Summary
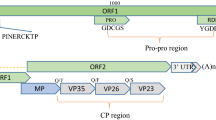
The complete nucleotide sequence of the genomic RNA of odontoglossum ringspot tobamovirus (ORSV) was determined. The RNA genome of ORSV is 6618 nucleotides long and contains five open reading frames (ORFs 1 to 5) coding for proteins of Mr 126 K, 181 K, 34 K, 18 K and 52 K, respectively. This is the longest RNA of the known viruses of theTobamovirus genus. The sequences of the ORSV RNA encoded proteins exhibit high homology to the proteins of the members of theTobamovirus genus. The genomic organization and sequence analysis showed that ORSV is more closely related to tobacco mild green mosaic virus (TMGMV), pepper mild mottle virus (PMMV), tomato mosaic virus (ToMV) and TMV than to cucumber green mottle mosaic virus (CGMMV) and sunn-hemp mosaic virus (SHMV).
Similar content being viewed by others
References
Alonso E, Garcia-Luque I, de la Cruz A, Wicke B, Avila-Rincon MJ, Serra MT, Castresana C, Diaz-Ruiz JR (1991) Nucleotide sequence of the genomic RNA pepper mild mottle virus, a resistance-breaking tobamovirus in pepper. J Gen Virol 72: 2875–2884
Argos P (1988) A sequence motif in many polymerases. Nucleic Acids Res 16: 9909–9916
Avila-Rincon MJ, Ferrero ML, Alonso E, Garcia-Luque I, Diaz-Ruiz JR (1989) Nucleotide sequences of 5′ and 3′ non-coding regions of pepper mild mottle virus strain S RNA. J Gen Virol 70: 3025–3031
Bruenn JA (1991) Relationships among the positive strand and double-strand RNA viruses as viewed through their RNA-dependent RNA polymerases. Nucleic Acids Res 19: 217–226
Calder VL, Palukaitis P (1992) Nucleotide sequence analysis of the movement genes of resistance breaking strains of tomato mosaic virus. J Gen Virol 73: 165–168
Carr JP, Marsh LE, Lomonossoff GP, Sekiya ME, Zaitlin M (1992) Resistance to tobacco mosaic virus induced by the 54-kDa gene sequence requires expression of the 54-kDa protein. Mol Plant Microbe Interact 5: 397–404
Culver JN, Lehto K, Close SM, Hilf ME, Dawson WO (1993) Genomic position affects the expression of tobacco mosaic virus movement and coat protein genes. Proc Natl Acad Sci USA 90: 2055–2059
Deom O, Oliver MJ, Beachy RN (1987) The 30-kilodalton gene product of tobacco mosaic virus potentiates virus movement. Science 237: 389–394
Dubs MC, Van Regenmortel MHV (1990) Odontoglossum ringspot virus coat protein: sequence and antigenic comparisons with other tobamoviruses. Arch Virol 115: 239–249
Edwardson JR, Zettler FW (1986) Odontoglossum ringspot virus. In: Van Regenmortel MHV, Fraenkel-Conrat H (eds) The plant viruses, vol 2. Plenum Press, New York, pp 233–247
Gallie DR, Sleat DE, Watts JW, Turner PC, Wilson TMA (1987) The 5′ leader sequence of tobacco mosaic virus RNA enhances the expression of foreign gene transcriptsin vitro andin vivo. Nucleic Acids Res 15: 3257–3273
Gallie DR, Sleat DE, Watts JW, Turner PC, Wilson TMA (1988) Mutational analysis of the tobacco mosaic virus 5′ leader sequence for altered ability to enhance translation. Nucleic Acids Res 16: 883–893
Garcia-Arenal F (1988) Sequence and structure at the genome 3′ end of the U2-strain of tobacco mosaic virus, a histidine-accepting tobamovirus. Virology 166: 201–206
Gibbs AJ (1986) Tobamovirus classification. In: Van Regenmortel MHV, Fraenkel-Conrat H (eds) The plant viruses, vol 2. Plenum Press, New York, pp 167–180
Goelet P, Lomonossoff GP, Butler PJG, Akam ME, Gait MJ, Karn J (1982) Nucleotide sequence of tobacco mosaic virus RNA. Proc Natl Acad Sci USA 79: 5818–5822
Gubler U, Hoffman BJ (1983) A simple and very efficient method for generating cDNA libraries. Gene 25: 263–269
Habili N, Symons RH (1989) Evolutionary relationship between luteoviruses and other RNA plant viruses based on sequence motifs in their putative RNA polymerases and nucleic acid helicases. Nucleic Acids Res 17: 9543–9555
Helmut B, Cribb P, Launert E (1992) The manual of cultivated orchid species, 3rd ed. American Orchid Society, Florida
Hodgman TC (1988) A new superfamily of replicative proteins. Nature 335: 22–23
Isomura Y, Matumoto Y, Murayama A, Chatani M, Inouye N, Ikegami M (1991) Molecular cloning, sequencing and expression inEscherichia coli of the odontoglossum ringspot virus coat protein gene. J Gen Virol 72: 2247–2249
Jensen DD, Gold AH (1951) A virus ring spot ofOdontoglossum orchids: symptoms, transmission, and electron microscopy. Phytopathology 41: 648–653
