Abstract
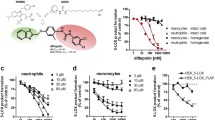
We previously showed that, in vitro, hyperforin from St. John’s wort (Hypericum perforatum) inhibits 5-lipoxygenase (5-LO), the key enzyme in leukotriene biosynthesis. Here, we demonstrate that hyperforin possesses a novel and unique molecular pharmacological profile as a 5-LO inhibitor with remarkable efficacy in vivo. Hyperforin (4 mg/kg, i.p.) significantly suppressed leukotriene B4 formation in pleural exudates of carrageenan-treated rats associated with potent anti-inflammatory effectiveness. Inhibition of 5-LO by hyperforin, but not by the iron-ligand type 5-LO inhibitor BWA4C or the nonredox-type inhibitor ZM230487, was abolished in the presence of phosphatidylcholine and strongly reduced by mutation (W13A-W75A-W102A) of the 5-LO C2-like domain. Moreover, hyperforin impaired the interaction of 5-LO with coactosin-like protein and abrogated 5-LO nuclear membrane translocation in ionomycin-stimulated neutrophils, processes that are typically mediated via the regulatory 5-LO C2-like domain. Together, hyperforin is a novel type of 5-LO inhibitor apparently acting by interference with the C2-like domain, with high effectiveness in vivo.








Similar content being viewed by others
References
Rådmark O, Werz O, Steinhilber D, Samuelsson B (2007) 5-Lipoxygenase: regulation of expression and enzyme activity. Trends Biochem Sci 32:332–341
Funk CD (2001) Prostaglandins and leukotrienes: advances in eicosanoid biology. Science 294:1871–1875
Peters-Golden M, Henderson WR Jr (2007) Leukotrienes. N Engl J Med 357:1841–1854
Werz O, Steinhilber D (2006) Therapeutic options for 5-lipoxygenase inhibitors. Pharmacol Ther 112:701–718
Capra V, Ambrosio M, Riccioni G, Rovati GE (2006) Cysteinyl-leukotriene receptor antagonists: present situation and future opportunities. Curr Med Chem 13:3213–3226
Werz O (2002) 5-Lipoxygenase: cellular biology and molecular pharmacology. Curr Drug Targets Inflamm Allergy 1:23–44
Ford-Hutchinson AW, Gresser M, Young RN (1994) 5-Lipoxygenase. Annu Rev Biochem 63:383–417
Werz O, Steinhilber D (2005) Pharmacological intervention with 5-lipoxygenase: new insights and novel compounds. Expert Opin Ther Pat 15:505–519
Rådmark O, Samuelsson B (2005) Regulation of 5-lipoxygenase enzyme activity. Biochem Biophys Res Commun 338:102–110
Pergola C, Dodt G, Rossi A, Neunhoeffer E, Lawrenz B, Northoff H, Samuelsson B, Rådmark O, Sautebin L, Werz O (2008) ERK-mediated regulation of leukotriene biosynthesis by androgens: a molecular basis for gender differences in inflammation and asthma. Proc Natl Acad Sci USA 105:19881–19886
Fischer L, Steinhilber D, Werz O (2004) Molecular pharmacological profile of the nonredox-type 5-lipoxygenase inhibitor CJ-13, 610. Br J Pharmacol 142:861–868
Fischer L, Szellas D, Rådmark O, Steinhilber D, Werz O (2003) Phosphorylation- and stimulus-dependent inhibition of cellular 5-lipoxygenase activity by nonredox-type inhibitors. FASEB J 17:949–951
Werz O, Szellas D, Henseler M, Steinhilber D (1998) Nonredox 5-lipoxygenase inhibitors require glutathione peroxidase for efficient inhibition of 5-lipoxygenase activity. Mol Pharmacol 54:445–451
Hammarberg T, Provost P, Persson B, Rådmark O (2000) The N-terminal domain of 5-lipoxygenase binds calcium and mediates calcium stimulation of enzyme activity. J Biol Chem 275:38787–38793
Kulkarni S, Das S, Funk CD, Murray D, Cho W (2002) A molecular basis of specific subcellular localization of the C2-like domain of 5-lipoxygenase. J Biol Chem 277:13167–13174
Rakonjac M, Fischer L, Provost P, Werz O, Steinhilber D, Samuelsson B, Rådmark O (2006) Coactosin-like protein supports 5-lipoxygenase enzyme activity and up-regulates leukotriene A4 production. Proc Natl Acad Sci USA 103:13150–13155
Albert D, Zundorf I, Dingermann T, Muller WE, Steinhilber D, Werz O (2002) Hyperforin is a dual inhibitor of cyclooxygenase-1 and 5-lipoxygenase. Biochem Pharmacol 64:1767–1775
Werz O, Burkert E, Samuelsson B, Rådmark O, Steinhilber D (2002) Activation of 5-lipoxygenase by cell stress is calcium independent in human polymorphonuclear leukocytes. Blood 99:1044–1052
Hornig C, Albert D, Fischer L, Hornig M, Rådmark O, Steinhilber D, Werz O (2005) 1-Oleoyl-2-acetylglycerol stimulates 5-lipoxygenase activity via a putative (phospho) lipid binding site within the N-terminal C2-like domain. J Biol Chem 280:26913–26921
Burkert E, Arnold C, Hammarberg T, Rådmark O, Steinhilber D, Werz O (2003) The C2-like beta-barrel domain mediates the Ca2+-dependent resistance of 5-lipoxygenase activity against inhibition by glutathione peroxidase-1. J Biol Chem 278:42846–42853
