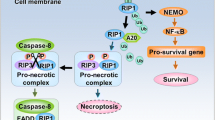
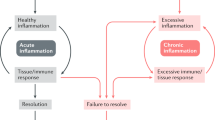
Abstract
Cell death is a major determinant of inflammatory disease severity. Whether cells live or die during inflammation largely depends on the relative success of the pro-survival process of autophagy versus the pro-death process of apoptosis. These processes interact and influence each other during inflammation and there is a checkpoint at which cells irrevocably commit to either one pathway or another. This review will discuss the concept of the autophagy/apoptosis checkpoint and its importance during inflammation, the mechanisms of inflammation leading up to the checkpoint, and how the checkpoint is regulated. Understanding these concepts is important since manipulation of the autophagy/apoptosis checkpoint represents a novel opportunity for treatment of inflammatory diseases caused by too much or too little cell death.

Similar content being viewed by others
Abbreviations
- PAMPs:
-
Pathogen-associated molecular pattern molecules
- LPS:
-
Lipopolysaccharide
- TNF-α:
-
Tumor necrosis factor alpha
- IL-1:
-
Interleukin-1
- IFN-γ:
-
Interferon-gamma
- DAMPs:
-
Damage-associated molecular pattern molecules
- HMGB1:
-
High mobility group box-1
- ATP:
-
Adenosine triphosphate
- FasL/CD95L:
-
Fas ligand
- TRAIL/Apo2L:
-
TNF-related apoptosis-inducing ligand
- TL1A:
-
TNF-like ligand 1A
- TLR:
-
Toll-like receptors
- NOD:
-
Nucleotide-oligomerization domain
- NLR:
-
NOD-like receptors
- AIM2:
-
Absent in melanoma-2
- ALRs:
-
AIM2-like receptors
- CLR:
-
C-type lectin receptors
- RIG-1:
-
Retinoic acid-inducible gene-I
- RLR:
-
RIG-I-like receptors
- TIR:
-
Toll/interleukin-1 receptor
- MyD88:
-
Myeloid differentiation primary-response protein 88
- MAPKs:
-
Mitogen-activated protein kinases
- NF-κB:
-
Nuclear factor-kappa B
- TRIF/TICAM1:
-
TIR-domain-containing adaptor protein inducing IFN-β
- MDA5:
-
Melanoma differentiation-associated gene 5
- LGP2:
-
Laboratory of genetics and physiology 2
- DD:
-
Death domain
- TRADD:
-
TNF-receptor-associated death domain
- FADD:
-
Fas-associated DD
- SOCS1:
-
Suppressor of cytokine signaling-1
- ER:
-
Endoplasmic reticulum
- PDI:
-
Protein disulfide isomerase
- UPR:
-
Unfolded protein response
- GRP78/BiP/HSPA5:
-
Glucose-regulated protein 78
- PERK:
-
PKR-like ER kinase
- IRE1:
-
Inositol requiring enzyme 1
- ATF6:
-
Activation transcription factor 6
- ERAD:
-
ER-associated degradation
- ROS:
-
Reactive oxygen species
- Nrf2:
-
NF-E2-related factor 2
- TNFAIP3/A20:
-
Tumor necrosis factor, alpha-induced protein 3
- mTOR:
-
Mammalian target of rapamycin
- CoA:
-
Coenzyme A
- S1P:
-
Sphingosine-1-phosphate
- PE:
-
Phosphatidylethanolamine
- CaMKK-β:
-
Calmodulin-dependent kinase kinase beta
- AMPK:
-
AMP kinase
- PCD:
-
Programmed cell death
- TRAF2:
-
TNF-receptor-associated factor 2
- cIAP1 or cIAP2:
-
Cellular inhibitors of apoptosis
- LUBAC:
-
Linear ubiquitin chain assembly complex
- TAK1:
-
TGF-activated kinase 1
- TAB:
-
TAK1-binding protein
- cFLIP:
-
Cellular FLICE-like inhibitory protein
- MLKL:
-
Mixed lineage kinase domain-like
- DISC:
-
Death-inducing signaling complex
- MOMP:
-
Mitochondrial outer membrane permeabilization
- Apaf-1:
-
Apoptosis protease activating factor-1
- BH3:
-
Bcl-2-homology 3
- Smac/DIABLO:
-
Second mitochondria-derived activator of caspase/direct IAP-binding protein with low PI
- HtrA2/OMI:
-
Serine protease high-temperature requirement protein A2
- IAPs:
-
Inhibitor of apoptosis proteins
- UVRAG:
-
UV irradiation resistance-associated gene
- DAPK:
-
Death-associated protein kinase
- DRP:
-
DAPK-related proteins kinase
References
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132:27–42. doi:10.1016/j.cell.2007.12.018
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35:495–516. doi:10.1080/01926230701320337
Levine B, Mizushima N, Virgin HW (2011) Autophagy in immunity and inflammation. Nature 469:323–335. doi:10.1038/nature09782
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147:728–741. doi:10.1016/j.cell.2011.10.026
Deretic V, Saitoh T, Akira S (2013) Autophagy in infection, inflammation and immunity. Nat Rev Immunol 13:722–737. doi:10.1038/nri3532
Boyle KB, Randow F (2013) The role of “eat-me” signals and autophagy cargo receptors in innate immunity. Curr Opin Microbiol. doi:10.1016/j.mib.2013.03.010
Sanjuan MA, Dillon CP, Tait SWG et al (2007) Toll-like receptor signalling in macrophages links the autophagy pathway to phagocytosis. Nature 450:1253–1257. doi:10.1038/nature06421
