Abstract
This paper is concerned with a mathematical analysis of the modified Guggenheim procedure. Theorems concerning the solutions of the differential equations which describe the general reaction
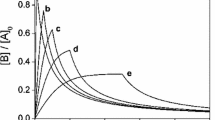
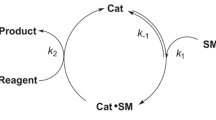
are derived and are used in a general discussion of the validity of the procedure. Numerical results are given to illustrate the general assertions and to obtain the range of values ofE 0 andS 0 for which the procedure is valid and enzymatic reaction such as α-chymotrypsin.
Similar content being viewed by others
Literature
Filmer, D. L. 1964. To appear.
Gutfreund, H. and J. M. Sturtevant. 1956. “The Mechanism of the Reaction of Chymotrypsin with p-Nitrophenyl Acetate.”Biochem. Jour.,63, 656.
Kezdy, F. J. and M. L. Bender. 1962. “The Kinetics of the α Chrymotrypsin-Catalyzed Hydrolysis of p-Nitrophenyl Acetate.”Biochemistry,1, 1097.
Spencer, T. and J. M. Sturtevant. 1959. “The Mechanism of Chymotrypsin-Catalyzed Reactions.”Jour. Am. Chem. Soc.,81, 1874.
Author information
Authors and Affiliations
Additional information
This work performed under the auspices of the U.S. Atomic Energy Commission.
Rights and permissions
About this article
Cite this article
Filmer, D.L., Cannon, J.R. Analysis of a procedure for the determination of kinetic rate constants. Bulletin of Mathematical Biophysics 27, 253–263 (1965). https://doi.org/10.1007/BF02478402
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02478402