Abstract
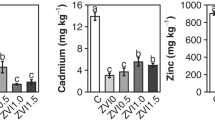
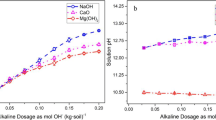
Because the economics of soil extraction processes depend on conservation and reuse of costly chelating agents, the ability of various electrolytes to modify EDTA extraction of Pb from a grossly-contaminated soil (PbT=21%) was investigated using batch equilibration experiments. In the absence of added electrolyte, a single 5-hr. extraction with 0.04 M EDTA (corresponding to 1∶1 PbT to EDTA ratio) released 65% of PbT over the pH 5 to 9 range. Under these conditions, Na+-, Li+-, and NH4ClO4 salts at 0.5 M increased Pb desorption to nearly 80%, probably from exchange displacement of soilbound Pb2+ and increased solubility of Pb-containing phases at higher ionic strength. Because Cl− and ClO4 − salts were equally effective, chlorocomplex formation was insignificant. Under slightly acidic conditions, Ca(ClO4)2 and Mg(ClO4)2 at 0.167 M caused roughly the same elevation in Pb recovery as 0.5M of the monovalent electrolytes. However, with progressively higher pH, Ca, and to a lesser extent Mg, suppressed Pb solubilization by competitive chelation of EDTA. Pb recovery by EDTA soil washing could be enhanced by addition of Ca salts at pH 4 to 6. Subsequent pH elevation in the presence of Ca would promote decomposition of Pb-EDTA complexes and separation of Pb as a hydroxide precipitate.
Similar content being viewed by others
References
Ahumada, I. A., Schalscha, E. B., Pratt, P. F., and Mattigod, S. V.: 1984, Soil Sci. Soc. Am. J. 48, 1240.
Borggaard, O. K.: 1979, J. Soil Sci. 30, 727.
Bowen, H. J. M.: 1966, Trace Elements in Biochemistry, Academic Press, New York.
Elliott, H. A. and Brown, G. A.: 1989, Water, Air, and Soil Pollution 45, 361.
Elliott, H. A., Linn, J. H., and Shields, G. A.: 1989, Hazard. Wastes Hazard. Mat. 6, 223.
Ellis, W. D., Fogg, T. R., and Taturi, A. N.: 1986, 12th Annual Research Symposium on Land Disposal, Remedial Action, Incineration and Treatment of Hazardous Waste, US EPA Hazardous Waste Engineering Research Laboratory, Cincinnati, OH.
Elsokkary, I. H.: 1980, Plant and Soil 54, 383.
Farrah, F. and Pickering, W. F.: 1977, Water, Air, and Soil Pollution 8, 189.
Hill-Cottingham, D. G. and Lloyd-Jones, C. P.: 1957, Plant and Soil 8, 263.
Jackson, D. R. and Watson, A. P.: 1977, J. Env. Qual. 6, 331.
Kobayashi, J., Morii, F., and Muramoto, S.: 1974, Trace Subst. Environ. Health 8, 179.
Lagerwerff, J. V. and Specht, A. W.: 1970, Environ. Sci. Tech. 4, 583.
Martell, A. E. and Smith R. M.: 1976, Critical Stability Constants, Plenum Press, New York.
Miller, W. P., Martens, D. C, and Zelazny, L. W.: 1986, Soil Sci. Soc. Am. Proc. 50, 598.
Motekaitis, R. J. and Martell, A. E.: 1985, J. Coord. Chem. 14, 49.
Neal, C: 1977, Clays and Clay Minerals 25, 253.
Norvell, W. A. and Lindsay, W. L.: 1969, Soil Sci. Soc. Am. Proc. 33, 86.
O'Connor, T. P. and Kester, D. R.: 1975, Geochim. Cosmochim. Acta 39, 1531.
Papanicolaou, E. P.: 1976, Soil Sci. 121, 65.
Pickering, W. F.: 1983, Water, Air, and Soil Pollution 20, 299.
Pickering, W. F.: 1986, Ore. Geology Reviews 1, 83.
PEL 1986, Hazardous Waste Consultant, March/April, 13.
Rhoades, J. D., and Krueger, D. R.: 1968, Soil Sci. Soc. Am. Proc. 32, 488.
Ringbom, A.: 1963, Complexation in Analytical Chemistry, Wiley Interscience, New York.
Stenstrom, T. and Vahter, M.: 1974, Ambio 3, 91.
Tessier, A., Campbell, P. G. C., and Bisson, M.: 1979, Anal. Chem. 51, 844.
Wiklander, L.: 1964, ‘Cation and Anion Exchange Phenomena’, in: F. E. Bear (ed.), Chemistry of the Soil, 2nd Ed., Reinhold, New York.
Wing, R., Doane, W., and Rayford, W.: 1977, Plating and Surface Finishing 64, 57.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brown, G.A., Elliott, H.A. Influence of electrolytes on EDTA extraction of Pb from polluted soil. Water Air Soil Pollut 62, 157–165 (1992). https://doi.org/10.1007/BF00478458
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00478458