Lee HK, Lund JM, Ramanathan B et al (2007) Autophagy-dependent viral recognition by plasmacytoid dendritic cells. Science 315:1398–1401. doi:10.1126/science.1136880
Schmid D, Pypaert M, Münz C (2007) Antigen-loading compartments for major histocompatibility complex class II molecules continuously receive input from autophagosomes. Immunity 26:79–92. doi:10.1016/j.immuni.2006.10.018
Mintern JD, Macri C, Chin WJ et al (2015) Differential use of autophagy by primary dendritic cells specialized in cross-presentation. Autophagy 11:906–917. doi:10.1080/15548627.2015.1045178
Deretic V, Kimura T, Timmins G et al (2015) Immunologic manifestations of autophagy. J Clin Invest 125:75–84. doi:10.1172/JCI73945
White E (2015) The role for autophagy in cancer. J Clin Invest 125:42–46. doi:10.1172/JCI73941
Kenific CM, Debnath J (2015) Cellular and metabolic functions for autophagy in cancer cells. Trends Cell Biol 25:37–45. doi:10.1016/j.tcb.2014.09.001
Dong X, Levine B (2013) Autophagy and viruses: adversaries or allies? J Innate Immun 5:480–493. doi:10.1159/000346388
Deretic V, Singh S, Master S et al (2006) Mycobacterium tuberculosis inhibition of phagolysosome biogenesis and autophagy as a host defence mechanism. Cell Microbiol 8:719–727. doi:10.1111/j.1462-5822.2006.00705.x
Nogueira CV, Lindsten T, Jamieson AM et al (2009) Rapid pathogen-induced apoptosis: a mechanism used by dendritic cells to limit intracellular replication of Legionella pneumophila. PLoS Pathog 5:e1000478. doi:10.1371/journal.ppat.1000478
Singhera GK, Chan TS, Cheng JY et al (2006) Apoptosis of viral-infected airway epithelial cells limit viral production and is altered by corticosteroid exposure. Respir Res 7:78. doi:10.1186/1465-9921-7-78
Chaudhuri AR, Jeor SS, Maciejewski JP (1999) Apoptosis induced by human cytomegalovirus infection can be enhanced by cytokines to limit the spread of virus. Exp Hematol 27:1194–1203. doi:10.1016/S0301-472X(99)00044-2
Marriott HM, Bingle CD, Read RC et al (2005) Dynamic changes in Mcl-1 expression regulate macrophage viability or commitment to apoptosis during bacterial clearance. J Clin Invest 115:359–368. doi:10.1172/JCI21766
Grassmé H, Kirschnek S, Riethmueller J et al (2000) CD95/CD95 ligand interactions on epithelial cells in host defense to Pseudomonas aeruginosa. Science 290:527–530
Flannagan RS, Cosío G, Grinstein S (2009) Antimicrobial mechanisms of phagocytes and bacterial evasion strategies. Nat Rev Microbiol 7:355–366. doi:10.1038/nrmicro2128
Wu Y-S, Chen S-N (2014) Apoptotic cell: linkage of inflammation and wound healing. Front Pharmacol 5:1. doi:10.3389/fphar.2014.00001
Kanaly ST, Nashleanas M, Hondowicz B, Scott P (1999) TNF receptor p55 is required for elimination of inflammatory cells following control of intracellular pathogens. J Immunol 163:3883–3889
Beattie L, d’El-Rei Hermida M, Moore JWJ et al (2013) A transcriptomic network identified in uninfected macrophages responding to inflammation controls intracellular pathogen survival. Cell Host Microbe 14:357–368. doi:10.1016/j.chom.2013.08.004
Vereecke L, Beyaert R, van Loo G (2011) Enterocyte death and intestinal barrier maintenance in homeostasis and disease. Trends Mol Med 17:584–593. doi:10.1016/j.molmed.2011.05.011
Bantel H, Schulze-Osthoff K (2003) Apoptosis in hepatitis C virus infection. Cell Death Differ 10(Suppl 1):S48–S58. doi:10.1038/sj.cdd.4401119
Rangel SM, Diaz MH, Knoten CA et al (2015) The role of ExoS in dissemination of Pseudomonas aeruginosa during Pneumonia. PLoS Pathog 11:e1004945. doi:10.1371/journal.ppat.1004945
Badley AD, Pilon AA, Landay A, Lynch DH (2000) Mechanisms of HIV-associated lymphocyte apoptosis. Blood 96:2951–2964
Selliah N, Finkel TH (2001) Biochemical mechanisms of HIV induced T cell apoptosis. Cell Death Differ 8:127–136. doi:10.1038/sj.cdd.4400822
Rodríguez-Grille J, Busch LK, Martínez-Costas J, Benavente J (2014) Avian reovirus-triggered apoptosis enhances both virus spread and the processing of the viral nonstructural muNS protein. Virology 462–463:49–59. doi:10.1016/j.virol.2014.04.039
Chung KF, Adcock IM (2008) Multifaceted mechanisms in COPD: inflammation, immunity, and tissue repair and destruction. Eur Respir J 31:1334–1356. doi:10.1183/09031936.00018908
Mogensen TH (2009) Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev 22:240–273. doi:10.1128/CMR.00046-08
Cha H-H, Hwang JR, Kim H-Y et al (2014) Autophagy induced by tumor necrosis factor α mediates intrinsic apoptosis in trophoblastic cells. Reprod Sci 21:612–622. doi:10.1177/1933719113508816
