Abstract
Background: European hypertension guidelines estimate that up to 15–20% of hypertensive patients are not controlled on a dual antihypertensive combination and require three or more different antihypertensive drug classes to achieve blood pressure (BP) control.
Objective: This study in patients with moderate-to-severe hypertension assessed the efficacy and safety of adding hydrochlorothiazide (HCTZ) 12.5 mg and 25 mg to a range of olmesartan medoxomil (OLM)/amlodipine (AML) doses. Study Design: This phase III, multicentre study had a randomized, double-blind, parallel-group design that included a double-blind safety run-in and a double-blind treatment period.
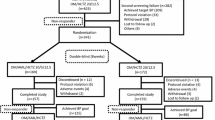
Intervention: Enrolled patients were screened and previous therapy was discontinued if required. During a 2-week, double-blind, safety run-in period (Weeks 0–2), patients were randomized to receive placebo, OLM/AML 20 mg/5 mg, OLM/AML 40 mg/5 mg or OLM/AML 40 mg/10 mg. During an 8-week, double-blind treatment period (Weeks 3–10), patients were allocated to eight groups depending on their initial treatment. They were either randomized to continue with the same dose of OLM/AML, or have HCTZ 12.5 mg or 25 mg added to treatment.
Main Outcome Measure: The primary endpoint was formulated before data collection began. It was the change in mean diastolic BP (DBP) from baseline to Week 10 in groups with HCTZ added to OLM/AML, compared with the corresponding dual OLM/AML therapy.
Results: Of 3195 patients who were screened, 2690 were randomized. Patients in every triple OLM/AML/HCTZ group had significantly greater mean reductions in DBP (p ≤ 0.032 for each comparison) and systolic BP (SBP) by Week 10 (p ≤ 0.0034 for each comparison), compared with patients on the corresponding OLM/AML therapy dose. The significant improvements in DBP and SBP reduction with triple OLM/AML/HCTZ therapy, compared with dual OLM/AML therapy, were observed after 4 and 6 weeks of therapy. Patients in each triple therapy group also had a significantly higher rate of BP < 140/90 mmHg threshold achievement (p ≤ 0.05 for each treatment comparison), compared with the dual OLM/AML groups. In three of the OLM/AML/HCTZ groups (40 mg/5 mg/25 mg, 40 mg/10 mg/12.5 mg and 40 mg/10 mg/25 mg), BP <140/90 mmHg threshold achievement by Week 10 was over 70%. Across the triple and dual combination therapy groups, treatment was well tolerated and no safety concerns for either treatment were identified.
Conclusion: Adding HCTZ to a range of OLM/AML dose combinations is well tolerated and improved BP control by significantly lowering DBP and SBP and significantly increasing BP threshold achievement in patients with moderate-to-severe hypertension.
Clinical Trial Registration: Registered at ClinicalTrials.gov identifier as NCT00923091.







Similar content being viewed by others
References
Mancia G, De Backer G, Dominiczak A, et al. 2007 Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens 2007; 25(6): 1105–87
Mancia G, Laurent S, Agabiti-Rosei E, et al. Reappraisal of European guidelines on hypertension management: a European Society of Hypertension Task Force document. J Hypertens 2009; 27(11): 2121–58
Wald DS, Law M, Morris JK, et al. Combination therapy versus monotherapy in reducing blood pressure: meta-analysis on 11,000 participants from 42 trials. Am J Med 2009; 122(3): 290–300
Jamerson K, Weber MA, Bakris GL, et al. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008; 359(23): 2417–28
Dahlof B, Sever PS, Poulter NR, et al. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA): a multicentre randomised controlled trial. Lancet 2005; 366(9489): 895–906
Sowers JR, Raij L, Jialal I, et al. Angiotensin receptor blocker/diuretic combination preserves insulin responses in obese hypertensives. J Hypertens 2010; 28(8): 1761–9
