Summary
Background
In the early phase after heart transplantation (HTX) patients are at high risk for infection because of intensified immunosuppression. This retrospective study evaluates the efficacy of a three-month antiviral cytomegalovirus (CMV) prophylaxis.
Patients and methods
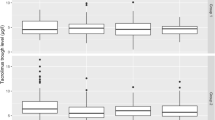
133 patients received a three-month combined intravenous and oral CMV prophylaxis with Ganciclovir (Cymevene®) after HTX between 1997 and April 2003 (group II). They were compared to a historical group consisting of 40 patients, who had undergone HTX between 1995 and 1996 (group I; CMV-prophylaxis: hyperimmune globuline (Cytotect®) for the first post-operative month in combination with orally administered aciclovir (Zovirax®) for 6 months).
Demographic data of organ recipients and donors in both groups were comparable, except for underlying cardiac diseases (p=0.016). All patients had identical postoperative immunosuppressive regimes.
Results
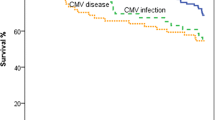
Group II had a significantly lower mortality rate (GI: 37.5%, GII: 9.8%; p<0.001); one year survival (p=0.001) and overall survival (p=0.001) were significantly better than in group I. Patients of group II had fewer rejection episodes ≥ grade II ISHLT requiring treatment (p<0.001).
Group II presented significantly fewer positive CMV blood samples (p=0.005) and CMV infections (26% versus 47,5% in GI; p=0.008), and a later onset of infections after HTX than group I (group I with a mean interval of 5.8 weeks after HTX, group II: 24.8 weeks after HTX; p<0.001).
Conclusion
Incidence of CMV infection was significantly lowered under ganciclovir prophylaxis, infections occurred at a later time point after HTX, when patients were immunologically more competent. The proportion of higher grade rejection episodes was markedly reduced and survival was improved.
Zusammenfassung
Hintergrund und Fragestellung
In der frühen Phase nach Herztransplantation (HTX), der Zeit intensivster Immunsuppression, haben Patienten ein hohes Infektionsrisiko. Diese retrospektive Kohortenstudie evaluiert die Wirksamkeit einer dreimonatigen antiviralen Cytomegalievirus (CMV)-Prophylaxe.
Patienten und Methodik
133 Patienten (Gruppe II: HTX 1997 — April 2003) erhielten eine dreimonatige kombiniert intravenös-orale CMV-Prophylaxe (Ganciclovir-Cymevene®) und wurden mit einer historischen Gruppe I (40 Patienten, HTX 1995-1996; CMV-Prophylaxe: CMV-Hyperimmunglobulin [Cytotect®] im 1. postoperativen Monat; Aciclovir [Zovirax®] oral über 6 Monate) verglichen.
Demographische Daten der Spender und Ernpfänqer beider Gruppen zeigten keine relevanten Unterschiede (ausgenommen cardiale Grunderkrankungen; p=0,016). In beiden Gruppen wurde ein identes immunsuppressives Regime verwendet.
Ergebnisse
In Gruppe II war die Mortalität hochsignifikant geringer (GI: 37,5%, GIl: 9,8%, p<0,001), das Einjahresüberleben (p=0,001) und das Gesamtüberleben (p 0,001) hochsignifikant besser als in Gruppe I. Patienten der Gruppe II hatten hochsignifikant weniger höhergradige, therapiewürdige Abstoßungen (≥ Grad II nach ISHLT) (p<0,001).
In Gruppe II fanden sich signifikant weniger positive CMV-Bluttests (p=0,005) und CMV-Infektionen (25,6% vs. 47,5% in GI; p=0,008), diese traten hochsignifikant später nach der HTX auf als in Gruppe I (GI: im Mittel 5,8 Wochen nach HTX, GII: 23,6 Wochen, p<0,001).