Jia G, Cheng G, Gangahar DM, Agrawal DK (2006) Insulin-like growth factor-1 and TNF-alpha regulate autophagy through c-jun N-terminal kinase and Akt pathways in human atherosclerotic vascular smooth cells. Immunol Cell Biol 84:448–454. doi:10.1111/j.1440-1711.2006.01454.x
Keller CW, Fokken C, Turville SG et al (2010) TNF-induces macroautophagy and regulates MHC class II expression in human skeletal muscle cells. J Biol Chem 286:3970–3980. doi:10.1074/jbc.M110.159392
Shi C-S, Shenderov K, Huang N-N et al (2012) Activation of autophagy by inflammatory signals limits IL-1β production by targeting ubiquitinated inflammasomes for destruction. Nat Immunol 13:255–263. doi:10.1038/ni.2215
de Luca A, Smeekens SP, Casagrande A et al (2014) IL-1 receptor blockade restores autophagy and reduces inflammation in chronic granulomatous disease in mice and in humans. Proc Natl Acad Sci USA 111:3526–3531. doi:10.1073/pnas.1322831111
Harris J, Hartman M, Roche C et al (2011) Autophagy controls IL-1beta secretion by targeting pro-IL-1beta for degradation. J Biol Chem 286:9587–9597. doi:10.1074/jbc.M110.202911
Gutierrez MG, Master SS, Singh SB et al (2004) Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 119:753–766. doi:10.1016/j.cell.2004.11.038
Berchtold LA, Prause M, Størling J, Mandrup-Poulsen T (2016) Cytokines and Pancreatic β-Cell Apoptosis. pp 99–158
Newton K, Dixit VM (2012) Signaling in innate immunity and inflammation. Cold Spring Harb Perspect Biol 4:a006049. doi:10.1101/cshperspect.a006049
Walczak H (2013) Death receptor-ligand systems in cancer, cell death, and inflammation. Cold Spring Harb Perspect Biol 5:a008698. doi:10.1101/cshperspect.a008698
Iwasaki A, Medzhitov R (2004) Toll-like receptor control of the adaptive immune responses. Nat Immunol 5:987–995. doi:10.1038/ni1112
Brubaker SW, Bonham KS, Zanoni I, Kagan JC (2015) Innate immune pattern recognition: a cell biological perspective. Annu Rev Immunol 33:257–290. doi:10.1146/annurev-immunol-032414-112240
Zarember KA, Godowski PJ (2002) Tissue expression of human Toll-like receptors and differential regulation of Toll-like receptor mRNAs in leukocytes in response to microbes, their products, and cytokines. J Immunol 168:554–561
Broz P, Monack DM (2013) Newly described pattern recognition receptors team up against intracellular pathogens. Nat Rev Immunol 13:551–565. doi:10.1038/nri3479
Takeuchi O, Akira S (2010) Pattern recognition receptors and inflammation. Cell 140:805–820. doi:10.1016/j.cell.2010.01.022
Meylan E, Tschopp J, Karin M (2006) Intracellular pattern recognition receptors in the host response. Nature 442:39–44. doi:10.1038/nature04946
Girardin SE, Boneca IG, Viala J et al (2003) Nod2 is a general sensor of peptidoglycan through muramyl dipeptide (MDP) detection. J Biol Chem 278:8869–8872. doi:10.1074/jbc.C200651200
Lamkanfi M, Dixit VM (2014) Mechanisms and functions of inflammasomes. Cell 157:1013–1022. doi:10.1016/j.cell.2014.04.007
Nunes T, de Souza HS (2013) Inflammasome in intestinal inflammation and cancer. Mediat Inflamm 2013:654963. doi:10.1155/2013/654963
Latz E, Xiao TS, Stutz A (2013) Activation and regulation of the inflammasomes. Nat Rev Immunol 13:397–411. doi:10.1038/nri3452
Lamkanfi M, Dixit VM (2012) Inflammasomes and their roles in health and disease. Annu Rev Cell Dev Biol 28:137–161. doi:10.1146/annurev-cellbio-101011-155745
van Kooyk Y, Rabinovich GA (2008) Protein-glycan interactions in the control of innate and adaptive immune responses. Nat Immunol 9:593–601. doi:10.1038/ni.f.203
Geijtenbeek TBH, Gringhuis SI (2009) Signalling through C-type lectin receptors: shaping immune responses. Nat Rev Immunol 9:465–479. doi:10.1038/nri2569
Wilson NS, Dixit V, Ashkenazi A (2009) Death receptor signal transducers: nodes of coordination in immune signaling networks. Nat Immunol 10:348–355. doi:10.1038/ni.1714
Lee H-J, Kim K-C, Han JA et al (2015) The early induction of suppressor of cytokine signaling 1 and the downregulation of toll-like receptors 7 and 9 induce tolerance in costimulated macrophages. Mol Cells 38:26–32. doi:10.14348/molcells.2015.2136
Matsuda N, Yamazaki H, Takano K et al (2008) Priming by lipopolysaccharide exaggerates acute lung injury and mortality in responses to peptidoglycan through up-regulation of Toll-like receptor-2 expression in mice. Biochem Pharmacol 75:1065–1075. doi:10.1016/j.bcp.2007.10.036
Wesemann DR, Benveniste EN (2003) STAT-1 alpha and IFN-gamma as modulators of TNF-alpha signaling in macrophages: regulation and functional implications of the TNF receptor 1:STAT-1 alpha complex. J Immunol 171:5313–5319