Konarska MM, Padgett RA, Sharp PA (1984) Recognition of cap structure in splicingin vitro of mRNA precursors. Cell 38: 731–736
Kukla BA, Guilley HA, Jonard GX, Richards KE, Mundry KW (1979) Characterization of long guanosine-free RNA sequences from Dahlamense and U2 strains of tobacco mosaic virus. Euro J Biochem 98: 61–66
MacFarlane SA, Davies JW (1992) Plants transformed with a region of the 201-kilodalton replicase gene from pea early browning virus RNA1 are resistant to virus infection. Proc Natl Acad Sci USA 89: 5829–5833
Meshi T, Ohno T, Iba H, Okada Y (1981) Nucleotide sequence of a cloned cDNA copy of TMV (cowpea strain) RNA, including the assembly origin, the coat protein cistron, and the 3′ noncoding region. Mol Gen Genet 184: 20–25
Meshi T, Ohno T, Okada Y (1982) Nucleotide sequence of the 30 K protein cistron of cowpea strain of tobacco mosaic virus. Nucleic Acids Res 10: 6111–6117
Mushegian AR, Koonin EV (1993) Cell-to-cell movement of plant viruses. Arch Virol 133: 239–257
Ohno T, Aoyagi M, Yamanashi Y, Saito H, Ikawa S, Meshi T, Okada Y (1984) Nucleotide sequence of the tobacco mosaic virus (tomato strain) genome and comparison with the common strain genome. J Biochem 96: 1915–1923
Padgett HS, Beachy RN (1993) Analysis of tobacco mosaic virus strain capable of overcoming N gene-mediated resistance. Plant Cell 5: 577–586
Park WM, Yoon KE, Chung SY, Ryu KH (1990) Identification of odontoglossum ringspot virus isolated fromCymbidium goeringii Reichenbach in Korea. Korean J Plant Pathol 6: 387–392
Park WM, Yoon KE, Chung SY, Ryu KH (1990) Purification and serological detection of odontoglossum ringspot virus isolated fromCymbidium goeringii in Korea. Korean J Plant Pathol 6: 474–481
Richards K, Jonard G, Guilley H, Keith G (1977) Leader sequence of 71 nucleotides devoid of G in tobacco mosaic virus RNA. Nature 267: 548–550
Rietveld K, Linschooten K, Pleij CWA, Bosch L (1984) The three dimensional folding of the tRNA-like structure of tobacco mosaic virus RNA: a new building principle applied twice. EMBO J 3: 2613–2619
Ryu KH, Park WM (1994) Construction of complementary DNA library and cDNA cloning for Cy strain of odontoglossum ringspot virus genomic RNA. Korean J Plant Pathol 10: 228–234
Ryu KH, Choi CW, Choi JK, Park WM (1995) Cloning of the 3′-terminal region encoding movement and coat proteins of a Korean isolate of odontoglossum ringspot virus. Arch Virol 140: 481–490
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74: 5463–5467
Sleat DE, Gallie DR, Jefferson RA, Bevan MW, Turner PC, Wilson TMA (1987) Characterization of the 5′-leader sequence of tobacco mosaic virus RNA as a general enhancer of translationin vitro. Gene 217: 217–225
Solis I, Garcia-Arenal F (1990) The complete nucleotide sequence of the genomic RNA of the tobamovirus tobacco mild green mosaic virus. Virology 177: 553–558
Sulzinsky MA, Gabard KA, Palukaitis P, Zaitlin M (1985) Replication of tobacco mosaic virus. VIII. Characterization of a third subgenomic TMV RNA. Virology 145: 132–140
Tyc K, Kornaska M, Gross HJ, Filipowicz W (1984) Multiple ribosome binding to the 5′-terminal leader sequence of tobacco mosaic virus RNA, assembly of an 80S ribosome mRNA complex at the AUU codon. Eur J Biochem 140: 503–511
Ugaki M, Tomiyama M, Kakutani T, Hidaka S, Kiguchi T, Nagata R, Sato T, Motoyoshi F, Nishiguchi M (1991) The complete nucleotide sequence of cucumber green mottle mosaic virus (SH strain) genomic RNA. J Gen Virol 72: 1487–1495
Valverde RA, Dodds JA, Heick JA (1986) Double-stranded ribonucleic acid from plants infected with viruses having elongated particles and undivided genomes. Phytopathology 76: 459–465
Zettler FW, Ko NJ, Wisler GC, Elliot MS, Wong SM (1990) Viruses of orchids and their control. Plant Dis 74: 621–626
Author information
Authors and Affiliations
Additional information
The nucleotide sequence reported in this paper will appear in the EMBL, GenBank and DDBJ nucleotide sequence databases under the accession number X82130.
Rights and permissions
About this article
Cite this article
Ryu, K.H., Park, W.M. The complete nucleotide sequence and genome organization of odontoglossum ringspot tobamovirus RNA. Archives of Virology 140, 1577–1587 (1995). https://doi.org/10.1007/BF01322531
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01322531