Summary
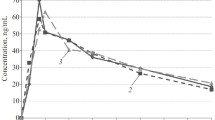
Hydrochlorothiazide (hct) was administered orally in four different doses (12.5, 25, 50 and 75 mg), to eight healthy volunteers. Plasma and urine concentrations of hct were determined by GLC. Maximal plasma levels were found at 1.5–5 h, and averaged 70, 142, 260 and 376 ng × ml−1 respectively. The peak plasma levels and AUC0→9h of hct were highly correlated (p<0.001) with the dose. The decline in the plasma curve was biphasic in those experiments in which the plasma levels of hct could be determined for at least 24 h. The half life of the slower phase lay between 5.6 and 14.8 h. The urinary recovery of hct, which represented the gastrointestinal absorption, averaged 65 to 72 per cent of the dose. The mean renal plasma clearance did not vary with the dose and averaged 319 to 345 ml × min−1. The diuresis during the 10 h after hct 12.5 mg exceeded that after placebo by a mean of 800 ml. The diureses was not increased further after higher doses of hct. The maximal natriuretic effect (+ 100 mmol), too, was found after the 12.5 mg dose. The excretion of potassium, however, rose with increasing doses; the maximal increment, after 75 mg hct, averaged 25 mmol. The excretion of calcium was significantly increased after 50 mg hct (+ 0.6 mmol). The maximal effect on magnesium excretion occurred after 25 mg hct (+ 0.5 mmol). In healthy volunteers there was no correlation between peak plasma level of hct or AUC0→9h and the renal excretion of water and electrolytes.
Similar content being viewed by others
References
Beermann, B., Groschinsky-Grind, M., Rosén, A.: Absorption, metabolism and excretion of hydrochlorothiazide. Clin. Pharmacol. Ther.19, 531–537 (1976)
Beermann, B., Groschinsky-Grind, M., Lindström, B.: Bioavailability of two hydrochlorothiazide preparations. Europ. J. clin. Pharmacol.10, 293–295 (1976)
Beerman, B., Groschinsky-Grind, M., Lindström, B.: On the pharmacokinetics of Bendroflumethiazide in man. Clin. Pharmacol. Ther. (In press)
Brickman, A. S., Coburn, J. W., Koppel, M H., Peacock, M., Massey, S. G.: The effect of Hydrochlorothiazide administration on serum and urinary calcium in normal, hypoparathyroid and hyperparathyroid subjects. Israel. J. med. Sci.7, 518–519 (1971)
Ford, R.: The clinical pharmacology of hydrochlorothiazide. South. Med. J.52, 40–45 (1959)
Gilmore, J. P., O'Brien, W. M., Brand, E. D., Peach, M. J., Westfall, T. C.: A student exercise in clinical pharmacology. Renal effects of diuretics. Curr. Ther. Res.12, 750–769 (1970)
Kill, F.: Permutation trial of diuretics chlorothiazide and hydroflumethiazide. Circulation21, 717 (1960)
Lindström, B., Molander, M., Groschinsky-Grind, M.: Gas chromatographic determination of hct in plasma, blood corpuscles and urine using extractive alkylation technique. J. Chromatogr.101, 219–221 (1975)
Mertz, D.: Pharmakologische Eigenschaften von Hydrochlorothiazide im Vergleich zur Wirkung anderer Diuretika. Naunyn-Schmiedeberg's Arch. Exp. Path. Pharmak.237, 71–93 (1959)
Middler, S., Pak Ch. Y. C., Murad, F., Bartter, F. C.: Thiazide Diuretics and Calcium metabolism. Metabolism22, 139–146 (1973)
Moyer, J. H., Fuchs, M., Shinazo, I., Tibor, B.: Some observations on the pharmacology of Hydrochlorothiazide. J. Cardiol.3, 113–117 (1959)
Parfitt, A. M.: The acute effects of mersalyl, chlorothiazide and mannitol on the renal excretion of calcium and other ions in man. Clin. Sci.36, 267–282 (1969)
Robinson, R. R., Murdaugh, H. V., Peschel, E.: Urinary excretion of magnesium in renal disease. Circulation18, 771 (1958)
Seitz, H., Jaworski, Z. F.: Effect of Hydrochlorothiazide on serum and urinary calcium and urinary citrate. Canad. med. Ass. J.90, 414–420 (1964)
Author information
Authors and Affiliations
Additional information
Financially supported by the Swedish Medical Research Council (Grant Nr. R 76-04x-227-12c) and Hässle-Ciba-Geigy AB, Gothenburg, Sweden
Rights and permissions
About this article
Cite this article
Beermann, B., Groschinsky-Grind, M. Pharmacokinetics of hydrochlorothiazide in man. Eur J Clin Pharmacol 12, 297–303 (1977). https://doi.org/10.1007/BF00607430
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00607430