Harris J, De Haro SA, Master SS et al (2007) T helper 2 cytokines inhibit autophagic control of intracellular Mycobacterium tuberculosis. Immunity 27:505–517. doi:10.1016/j.immuni.2007.07.022
Schönthal AH (2012) Endoplasmic reticulum stress: its role in disease and novel prospects for therapy. Scientifica (Cairo) 2012:857516. doi:10.6064/2012/857516
Xu C, Bailly-Maitre B, Reed JC (2005) Endoplasmic reticulum stress: cell life and death decisions. J Clin Invest 115:2656–2664. doi:10.1172/JCI26373
Jäger R, Bertrand MJM, Gorman AM et al (2012) The unfolded protein response at the crossroads of cellular life and death during endoplasmic reticulum stress. Biol Cell 104:259–270. doi:10.1111/boc.201100055
Rath E, Haller D (2011) Inflammation and cellular stress: a mechanistic link between immune-mediated and metabolically driven pathologies. Eur J Nutr 50:219–233. doi:10.1007/s00394-011-0197-0
He B (2006) Viruses, endoplasmic reticulum stress, and interferon responses. Cell Death Differ 13:393–403. doi:10.1038/sj.cdd.4401833
Martinon F, Chen X, Lee A-H, Glimcher LH (2010) TLR activation of the transcription factor XBP1 regulates innate immune responses in macrophages. Nat Immunol 11:411–418. doi:10.1038/ni.1857
Messlik A, Schmechel S, Kisling S et al (2009) Loss of Toll-like receptor 2 and 4 leads to differential induction of endoplasmic reticulum stress and proapoptotic responses in the intestinal epithelium under conditions of chronic inflammation. J Proteome Res 8:4406–4417. doi:10.1021/pr9000465
Woo CW, Cui D, Arellano J et al (2009) Adaptive suppression of the ATF4-CHOP branch of the unfolded protein response by toll-like receptor signalling. Nat Cell Biol 11:1473–1480. doi:10.1038/ncb1996
Janssens S, Pulendran B, Lambrecht BN (2014) Emerging functions of the unfolded protein response in immunity. Nat Immunol 15:910–919. doi:10.1038/ni.2991
Deegan S, Saveljeva S, Gorman AM, Samali A (2013) Stress-induced self-cannibalism: on the regulation of autophagy by endoplasmic reticulum stress. Cell Mol Life Sci 70:2425–2441. doi:10.1007/s00018-012-1173-4
Ogata M, Hino S, Saito A et al (2006) Autophagy is activated for cell survival after endoplasmic reticulum stress. Mol Cell Biol 26:9220–9231. doi:10.1128/MCB.01453-06
Szegezdi E, Logue SE, Gorman AM, Samali A (2006) Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep 7:880–885. doi:10.1038/sj.embor.7400779
Mittal M, Siddiqui MR, Tran K et al (2014) Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal 20:1126–1167. doi:10.1089/ars.2012.5149
Zhang K, Kaufman RJ (2008) From endoplasmic-reticulum stress to the inflammatory response. Nature 454:455–462. doi:10.1038/nature07203
Adam-Vizi V, Chinopoulos C (2006) Bioenergetics and the formation of mitochondrial reactive oxygen species. Trends Pharmacol Sci 27:639–645. doi:10.1016/j.tips.2006.10.005
Zhang K (2010) Integration of ER stress, oxidative stress and the inflammatory response in health and disease. Int J Clin Exp Med 3:33–40
Scherz-Shouval R, Elazar Z (2011) Regulation of autophagy by ROS: physiology and pathology. Trends Biochem Sci 36:30–38. doi:10.1016/j.tibs.2010.07.007
Zhang L, Wang K, Lei Y et al (2015) Redox signaling: potential arbitrator of autophagy and apoptosis in therapeutic response. Free Radic Biol Med 89:452–465. doi:10.1016/j.freeradbiomed.2015.08.030
Schauvliege R, Vanrobaeys J, Schotte P, Beyaert R (2002) Caspase-11 gene expression in response to lipopolysaccharide and interferon-gamma requires nuclear factor-kappa B and signal transducer and activator of transcription (STAT) 1. J Biol Chem 277:41624–41630. doi:10.1074/jbc.M207852200
Wang S, Miura M, Jung YK et al (1998) Murine caspase-11, an ICE-interacting protease, is essential for the activation of ICE. Cell 92:501–509
Fontana L Neuroendocrine factors in the regulation of inflammation: excessive adiposity and calorie restriction. Exp Gerontol 44:41–45. doi:10.1016/j.exger.2008.04.005
Reuter S, Gupta SC, Chaturvedi MM, Aggarwal BB (2010) Oxidative stress, inflammation, and cancer: how are they linked? Free Radic Biol Med 49:1603–1616. doi:10.1016/j.freeradbiomed.2010.09.006
Hitotsumatsu O, Ahmad R-C, Tavares R et al (2008) The ubiquitin-editing enzyme A20 restricts nucleotide-binding oligomerization domain containing 2-triggered signals. Immunity 28:381–390. doi:10.1016/j.immuni.2008.02.002
Boone DL, Turer EE, Lee EG et al (2004) The ubiquitin-modifying enzyme A20 is required for termination of Toll-like receptor responses. Nat Immunol 5:1052–1060. doi:10.1038/ni1110
Lee EG, Boone DL, Chai S et al (2000) Failure to regulate TNF-induced NF-kappaB and cell death responses in A20-deficient mice. Science 289:2350–2354