Werz O, Klemm J, Samuelsson B, Rådmark O (2001) Phorbol ester up-regulates capacities for nuclear translocation and phosphorylation of 5-lipoxygenase in Mono Mac 6 cells and human polymorphonuclear leukocytes. Blood 97:2487–2495
Jones G, Willett P, Glen RC (1995) Molecular recognition of receptor sites using a genetic algorithm with a description of desolvation. J Mol Biol 245:43–53
Werz O, Tretiakova I, Michel A, Ulke-Lemee A, Hornig M, Franke L, Schneider G, Samuelsson B, Rådmark O, Steinhilber D (2005) Caspase-mediated degradation of human 5-lipoxygenase in B lymphocytic cells. Proc Natl Acad Sci USA 102:13164–13169
DeLano WL (2002) The PyMOL molecular graphics system (2002). DeLano Scientific, San Carlos, CA, USA. http://www.pymol.org
Carter GW, Young PR, Albert DH, Bouska J, Dyer R, Bell RL, Summers JB, Brooks DW (1991) 5-Lipoxygenase inhibitory activity of zileuton. J Pharmacol Exp Ther 256:929–937
Werz O, Schneider N, Brungs M, Sailer ER, Safayhi H, Ammon HPT, Steinhilber D (1997) A test system for leukotriene synthesis inhibitors based on the in vitro differentiation of the human leukemic cell lines HL-60 and Mono Mac 6. Naunyn Schmiedebergs Arch Pharmacol 356:441–445
Tateson JE, Randall RW, Reynolds CH, Jackson WP, Bhattacherjee P, Salmon JA, Garland LG (1988) Selective inhibition of arachidonate 5-lipoxygenase by novel acetohydroxamic acids: biochemical assessment in vitro and ex vivo. Br J Pharmacol 94:528–539
Moore LB, Goodwin B, Jones SA, Wisely GB, Serabjit-Singh CJ, Willson TM, Collins JL, Kliewer SA (2000) St. John’s wort induces hepatic drug metabolism through activation of the pregnane X receptor. Proc Natl Acad Sci USA 97:7500–7502
Albert D, Buerkert E, Steinhilber D, Werz O (2003) Induction of 5-lipoxygenase activation in polymorphonuclear leukocytes by 1-oleoyl-2-acetylglycerol. Biochim Biophys Acta 1631:85–93
Provost P, Doucet J, Hammarberg T, Gerisch G, Samuelsson B, Rådmark O (2001) 5-Lipoxygenase interacts with coactosin-like protein. J Biol Chem 276:16520–16527
Chen XS, Funk CD (2001) The N-terminal “beta-barrel” domain of 5-lipoxygenase is essential for nuclear membrane translocation. J Biol Chem 276:811–818
Rao TS, Currie JL, Shaffer AF, Isakson PC (1993) Evaluation of 5-lipoxygenase inhibitors, zileuton, A-78773 and ICI-D-2138 in an ionophore (A-23187)-induced pleural inflammation model in the rat. Life Sci 53:PL147–PL152
Cuzzocrea S, Rossi A, Serraino I, Mazzon E, Di Paola R, Dugo L, Genovese T, Calabro B, Caputi AP, Sautebin L (2003) 5-Lipoxygenase knockout mice exhibit a resistance to pleurisy and lung injury caused by carrageenan. J Leukoc Biol 73:739–746
Heilmann J, Winkelmann K, Sticher O (2003) Studies on the antioxidative activity of phloroglucinol derivatives isolated from hypericum species. Planta Med 69:202–206
Feisst C, Werz O (2004) Suppression of receptor-mediated Ca2+ mobilization and functional leukocyte responses by hyperforin. Biochem Pharmacol 67:1531–1539
Puustinen T, Scheffer MM, Samuelsson B (1988) Regulation of the human leukocyte 5-lipoxygenase: stimulation by micromolar calcium levels and phosphatidylcholine vesicles. Biochim Biophys Acta 960:261–267
Noguchi M, Miyano M, Matsumoto T, Noma M (1994) Human 5-lipoxygenase associates with phosphatidylcholine liposomes and modulates LTA(4) synthetase activity. Biochim Biophys Acta 1215:300–306
Dell’Aica I, Niero R, Piazza F, Cabrelle A, Sartor L, Colalto Brunetta E, Lorusso G, Benelli R, Albini A, Calabrese F, Agostini C, Garbisa S (2007) Hyperforin blocks neutrophil activation of matrix metalloproteinase-9, motility and recruitment, and restrains inflammation-triggered angiogenesis and lung fibrosis. J Pharmacol Exp Ther 321:492–500
Acknowledgments
We thank Sven George and Bianca Jazzar for expert technical assistance. The study was supported by the Deutsche Forschungsgemeinschaft, the Beilstein-Institut zur Förderung der chemischen Wissenschaften, the Swedish Research Council, 03X-217, and the European Union (EICOSANOX, LSHM-CT-2004-005033. Disclaimer: The report reflects only the author’s views and the Community is not liable for any use that may be made of the information herein). C.P. received a Carl-Zeiss stipend.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feißt, C., Pergola, C., Rakonjac, M. et al. Hyperforin is a novel type of 5-lipoxygenase inhibitor with high efficacy in vivo. Cell. Mol. Life Sci. 66, 2759–2771 (2009). https://doi.org/10.1007/s00018-009-0078-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-009-0078-3