S, Poirier G (2010) PARP inhibition: PARP1 and beyond. Nat Rev Cancer 10(4):293–301
Bryant H, Schultz N, Thomas H, Parker K, Flower D, Lopez E, Kyle S, Meuth M, Curtin N, Helleday T (2005) Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nature 343(7035):913–917
Farmer H, McCabe N, Lord C, Tutt A, Johnson D, Richardson T, Santarosa M, Dillon K, Hickson I, Knights C, Martin N, Jackson S, Smith G, Ashworth A (2005) Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434(7035):917–921
Powell C, Mikropoulos C, Kaye S, Nutting C, Bhide S, Newbold K, Harrington K (2010) Pre-clinical and clinical evaluation of PARP inhibitors as tumour-specific radiosensitisers. Cancer Treat Rev 36(7):566–575
Senra J, Telfer B, Cherry K, McCrudden C, Hirst D, O’Connor M, Wedge S, Stratford I (2011) Inhibition of PARP-1 by olaparib (AZD2281) increases the radiosensitivity of a lung tumor xenograft. Mol Cancer Ther 10(10):1949–1958
Bonnet D, Dick J (1997) Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nature Med 3(7):730–737
Galli R, Binda E, Orfanelli U, Cipelletti B, Gritti A, De Vitis S, Fiocco R, Foroni C, Dimeco F, Vescovi A (2004) Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 64(19):7011–7021
Beier D, Röhrl S, Pillai D, Schwarz S, Kunz-Schughart L, Leukel P, Proescholdt M, Brawanski A, Bogdahn U, Trampe-Kieslich A, Giebel B, Wischhusen J, Reifenberger G, Hau P, Beier C (2008) Temozolomide preferentially depletes cancer stem cells in glioblastoma. Cancer Res 68(14):5706–5715
Hegi M, Diserens A, Gorlia T, Hamou M, de Tribolet N, Weller M, Kros J, Hainfellner J, Mason W, Mariani L, Bromberg J, Hau P, Mirimanoff R, Cairncross J, Janzer R, Stupp R (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352(10):997–1003
Lathia J, Mack S, Mulkearns-Hubert E, Valentim C, Rich J (2015) Cancer stem cells in glioblastoma. Genes Dev 29(12):1203–1217
Gupta P, Onder T, Jiang G, Tao K, Kuperwasser C, Weinberg R, Lander E (2009) Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138(4):645–659
Qin L, Jia P, Zhang Z, Zhang S (2015) ROS-p53-cyclophilin-D signaling mediates salinomycin-induced glioma cell necrosis. J Exp Clin Canc Res 34:57
Calzolari A, Saulle E, De Angelis M, Pasquini L, Boe A, Pelacchi F, Ricci-Vitiani L, Baiocchi M, Testa U (2014) Salinomycin potentiates the cytotoxic effects of TRAIL on glioblastoma cell lines. PLoS One 9(4):e94438
Pardridge W (2007) Blood–brain barrier delivery. Drug Discov Today 12(1–2):54–61
Vogelbaum M, Aghi M (2015) Convection-enhanced delivery for the treatment of glioblastoma. Neuro-Oncology 17 (Suppl 2):3–8
Hynynen K, McDannold N, Vykhodtseva N, Jolesz F (2003) Non-invasive opening of BBB by focused ultrasound. Acta Neurochir Suppl 86:555–558
Choi J, Feshitan J, Baseri B, Wang S, Tung Y, Borden M, Konofagou E (2010) Microbubble-size dependence of focused ultrasound-induced blood–brain barrier opening in mice in vivo. IEEE Trans Bio-Med Eng 57(1):145–154
Liu H, Hsu P, Lin C, Huang C, Chai W, Chu P, Huang C, Chen P, Yang L, Kuo J, Wei K (2016) Focused ultrasound enhances central nervous system delivery of bevacizumab for malignant glioma treatment. Radiology 281(1):99–108
Carpentier A, Canney M, Vignot A, Reina V, Beccaria K, Horodyckid C, Karachi C, Leclercq D, Lafon C, Chapelon J, Capelle L, Cornu P, Sanson M, Hoang-Xuan K, Delattre J, Idbaih A (2016) Clinical trial of blood–brain barrier disruption by pulsed ultrasound. Sci Transl Med 8 (343):343re342
Sarkar G, Curran G, Sarkaria J, Lowe V, Jenkins R (2014) Peptide carrier-mediated non-covalent delivery of unmodified cisplatin, methotrexate and other agents via intravenous route to the brain. PLoS One 9(5):e97655
Allen T, Cullis P (2004) Drug delivery systems: entering the mainstream. Science 303(5665):1818–1822
Peer D, Karp J, Hong S, Farokhzad O, Margalit R, Langer R (2007) Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol 2(12):751–760
Gutkin A, Cohen Z, Peer D (2016) Harnessing nanomedicine for therapeutic intervention in glioblastoma. Expert Opin Drug Deliv 13(11):1573–1582
Kim S, Harford J, Pirollo K, Chang E (2015) Effective treatment of glioblastoma requires crossing the blood–brain barrier and targeting tumors including cancer stem cells: the promise of nanomedicine. Biochem Biophys Res Commun 468(3):485–489