Russell RC, Yuan H-X, Guan K-L (2013) Autophagy regulation by nutrient signaling. Cell Res 24:42–57. doi:10.1038/cr.2013.166
Li Y, Li S, Qin X et al (2014) The pleiotropic roles of sphingolipid signaling in autophagy. Cell Death Dis 5:e1245. doi:10.1038/cddis.2014.215
Garcia-Ruiz C, Morales A, Fernández-Checa JC (2015) Glycosphingolipids and cell death: one aim, many ways. Apoptosis 20:607–620. doi:10.1007/s10495-015-1092-6
Maceyka M, Spiegel S (2014) Sphingolipid metabolites in inflammatory disease. Nature 510:58–67. doi:10.1038/nature13475
Lee J, Yeganeh B, Ermini L, Post M (2015) Sphingolipids as cell fate regulators in lung development and disease. Apoptosis 20:740–757. doi:10.1007/s10495-015-1112-6
Yang Z, Klionsky DJ (2010) Mammalian autophagy: core molecular machinery and signaling regulation. Curr Opin Cell Biol 22:124–31. doi:10.1016/j.ceb.2009.11.014
Tanida I (2011) Autophagy basics. Microbiol Immunol 55:1–11. doi:10.1111/j.1348-0421.2010.00271.x
Ravikumar B, Sarkar S, Davies JE et al (2010) Regulation of mammalian autophagy in physiology and pathophysiology. Physiol Rev 90:1383–1435. doi:10.1152/physrev.00030.2009
Burman C, Ktistakis NT (2010) Autophagosome formation in mammalian cells. Semin Immunopathol 32:397–413. doi:10.1007/s00281-010-0222-z
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36:2503–2518. doi:10.1016/j.biocel.2004.05.009
Hemelaar J, Lelyveld VS, Kessler BM, Ploegh HL (2003) A single protease, Apg4B, is specific for the autophagy-related ubiquitin-like proteins GATE-16, MAP1-LC3, GABARAP, and Apg8L. J Biol Chem 278:51841–51850. doi:10.1074/jbc.M308762200
Geng J, Klionsky DJ (2008) The Atg8 and Atg12 ubiquitin-like conjugation systems in macroautophagy. “Protein modifications: beyond the usual suspects” review series. EMBO Rep 9:859–864. doi:10.1038/embor.2008.163
Mizushima N, Yoshimori T, Ohsumi Y (2011) The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol 27:107–32. doi:10.1146/annurev-cellbio-092910-154005
Kimura S, Noda T, Yoshimori T (2008) Dynein-dependent movement of autophagosomes mediates efficient encounters with lysosomes. Cell Struct Funct 33:109–122
Eskelinen E-L, Saftig P (2009) Autophagy: a lysosomal degradation pathway with a central role in health and disease. Biochim Biophys Acta 1793:664–673. doi:10.1016/j.bbamcr.2008.07.014
Delgado MA, Elmaoued RA, Davis AS et al (2008) Toll-like receptors control autophagy. EMBO J 27:1110–1121. doi:10.1038/emboj.2008.31
Anand PK, Tait SWG, Lamkanfi M et al (2011) TLR2 and RIP2 pathways mediate autophagy of Listeria monocytogenes via extracellular signal-regulated kinase (ERK) activation. J Biol Chem 286:42981–42991. doi:10.1074/jbc.M111.310599
Shi C-S, Kehrl JH (2008) MyD88 and Trif target Beclin 1 to trigger autophagy in macrophages. J Biol Chem 283:33175–33182. doi:10.1074/jbc.M804478200
Xu Y, Jagannath C, Liu X-D et al (2007) Toll-like receptor 4 is a sensor for autophagy associated with innate immunity. Immunity 27:135–144. doi:10.1016/j.immuni.2007.05.022
Shin D-M, Yuk J-M, Lee H-M et al (2010) Mycobacterial lipoprotein activates autophagy via TLR2/1/CD14 and a functional vitamin D receptor signalling. Cell Microbiol 12:1648–1665. doi:10.1111/j.1462-5822.2010.01497.x
Henault J, Martinez J, Riggs JM et al (2012) Noncanonical autophagy is required for type I interferon secretion in response to DNA-immune complexes. Immunity 37:986–997. doi:10.1016/j.immuni.2012.09.014
Shi C-S, Kehrl JH (2010) TRAF6 and A20 regulate lysine 63-linked ubiquitination of Beclin-1 to control TLR4-induced autophagy. Sci Signal 3:ra42. doi:10.1126/scisignal.2000751
Travassos LH, Carneiro LAM, Ramjeet M et al (2010) Nod1 and Nod2 direct autophagy by recruiting ATG16L1 to the plasma membrane at the site of bacterial entry. Nat Immunol 11:55–62. doi:10.1038/ni.1823
Cooney R, Baker J, Brain O et al (2010) NOD2 stimulation induces autophagy in dendritic cells influencing bacterial handling and antigen presentation. Nat Med 16:90–97. doi:10.1038/nm.2069
Wang L-J, Huang H-Y, Huang M-P et al (2014) The microtubule-associated protein EB1 links AIM2 inflammasomes with autophagy-dependent secretion. J Biol Chem 289:29322–29333. doi:10.1074/jbc.M114.559153
Lei Y, Wen H, Yu Y et al (2012) The mitochondrial proteins NLRX1 and TUFM form a complex that regulates type I interferon and autophagy. Immunity 36:933–946. doi:10.1016/j.immuni.2012.03.025
Pilli M, Arko-Mensah J, Ponpuak M et al (2012) TBK-1 promotes autophagy-mediated antimicrobial defense by controlling autophagosome maturation. Immunity 37:223–234. doi:10.1016/j.immuni.2012.04.015
