Abstract
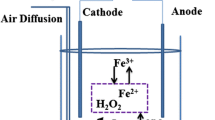
Deepwater discharges of sulfide flotation tailings have been practiced for several decades. Oxygen penetration and organic matter content in such environments are the two major factors that determine their reduction-oxidation condition and hence the rate of sulfide mineral dissolution under such discharge environments. In this study, the effects of solution redox potential and pH on the dissolution of some heavy metals from sulfide flotation tailings were examined. Zn, Pb and Cu concentrations in solution were all higher at a higher redox potential and lower pH. Dissolved Zn concentrations reached 100 ppm even at a low redox potential of 105 mV and near neutral pH after 33 days. Pb concentrations were below 2 ppm at 105 mV and near neutral pH after 165 days. At 105 mV, the Cu concentrations were below 0.2 ppm at near neutral pH after 165 days. At pH 2.3, the Fe concentrations were higher at 420 mV than at 105 mV. But at pH 6.8, the Fe concentrations were lower at 420 mV than at 105 mV, due to the oxidation of Fe2+ to Fe3+ and the subsequent precipitation of ferric hydroxide. Arsenic concentrations in solution were all less than 0.002 ppm. For Zn, Cu and Fe, no significant differences were observed between the tests in solutions prepared using seawater and those using distilled water. However, Pb concentrations reached higher levels in solutions prepared using seawater.
Similar content being viewed by others
References
Alpers, C.N. and Blowes, D.W., eds., 1994, Environmental Geochemistry of Sulfide Oxidation, ACS Symposium Series 550, Washington, D. C., American Chemistry Society, 681 pp.
Choudhury, A., 2005, Pretreatment of Aqueous Phase of Mine Plant Tailings for Submarine Disposal, M.S. thesis, University of Alaska Fairbanks, 70 pp.
Evangelou, V.P. and Zhang, Y.L., 1995, “A Review: Pyrite Oxidation Mechanisms and Acid Mine Drainage Prevention”, Critical Reviews in Environmental Science and Technology, Vol. 25, No. 2, pp. 141–191.
Lin, H. K., Walsh, D.E., Oleson, J.L. and Chen, X., 2008(a), “Reduction of Cyanate to Cyanide in Cyanidation Tailings under Reducing Environments,” Minerals and Metallurgical Processing, Vol. 25, No. 1, pp. 41–45.
Lin, H.K., Walsh, D.E., Chen, X., and Oleson, J.L., 2008(b), “Simulating deepwater discharge of sulfide tailings,” Research report to Minerals Management Service, Department of Interior, 23 pp.
Lin, H.K. and Rao, P.D., 1988, “Ferric Chloride Leaching of the Delta Sulfide Ores and Gold Extraction from the Leaching Residue”, in Hydrometallurgy of Complex Sulfide Ores, P.D. Rao, ed., Mineral Industry Research Laboratory Report No. 81, University of Alaska Fairbanks, pp. 106–123.
Marcus, J.J., ed., 1997, Mining Environmental Handbook, Imperial College Press, London, chap. 13.
Poling, G.W., Ellis, D.V., Murray, J.W., Parsons, T.R. and Pelletier, C.A., 2002(a), Underwater Tailing Placement at Island Copper Mine, Society for Mining, Metallurgy, and Exploration, Inc., Littleton, Colorado, chap. 8.
Poling, G.W., Ellis, D.V., Murray, J.W., Parsons, T.R. and Pelletier, C.A., 2002(b), Underwater Tailing Placement at Island Copper Mine, Society for Mining, Metallurgy, and Exploration, Inc., Littleton, Colorado, p. 21.
Ritcey, G.M., 1989, Tailings Management: Problems and Solutions in the Mining Industry, Elsevier Science Publishes, Amsterdam, The Netherlands, p. 444.
U.S. Environmental Protection Agency, 1977, Proposed Revisions to Ocean Dumping Criteria, Final Environmental Impact Statement, 201 pp.
Author information
Authors and Affiliations
Additional information
Paper number MMP-08-038.
Discussion of this peer-reviewed and approved paper is invited and must be submitted to SME Publications Dept. prior to Feb. 28, 2010.
Rights and permissions
About this article
Cite this article
Lin, H.K., Walsh, D.E., Chen, X. et al. Release of heavy metals from sulfide flotation tailings under deepwater discharge environments. Mining, Metallurgy & Exploration 26, 174–178 (2009). https://doi.org/10.1007/BF03402232
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03402232