Danhier F, Messaoudi K, Lemaire L, Benoit J, Lagarce F (2015) Combined anti-Galectin-1 and anti-EGFR siRNA-loaded chitosan-lipid nanocapsules decrease temozolomide resistance in glioblastoma: in vivo evaluation. Int J Pharm 481(1–2):154–161
Bastiancich C, Vanvarenberg K, Ucakar B, Pitorre M, Bastiat G, Lagarce F, Préat V, Danhier F (2016) Lauroyl-gemcitabine-loaded lipid nanocapsule hydrogel for the treatment of glioblastoma. J Control Release 225:283–293
Hoare T, DS K (2008) Hydrogels in drug delivery: progress and challenges. Polymer 49(8):1993–2007
Bastiancich C, Danhier P, Préat V, Danhier F (2016) Anticancer drug-loaded hydrogels as drug delivery systems for the local treatment of glioblastoma. J Control Release 243:29–42
Vellimana A, Recinos V, Hwang L, Fowers K, Li K, Zhang Y, Okonma S, Eberhart C, Brem H, Tyler B (2013) Combination of paclitaxel thermal gel depot with temozolomide and radiotherapy significantly prolongs survival in an experimental rodent glioma model. J Neurooncol 111(3):229–236
Fourniols T, Randolph L, Staub A, Vanvarenberg K, Leprince J, Préat V, des Rieux A, Danhier F (2015) Temozolomide-loaded photopolymerizable PEG-DMA-based hydrogel for the treatment of glioblastoma. J Control Release 210:95–104
Marchesi F, Turriziani M, Tortorelli G, Avvisati G, Torino F, De Vecchis L (2007) Triazene compounds: mechanism of action and related DNA repair systems. Pharmacol Res 56(4):275–287
Perazzoli G, Prados J, Ortiz R, Caba O, Cabeza L, Berdasco M, Gónzalez B, Melguizo C (2015) Temozolomide resistance in glioblastoma cell lines: implication of MGMT, MMR, P-glycoprotein and CD133 expression. PLoS One 10(10):e0140131
Weller M, Stupp R, Reifenberger G, Brandes A, van den Bent M, Wick W, Hegi M (2010) MGMT promoter methylation in malignant gliomas: ready for personalized medicine? Nat Rev Neurol 6(1):39–51
Parker N, Khong P, Parkinson J, Howell V, Wheeler H (2015) Molecular heterogeneity in glioblastoma: potential clinical implications. Front Oncol 5:55
Messaoudi K, Clavreul C, Danhier F, Saulnier P, Benoît J-P, Lagarce F (2015) Combined silencing expression of MGMT with EGFR or galectin-1 enhances the sensitivity of glioblastoma to temozolomide. Eur J Nanomed 7(2):97–107
Kaina B, Margison G, Christmann M (2010) Targeting O 6-methylguanine-DNA methyltransferase with specific inhibitors as a strategy in cancer therapy. Cell Mol Life Sci 67(21):3663–3681
Ramirez Y, Mladek A, Phillips R, Gynther M, Rautio J, Ross A, Wheelhouse R, Sakaria J (2015) Evaluation of novel imidazotetrazine analogues designed to overcome temozolomide resistance and glioblastoma regrowth. Mol Cancer Ther 14(1):111–119
Rich J, Bigner D (2004) Development of novel targeted therapies in the treatment of malignant glioma. Nat Rev Drug Discov 3(5):430–446
Verschuere T, Toelen J, Maes W, Poirier F, Boon L, Tousseyn T, Mathivet T, Gerhardt H, Mathieu V, Kiss R, Lefranc F, Van Gool S, De Vleeschouwer S (2014) Glioma-derived galectin-1 regulates innate and adaptive antitumor immunity. Int J Cancer 134(4):873–884
Le Mercier M, Fortin S, Mathieu V, Kiss R, Lefranc F (2010) Galectins and gliomas. Brain Pathol 20(1):17–27
Camby I, Decaestecker C, Lefranc F, Kaltner H, Gabius H, Kiss R (2005) Galectin-1 knocking down in human U87 glioblastoma cells alters their gene expression pattern. Biochem Biophys Res Commun 335(1):27–35
Acknowledgements
The authors are recipients of subsidies from the Fonds National de la Recherche Scientifique (FNRS), the Fonds Spéciaux de Recherche Scientifique (FSR, UCL), as well as the BEWARE Academia Programme (COFUND). The authors would also like to thank Professor Samuel H. Greenblatt for personal communication.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
J. Bianco and C. Bastiancich contributed equally.
Rights and permissions
About this article
Cite this article
Bianco, J., Bastiancich, C., Jankovski, A. et al. On glioblastoma and the search for a cure: where do we stand?. Cell. Mol. Life Sci. 74, 2451–2466 (2017). https://doi.org/10.1007/s00018-017-2483-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-017-2483-3