Harris J (2013) Autophagy and IL-1 Family Cytokines. Front Immunol 4:83. doi:10.3389/fimmu.2013.00083
Rashid H-O, Yadav RK, Kim H-R, Chae H-J (2015) ER stress: autophagy induction, inhibition and selection. Autophagy 11:1956–1977. doi:10.1080/15548627.2015.1091141
Høyer-Hansen M, Jäättelä M (2007) Connecting endoplasmic reticulum stress to autophagy by unfolded protein response and calcium. Cell Death Differ 14:1576–1582. doi:10.1038/sj.cdd.4402200
Høyer-Hansen M, Bastholm L, Szyniarowski P et al (2007) Control of macroautophagy by calcium, calmodulin-dependent kinase kinase-β, and Bcl-2. Mol Cell 25:193–205. doi:10.1016/j.molcel.2006.12.009
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26:239–257
Silva MT (2010) Secondary necrosis: the natural outcome of the complete apoptotic program. FEBS Lett 584:4491–4499. doi:10.1016/j.febslet.2010.10.046
Haslett C (1999) Granulocyte apoptosis and its role in the resolution and control of lung inflammation. Am J Respir Crit Care Med 160:S5–S11. doi:10.1164/ajrccm.160.supplement_1.4
Lamkanfi M, Dixit VM (2010) Manipulation of host cell death pathways during microbial infections. Cell Host Microbe 8:44–54. doi:10.1016/j.chom.2010.06.007
Pasparakis M, Vandenabeele P (2015) Necroptosis and its role in inflammation. Nature 517:311–320. doi:10.1038/nature14191
Micheau O, Tschopp J (2003) Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114:181–190
Han J, Zhong C-Q, Zhang D-W (2011) Programmed necrosis: backup to and competitor with apoptosis in the immune system. Nat Immunol 12:1143–1149. doi:10.1038/ni.2159
Li H, Zhu H, Xu CJ, Yuan J (1998) Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94:491–501
Ow Y-LP, Green DR, Hao Z, Mak TW (2008) Cytochrome c: functions beyond respiration. Nat Rev Mol Cell Biol 9:532–542. doi:10.1038/nrm2434
Saelens X, Festjens N, Vande Walle L et al (2004) Toxic proteins released from mitochondria in cell death. Oncogene 23:2861–2874. doi:10.1038/sj.onc.1207523
Duprez L, Wirawan E, Vanden Berghe T, Vandenabeele P (2009) Major cell death pathways at a glance. Microbes Infect 11:1050–1062. doi:10.1016/j.micinf.2009.08.013
Łopatniuk P, Witkowski JM (2011) Conventional calpains and programmed cell death. Acta Biochim Pol 58:287–296
Gafni J, Cong X, Chen SF et al (2009) Calpain-1 cleaves and activates caspase-7. J Biol Chem 284:25441–25449. doi:10.1074/jbc.M109.038174
Martinez JA, Zhang Z, Svetlov SI et al (2010) Calpain and caspase processing of caspase-12 contribute to the ER stress-induced cell death pathway in differentiated PC12 cells. Apoptosis 15:1480–1493. doi:10.1007/s10495-010-0526-4
Vaisid T, Barnoy S, Kosower NS (2009) Calpain activates caspase-8 in neuron-like differentiated PC12 cells via the amyloid-beta-peptide and CD95 pathways. Int J Biochem Cell Biol 41:2450–2458. doi:10.1016/j.biocel.2009.07.010
Tan Y, Wu C, De Veyra T, Greer PA (2006) Ubiquitous calpains promote both apoptosis and survival signals in response to different cell death stimuli. J Biol Chem 281:17689–17698. doi:10.1074/jbc.M601978200
Weber H, Müller L, Jonas L et al (2013) Calpain mediates caspase-dependent apoptosis initiated by hydrogen peroxide in pancreatic acinar AR42J cells. Free Radic Res 47:432–446. doi:10.3109/10715762.2013.785633
Shi M, Zhang T, Sun L et al (2013) Calpain, Atg5 and Bak play important roles in the crosstalk between apoptosis and autophagy induced by influx of extracellular calcium. Apoptosis 18:435–451. doi:10.1007/s10495-012-0786-2
Wood DE, Thomas A, Devi LA et al (1998) Bax cleavage is mediated by calpain during drug-induced apoptosis. Oncogene 17:1069–1078. doi:10.1038/sj.onc.1202034
Gao G, Dou QP (2000) N-terminal cleavage of bax by calpain generates a potent proapoptotic 18-kDa fragment that promotes bcl-2-independent cytochrome C release and apoptotic cell death. J Cell Biochem 80:53–72
Chen M (2001) Bid is cleaved by calpain to an active fragment in vitro and during myocardial ischemia/reperfusion. J Biol Chem 276:30724–30728. doi:10.1074/jbc.M103701200
Smith MA, Schnellmann RG (2012) Calpains, mitochondria, and apoptosis. Cardiovasc Res 96:32–37. doi:10.1093/cvr/cvs163
Tabas I, Ron D (2011) Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol 13:184–190. doi:10.1038/ncb0311-184
Kroemer G, Levine B (2008) Autophagic cell death: the story of a misnomer. Nat Rev Mol Cell Biol 9:1004–1010. doi:10.1038/nrm2529
Shen S, Kepp O, Kroemer G (2012) The end of autophagic cell death? Autophagy 8:1–3. doi:10.4161/auto.8.1.16618
