Abstract
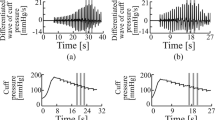
This double-blind, placebo-controlled, randomized cross-over study investigates the influence of aescin on the capillary resistance tested on 12 persons with healthy vessels. The quantification of the human-pharmacological diagnosis ensued from a computerized variant of the capillary-resistometry (petechiae test) developed by us, which can determine and document the capillary resistance under standardized conditions independent of the examiner. The isochronous parallel measurements carried out on each test person differ considerably from one another—with placebo as well as with verum treatment. The verum-treated test persons in phase I show remarkable lower petechiae values in phase II (carry-over effect). After a distinct decrease of the values at the beginning of the treatment, the values of those tested remain at a low level until the end of the period of observation. In the verum treatment the amount of petechiae as well as the petechiae area is already significantly lower after 7 days than in the initial examination. A decrease of the values in the placebo treatment cannot be detected.
Similar content being viewed by others
References
Küchmeister H (1953) Läßt sich die Wirkung des Roßkastanienextraktes auf die Kapillarfunktion objektivieren? Ärztl. Forschung 7:102–104.
Galley P (1987) Study of the activity of Daflon 500 mg on capillary resistance. J Inter Med (suppl) 88:25–26.
Wienert V (1994) The measurement of capillary permeability using computer-aided capillary resistometry clinical hemorheology 14:47–52.
Author information
Authors and Affiliations
About this article
Cite this article
Wienert, V. Efficacy of aescin on the capillary fragility in men. International Journal of Angiology 6, 115–117 (1997). https://doi.org/10.1007/BF01616680
Issue Date:
DOI: https://doi.org/10.1007/BF01616680