Folgerungen
Unter Ganciclovirprophylaxe war die Inzidenz der CMV-Infekte nicht nur signifikant reduziert, sie wurden zudem in eine spätere Phase nach HTX, in der Patienten immunologisch wiederum kompetenter sind, verschoben. Höhergradige Abstoßungen waren in der Ganciclovirgruppe hochsignifikant seltener, die zudem ein verbessertes Einjahres- und Langzeitüberleben aufweist.
Article PDF
Similar content being viewed by others
Literatur
Laczkovics A, Havel M, Laufer G, Teufelsbauer H, Gollner C, Wolner E (1987) Orthotopic heart transplantation — experiences at the University Surgical Clinic II in Vienna (status June 1986). Wien Klin Wochenschr 99: 333–340
Laufer G, Havel M, Holzinger C, Wollenek G, Laczkovics A, Wolner E (1989) Orthotopic heart transplantation at the 2nd Surgical Department of the University of Vienna: 4-year experience. Wien Klin Wochenschr 101: 53–57
Petri WA (1994) Infections in heart transplant recipients. Clin Infect Dis 18: 141–148
Aulitzky WE, Hengster P, Tilg H, Schulz T, Dierich M, Huber C (1988) Cytomegalovirus infection. Wien Klin Wochenschr 100: 33–43
Avery RK, Pappas PG (2002) Infections after heart (and heart/lung) transplantation. In: Kirklin JK, Young JB, McGiffin DC (eds) Heart Transplantation. Churchill Livingstone, New York Edinburgh London Philadelphia, pp 521–583
Rubin RH (2001) Gastrointestinal infectious disease complications following transplantation and their differentiation from immunosuppressant-induced gastrointestinal toxicities. Clin Transplantation 15 [Suppl 4]: 11–22
Grattan MT, Moreno-Cabral CE, Starnes VA, Oyer PE, Stinson EB, Shumway NE (1989) Cytomegalovirus infection is associated with cardiac allograft rejection and atherosclerosis. JAMA 261: 3561–3566
Loebe M, Schüler S, Spiegelsberger S, Warnecke H, Fleck E, Hetzer R (1990) Cytomegalievirus-Infektion und Koronarsklerose nach Herztransplantation. Deutsch Med Wschr 115: 1266–1269
Valantine HA, Gao SZ, Menon SG et al (1999) Impact of prophylactic immediate posttransplant ganciclovir on development of transplant atherosclerosis. A post-hoc analysis of a randimized, placebo-controlled study. Circulation 100: 61–66
George MJ, Snydman DR, Werner BG, Griffith J, Falagas ME, Dougherty NN, Rubin RH (1997) The independent role of cytomegalovirus as a risk factor for invasive fungal disease in orthotopic liver transplant recipients. Am J Med 103: 106–113
Gorensek MJ, Stewart RW, Keys TF, McHenry MC, Babiak T, Goormastic M (1988) Symptomatic cytomegalovirus infection as a significant risk factor for major infections after cardiac transplantation. J Infect Dis 158: 884–887
Manez R, Breinig MC, Linden P, Wilson J, Torre-Cisneros J, Kusne S et al (1997) Posttransplant lymphoproliferative disease in primary Epstein-Barr virus infection after liver transplantation: the role of cytomegalovirus disease. J Infect Dis 176: 1462–1467
Rubin RH (2000) Prevention of cytomegalovirus infection in organ transplant recipients. Editorial. Transplant Infectious Disease 2: 99–100
Griffiths PD (1997) Prophylaxis against CMV infection in transplant patients. J Antimicrob Chemother 39: 299–301
Costanzo-Nordin MR, Swinnen LJ, Fisher SG, et al (1992) Cytomegalovirus infections in heart transplant recipients: relationship to immunosuppression. J Heart Lung Transplant 11: 837–846
Valenza M, Czer LSC, Pan S-H, et al (1995) Combined antiviral and immunoglobulin therapy as a prophylaxis against cytomegalovirus infection after heart transplantation. J Heart Lung Transplant 14: 659–665