Xue L, Fletcher GC, Tolkovsky AM (2001) Mitochondria are selectively eliminated from eukaryotic cells after blockade of caspases during apoptosis. Curr Biol 11:361–365
Zhang Y, Qi H, Taylor R et al The role of autophagy in mitochondria maintenance: characterization of mitochondrial functions in autophagy-deficient S. cerevisiae strains. Autophagy 3:337–346
Mitter SK, Song C, Qi X et al (2014) Dysregulated autophagy in the RPE is associated with increased susceptibility to oxidative stress and AMD. Autophagy 10:1989–2005. doi:10.4161/auto.36184
Paul S, Kashyap AK, Jia W et al (2012) Selective autophagy of the adaptor protein Bcl10 modulates T cell receptor activation of NF-κB. Immunity 36:947–958. doi:10.1016/j.immuni.2012.04.008
Shibata Y, Oyama M, Kozuka-Hata H et al (2012) p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO. Nat Commun 3:1061. doi:10.1038/ncomms2068
Kimura T, Jain A, Choi SW et al (2015) TRIM-mediated precision autophagy targets cytoplasmic regulators of innate immunity. J Cell Biol. doi:10.1083/jcb.201503023
Hou W, Han J, Lu C et al (2014) Autophagic degradation of active caspase-8. Autophagy 6:891–900. doi:10.4161/auto.6.7.13038
Chen S, Zhou L, Zhang Y et al (2014) Targeting SQSTM1/p62 induces cargo loading failure and converts autophagy to apoptosis via NBK/Bik. Mol Cell Biol 34:3435–3449. doi:10.1128/MCB.01383-13
Young MM, Takahashi Y, Khan O et al (2012) Autophagosomal membrane serves as platform for intracellular death-inducing signaling complex (iDISC)-mediated caspase-8 activation and apoptosis. J Biol Chem 287:12455–12468. doi:10.1074/jbc.M111.309104
Laussmann MA, Passante E, Düssmann H et al (2011) Proteasome inhibition can induce an autophagy-dependent apical activation of caspase-8. Cell Death Differ 18:1584–1597. doi:10.1038/cdd.2011.27
Deegan S, Saveljeva S, Logue SE et al (2014) Deficiency in the mitochondrial apoptotic pathway reveals the toxic potential of autophagy under ER stress conditions. Autophagy 10:1921–1936. doi:10.4161/15548627.2014.981790
Rubinstein AD, Eisenstein M, Ber Y et al (2011) The autophagy protein Atg12 associates with antiapoptotic Bcl-2 family members to promote mitochondrial apoptosis. Mol Cell 44:698–709. doi:10.1016/j.molcel.2011.10.014
Yu L, Alva A, Su H et al (2004) Regulation of an ATG7-beclin 1 program of autophagic cell death by caspase-8. Science 304:1500–1502. doi:10.1126/science.1096645
Yin X, Cao L, Kang R et al (2011) UV irradiation resistance-associated gene suppresses apoptosis by interfering with BAX activation. EMBO Rep 12:727–734. doi:10.1038/embor.2011.79
Han J, Hou W, Goldstein LA et al (2014) A Complex between Atg7 and Caspase-9: a novel mechanism of cross-regulation between autophagy and apoptosis. J Biol Chem 289:6485–6497. doi:10.1074/jbc.M113.536854
Jeong H-S, Choi HY, Lee E-R et al (2011) Involvement of caspase-9 in autophagy-mediated cell survival pathway. Biochim Biophys Acta 1813:80–90. doi:10.1016/j.bbamcr.2010.09.016
Demarchi F, Schneider C (2007) The calpain system as a modulator of stress/damage response. Cell Cycle 6:136–138
Norman JM, Cohen GM, Bampton ETW (2010) The in vitro cleavage of the hAtg proteins by cell death proteases. Autophagy 6:1042–1056. doi:10.4161/auto.6.8.13337
Yousefi S, Perozzo R, Schmid I et al (2006) Calpain-mediated cleavage of Atg5 switches autophagy to apoptosis. Nat Cell Biol 8:1124–1132. doi:10.1038/ncb1482
Wirawan E, Vande Walle L, Kersse K et al (2010) Caspase-mediated cleavage of Beclin-1 inactivates Beclin-1-induced autophagy and enhances apoptosis by promoting the release of proapoptotic factors from mitochondria. Cell Death Dis 1:e18. doi:10.1038/cddis.2009.16
You M, Savaraj N, Kuo MT et al (2012) TRAIL induces autophagic protein cleavage through caspase activation in melanoma cell lines under arginine deprivation. Mol Cell Biochem 374:181–190. doi:10.1007/s11010-012-1518-1
Rohn TT, Wirawan E, Brown RJ et al (2011) Depletion of Beclin-1 due to proteolytic cleavage by caspases in the Alzheimer’s disease brain. Neurobiol Dis 43:68–78. doi:10.1016/j.nbd.2010.11.003
Russo R, Berliocchi L, Adornetto A et al (2011) Calpain-mediated cleavage of Beclin-1 and autophagy deregulation following retinal ischemic injury in vivo. Cell Death Dis 2:e144. doi:10.1038/cddis.2011.29
Pagliarini V, Wirawan E, Romagnoli A et al (2012) Proteolysis of Ambra1 during apoptosis has a role in the inhibition of the autophagic pro-survival response. Cell Death Differ 19:1495–1504. doi:10.1038/cdd.2012.27
Betin VMS, Lane JD (2009) Caspase cleavage of Atg4D stimulates GABARAP-L1 processing and triggers mitochondrial targeting and apoptosis. J Cell Sci 122:2554–2566. doi:10.1242/jcs.046250