Chrysant S, Melino M, Karki S, et al. The combination of olmesartan medoxomil and amlodipine besylate in controlling high blood pressure: COACH, a randomized, double-blind, placebo-controlled, 8-week factorial efficacy and safety study. Clin Ther 2008; 30(4): 587–604
Pepine CJ, Handberg EM, Cooper-DeHoff RM, et al. A calcium antagonist vs a non-calcium antagonist hypertension treatment strategy for patients with coronary artery disease. The International Verapamil-Trandolapril Study (INVEST): a randomized controlled trial. JAMA 2003; 290(21): 2805–16
Oparil S, Melino M, Lee J, et al. Triple therapy with olmesartan medoxomil, amlodipine besylate, and hydrochlorothiazide in adult patients with hypertension: The TRINITY multicenter, randomized, double-blind, 12-week, parallel-group study. Clin Ther 2010; 32(7): 1252–69
Lloyd-Jones DM, Evans JC, Larson MG, et al. Treatment and control of hypertension in the community: a prospective analysis. Hypertension 2002; 40(5): 640–6
Weycker D, Edelsberg J, Vincze G, et al. Blood pressure control in patients initiating antihypertensive therapy. Ann Pharmacother 2008; 42(2): 169–76
Volpe M, Miele C, Haag U. Efficacy and safety of a stepped-care regimen using olmesartan medoxomil, amlodipine and hydrochlorothiazide in patients with moderate-to-severe hypertension: an open-label, long-term study. Clin Drug Investig 2009; 29(6): 381–91
Chrysant SG, Oparil S, Melino M, et al. Efficacy and safety of long-term treatment with the combination of amlodipine besylate and olmesartan medoxomil in patients with hypertension. J Clin Hypertens (Greenwich) 2009; 11(9): 475–82
Calhoun DA, Lacourciere Y, Chiang YT, et al. Triple anti-hypertensive therapy with amlodipine, valsartan, and hydrochlorothiazide: a randomized clinical trial. Hypertension 2009; 54(1): 32–9
Volpe M, Tocci G. Redefining blood pressure targets in high-risk patients? lessons from coronary endpoints in recent randomized clinical trials. Am J Hypertens 2011; 24(10): 1060–8
Tocci G, Volpe M. Modern clinical management of arterial hypertension: fixed or free combination therapies? High Blood Press Cardiovasc Prev 2011; 18 Suppl. 1: 3–11
Tocci G, Volpe M. Fixed-combination therapy to improve blood pressure control: experience with olmesartan-based therapy. Expert Rev Cardiovasc Ther 2011; 9(7): 829–40
Gupta AK, Arshad S, Poulter NR. Compliance, safety, and effectiveness of fixed-dose combinations of antihypertensive agents: a meta-analysis. Hypertension 2010; 55(2): 399–407
World Health Organization. Reducing risks, promoting healthy life. The World Health Report 2002 [online]. Available from URL: http://www.who.int/whr/2002/en/whr02_en.pdf [Accessed 2008 Sep 17]
Chrysant SG, Weber MA, Wang AC, et al. Evaluation of antihypertensive therapy with the combination of olmesartan medoxomil and hydrochlorothiazide. Am J Hypertens 2004; 17(3): 252–9
Volpe M, Brommer P, Haag U, et al. Efficacy and tolerability of olmesartan medoxomil combined with amlodipine in patients with moderate to severe hypertension after amlodipine monotherapy: a randomized, double-blind, parallel-group, multicentre study. Clin Drug Investig 2009; 29(1): 11–25
Oparil S, Chrysant SG, Kereiakes D, et al. Results of an olmesartan medoxomil-based treatment regimen in hypertensive patients. J Clin Hypertens (Greenwich) 2008; 10(12): 911–21
Rump LC, Girerd X, Sellin L, et al. Effects of high dose olmesartan medoxomil plus hydrochlorothiazide on blood pressure control in patients with grade 2 and grade 3 hypertension. J Hum Hypertens 2010; 25(9): 565–74
Fogari R, Taddei S, Holm-Bentzen M, et al. Efficacy and safety of olmesartan medoxomil 40 mg/hydrochlorothiazide 12.5 mg combination therapy versus olmesartan medoxomil 40 mg monotherapy in patients with moderate to severe hypertension: a randomized, double-blind, parallel-group, multicentre, multinational, phase III study. Clin Drug Investig 2010; 30(9): 581–97