McCarthy JF, McCarthy PM, Massad MG, et al (1998) Risk factors for death after heart transplantation: does a single-center experience correlate with multicenter registries? Ann Thorac Surg 65: 1574–1579
Akposso K, Rondeau E, Haymann JP, Peraldi MN, Marlin C, Sraer JD (1997) Long-term prognosis of renal transplantation after preemptive treatment of cytomegalovirus infection. Transplantation 63: 974–976
Antretter H, Höfer D, Klaus A, Larcher C, Margreiter J, Margreiter R (2000) Cytomegalievirus-Infektion nach Herztransplantation. Retrospektive Analyse einer antiviralen CMV-Prophylaxe. Dtsch Med Wochenschr 125: 445–451
Künzle N, Petignat C, Francioli P, et al (2000) Preemptive treatment approach to cytomegalovirus (CMV) infection in solid organ transplant patients: relationship between compliance with the guidelines and prevention of CMV morbidity. Transpl Infect Dis 2: 118–126
Merigan TC, Renlund DG, Keay S, et al (1992) A controlled trial of ganciclovir to prevent cytomegalovirus disease after heart transplantation. N Engl J Med 326: 1182–1186
Patel R, Snydman DR, Rubin RH, et al (1996) Cytomegalovirus prophylaxis in solid organ transplant recipients. Transplantation 61: 1279–1289
Winston DJ (1995) Prevention of cytomegalovirus disease in transplant recipients. Lancet 346: 1380–1381
Antretter H, Pölzl G, Margreiter J, Hörmann Ch, Ott H, Margreiter R, Laufer G (2002) Successful transfer of a cardiac allograft from a heterotop to an orthotopic position 16 years after heart transplantation. Transplantation 74: 540–543
Shumway NE, Lower RR, Stofer RC (1966) Transplantation of the heart. Adv Surg 2: 265–284
Antretter H, Laufer G. Surgical techniques for cardiac transplantation (2001) Acta Chir Austriaca 33: 17–24
Iberer F, Tscheliessnigg KH, Halwachs G, Auer T, Wasler A, Petutschnigg B, et al (1995) CMV antigenemia (the lower matrix protein PP65), a marker for the guidance of antiviral therapy in cytomegalovirus disease after orthotopic heart transplantation. Wien Klin Wochenschr 107: 718–722
Fisman JA, Rubin RH (1998) Infection in organ transplant recipients. N Engl J Med 338: 1741–1751
Koskinen PK, Kallio EA, Tikkanen JM, Sihvola RK, Hayry PJ, Lemstrom KB (1999) Cytomegalovirus infection and cardiac allograft vasculopathy. Transpl Infect Dis 1: 115–126
Paniagua MJ, Crespo-Leiro MG, De la Fuente L, et al (2002) Prevention of cytomegalovirus disease after heart transplantation: preemptive therapy with 7 days’ intravenous ganciclovir. Transplant Proceedings 34: 69–70
Schirm J, Kooistra A, van Son WJ, van der Bij W, Verschuuren E, Sprenger HG et al (1999) Comparison of the Murex Hybrid Capture CMV DNA (v2.0) assay and the pp65 CMV antigenemia test for the detection and quantitation of CMV in blood samples from immunocompromised patients. J Cin Virol 14: 153–165
Valantine HA, Luikart H, Ramona Doyle, et al (2001) Impact of cytomegalovirus hyperimmune globuline on outcome after cardiothoracic transplantation. Transplantation 72: 1647–1652
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Antretter, H., Höfer, D., Hangler, H. et al. Können CMV-Infekte nach Herztransplantation durch dreimonatige antivirale Prophylaxe reduziert werden? 7 Jahre Erfahrung mit Ganciclovir. Wien Klin Wochenschr 116, 542–551 (2004). https://doi.org/10.1007/BF03217708
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03217708