Betin VMS, MacVicar TDB, Parsons SF et al (2012) A cryptic mitochondrial targeting motif in Atg4D links caspase cleavage with mitochondrial import and oxidative stress. Autophagy 8:664–676. doi:10.4161/auto.19227
Siddiqui MA, Mukherjee S, Manivannan P, Malathi K (2015) RNase L cleavage products promote switch from autophagy to apoptosis by caspase-mediated cleavage of Beclin-1. Int J Mol Sci 16:17611–17636. doi:10.3390/ijms160817611
Luo S, Rubinsztein DC (2010) Apoptosis blocks Beclin 1-dependent autophagosome synthesis: an effect rescued by Bcl-xL. Cell Death Differ 17:268–277. doi:10.1038/cdd.2009.121
Ciechomska IA, Goemans CG, Tolkovsky AM (2009) Why doesn’t Beclin 1, a BH3-only protein, suppress the anti-apoptotic function of Bcl-2? Autophagy 5:880–881
Kang R, Zeh HJ, Lotze MT, Tang D (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18:571–580. doi:10.1038/cdd.2010.191
R Safa A (2013) Roles of c-FLIP in apoptosis, necroptosis, and autophagy. J Carcinog Mutagen. doi:10.4172/2157-2518.S6-003
Gozuacik D, Bialik S, Raveh T et al (2008) DAP-kinase is a mediator of endoplasmic reticulum stress-induced caspase activation and autophagic cell death. Cell Death Differ 15:1875–1886. doi:10.1038/cdd.2008.121
Inbal B, Bialik S, Sabanay I et al (2002) DAP kinase and DRP-1 mediate membrane blebbing and the formation of autophagic vesicles during programmed cell death. J Cell Biol 157:455–468. doi:10.1083/jcb.200109094
Levin-Salomon V, Bialik S, Kimchi A (2014) DAP-kinase and autophagy. Apoptosis 19:346–356. doi:10.1007/s10495-013-0918-3
Luo S, Rubinsztein DC (2007) Atg5 and Bcl-2 provide novel insights into the interplay between apoptosis and autophagy. Cell Death Differ 14:1247–1250. doi:10.1038/sj.cdd.4402149
Maiuri MC, Le Toumelin G, Criollo A et al (2007) Functional and physical interaction between Bcl-X(L) and a BH3-like domain in Beclin-1. EMBO J 26:2527–2539. doi:10.1038/sj.emboj.7601689
Klionsky DJ, Abdalla FC, Abeliovich H et al (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8:445–544
Erlich S, Mizrachy L, Segev O et al Differential interactions between Beclin 1 and Bcl-2 family members. Autophagy 3:561–568
Wei Y, Sinha S, Levine B (2008) Dual role of JNK1-mediated phosphorylation of Bcl-2 in autophagy and apoptosis regulation. Autophagy 4:949–951
Zalckvar E, Berissi H, Mizrachy L et al (2009) DAP-kinase-mediated phosphorylation on the BH3 domain of beclin 1 promotes dissociation of beclin 1 from Bcl-XL and induction of autophagy. EMBO Rep 10:285–292. doi:10.1038/embor.2008.246
Zalckvar E, Berissi H, Eisenstein M, Kimchi A (2009) Phosphorylation of Beclin 1 by DAP-kinase promotes autophagy by weakening its interactions with Bcl-2 and Bcl-XL. Autophagy 5:720–722
Tang D, Kang R, Livesey KM et al (2010) Endogenous HMGB1 regulates autophagy. J Cell Biol 190:881–892. doi:10.1083/jcb.200911078
Pyo J-O, Jang M-H, Kwon Y-K et al (2005) Essential roles of Atg5 and FADD in autophagic cell death: dissection of autophagic cell death into vacuole formation and cell death. J Biol Chem 280:20722–20729. doi:10.1074/jbc.M413934200
Harris HE, Andersson U, Pisetsky DS (2012) HMGB1: a multifunctional alarmin driving autoimmune and inflammatory disease. Nat Rev Rheumatol 8:195–202. doi:10.1038/nrrheum.2011.222
Zhu X, Messer JS, Wang Y et al (2015) Cytosolic HMGB1 controls the cellular autophagy/apoptosis checkpoint during inflammation. J Clin Invest 125:1098–1110. doi:10.1172/JCI76344
Lin HH, Lin S-M, Chung Y et al (2014) Dynamic involvement of ATG5 in cellular stress responses. Cell Death Dis 5:e1478. doi:10.1038/cddis.2014.428
Lépine S, Allegood JC, Edmonds Y et al (2011) Autophagy induced by deficiency of sphingosine-1-phosphate phosphohydrolase 1 is switched to apoptosis by calpain-mediated autophagy-related gene 5 (Atg5) cleavage. J Biol Chem 286:44380–44390. doi:10.1074/jbc.M111.257519
Shimizu S, Kanaseki T, Mizushima N et al (2004) Role of Bcl-2 family proteins in a non-apoptotic programmed cell death dependent on autophagy genes. Nat Cell Biol 6:1221–1228. doi:10.1038/ncb1192
Acknowledgements
Funding was provided by Crohn’s and Colitis Foundation of America and National Institute of Diabetes and Digestive and Kidney Diseases (Grant Nos. http://dx.doi.org/10.13039/100001063, P30 DK42086).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Messer, J.S. The cellular autophagy/apoptosis checkpoint during inflammation. Cell. Mol. Life Sci. 74, 1281–1296 (2017). https://doi.org/10.1007/s00018-016-2403-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-016-2403-y