Chrysant SG, Lee J, Melino M, et al. Efficacy and tolerability of amlodipine plus olmesartan medoxomil in patients with difficult-to-treat hypertension. J Hum Hypertens 2010; 24(11): 730–8
Chrysant SG, Chavanu KJ, Xu J. Combination therapy with olmesartan medoxomil and hydrochlorothiazide: secondary analysis of the proportion of patients achieving recommended blood pressure goals from a randomized, double-blind, factorial study. Am J Cardiovasc Drugs 2009; 9(4): 241–51
Mourad JJ, Le Jeune S. Effective systolic blood pressure reduction with olmesartan medoxomil/amlodipine combination therapy: post hoc analysis of data from a randomized, double-blind, parallel-group, multicentre study. Clin Drug Investig 2009; 29(6): 419–25
Punzi H, Neutel JM, Kereiakes DJ, et al. Efficacy of amlodipine and olmesartan medoxomil in patients with hypertension: the AZOR Trial Evaluating Blood Pressure Reductions and Control (AZTEC) study. Ther Adv Cardiovasc Dis 2010; 4(4): 209–21
Oparil S, Lee J, Karki S, et al. Subgroup analyses of an efficacy and safety study of concomitant administration of amlodipine besylate and olmesartan medoxomil: evaluation by baseline hypertension stage and prior antihypertensive medication use. J Cardiovasc Pharmacol 2009; 54(5): 427–36
Ram CV, Sachson R, Littlejohn T, et al. Management of hypertension in patients with diabetes using an amlodipine-, olmesartan medoxomil-, and hydrochlorothiazide-based titration regimen. Am J Cardiol 2011; 107(9): 1346–52
Weir MR, Hsueh WA, Nesbitt SD, et al. A titrate-to-goal study of switching patients uncontrolled on antihypertensive monotherapy to fixed-dose combinations of amlodipine and olmesartan medoxomil +/- hydrochlorothiazide. J Clin Hypertens (Greenwich) 2011; 13(6): 404–12
Neutel JM, Kereiakes DJ, BENIFICIARY Investigators. An olmesartan medoxomil-based treatment algorithm is effective in achieving 24-hour BP control in patients with type 2 diabetes mellitus, regardless of age, race, sex, or severity of hypertension: subgroup analysis of the BENIFICIARY study. Am J Cardiovasc Drugs 2010; 10(5): 289–303
Neutel JM, Kereiakes DJ, Waverczak WF, et al. Effects of an olmesartan medoxomil based treatment algorithm on 24-hour blood pressure control in patients with hypertension and type 2 diabetes. Curr Med Res Opin 2010; 26(3): 721–8
Kereiakes DJ, Neutel J, Stoakes KA, et al. The effects of an olmesartan medoxomil-based treatment algorithm on 24-hour blood pressure levels in elderly patients aged 65 and older. J Clin Hypertens (Greenwich) 2009; 11(8): 411–21
Oparil S, Chrysant SG, Melino M, et al. Long-term efficacy of a combination of amlodipine and olmesartan medoxomil +/- hydrochlorothiazide in patients with hypertension stratified by age, race and diabetes status: a substudy of the COACH trial. J Hum Hypertens 2010; 24(12): 831–8
Acknowledgements
This study was designed and sponsored by Daiichi Sankyo Europe GmbH, Munich, Germany. Prof. Lars Christian Rump served as Coordinating Investigator and Medical Expert for the study. Medical writing assistance during the preparation of this manuscript was funded by Daiichi Sankyo Europe GmbH and provided by Faysal Riaz, PhD, of inScience Communications, Springer Healthcare, Chester, UK.
Prof. Massimo Volpe has participated in advisory boards and received honoraria for lectures from Daiichi Sankyo and Novartis Pharma. Prof. Lars Christian Rump has received honoraria from Daiichi Sankyo for lectures and consultancy work.
The authors thank all the investigators, study nurses and coordinators who participated in the study for their work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volpe, M., Rump, L.C., Ammentorp, B. et al. Efficacy and Safety of Triple Antihypertensive Therapy with the Olmesartan/Amlodipine/Hydrochlorothiazide Combination. Clin Drug Investig 32, 649–664 (2012). https://doi.org/10.1007/BF03261919
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03261919