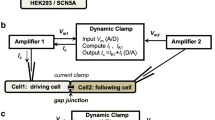
Abstract
Cell pairs of neonatal rat hearts were used to study the influence of temperature on the electrical properties of gap junctions. A dual voltage-clamp method was adopted, which allowed the voltage gradient between the cells to be controlled and the intercellular current flow to be measured. Cell pairs with normal coupling revealed a positive correlation between the conductance of the junctional membranes, g j, and temperature. Cooling from 37° C to 14° C led to a steeper decrease in g j, cooling from 14° C to -2° C to a shallower decrease (37° C: g j=48.3 nS; 14° C: g j=21.4 nS;-2°C: g j=17.5 nS), corresponding to a temperature coefficient, Q 10, of 1.43 and 1.14 respectively. The existence of two Q 10 values implies that g j may be controlled by enzymatic reactions. When g j was low, i. e. below 5 nS (conditions: low temperature; treatment with 3 mM heptanol), it showed voltage-dependent gating. This property was not visible when g j was large, i. e. 20–70 nS (conditions: high temperature; normal saline), presumably because of series resistances (pipette resistance). Cell pairs with weak intrinsic coupling and normally coupled cell pairs treated with 3 mM heptanol revealed a positive correlation between the conductance of single gap-junction channels, γ j, and temperature (37° C: 75.6 pS; -2°C: 19.6 pS), corresponding to a Q 10 of 1.41.
Similar content being viewed by others
References
Burt JM, Spray DC (1988) Single-channel events and gating behavior of the cardiac gap junction channel. Proc Natl Acad Sci USA 85:3431–3434
Chen Y, DeHaan R (1989) Cardiac gap junction channels shift to lower conductance states when temperature is reduced. Biophys 155:152 a
Coraboeuf E, Weidmann S (1954) Temperature effects on the electrical activity of Purkinje fibres. Helv Physiol Pharmacol Acta 12:32–41
Fatt P, Katz B (1953) The electrical properties of crustacean muscle fibres. J Physiol (Lond) 120:171–204
Hille B (1984) Ionic channels of excitable membranes. Sinauer, Sunderland, pp 49, 195–198
Hladky SB, Haydon DA (1972) Ion transfer across lipid membranes in the presence of gramicidin A. Biochim Biophys Acta 274:294–312
Imanaga I, Kameyama M, Irisawa H (1987) Cell-to-cell diffusion of fluorescent dyes in paired ventricular cells. Am J Physiol 252: H 223-H 232
Kolb H-A, Somogyi R (1991) Biochemical and biophysical analysis of cell-to-cell channels and regulation of gap junction permeability. Rev Physiol Biochem Pharmacol 118:1–47
Lau AF, Hatch-Pigott V, Crow DS (1991) Evidence that heart connexin 43 is a phosphoprotein. J Mol Cell Cardiol 23:659–663
Moreno AP, Fishman GI, Spray DC (1992) Phosphorylation shifts unitary conductance and modifies voltage dependent kinetics of human connexin 43 gap junction channels. Biophys J 62:51–53
Niggli E, Rüdisüli A, Maurer P, Weingart R (1989) Effects of general anesthetics on current flow across membranes in guinea pig myocytes. Am J Physiol 256:C 273-C 281
Payton BW, Bennettt MVL, Pappas GD (1969) Temperaturedependence of resistance at an electrotonic synapse. Science 163:594–597
Robinson RA, Stokes RH (1970) Electrolyte solutions. Butterworths, London, p 465
Rook MB, Jongsma HJ, van Ginneken ACG (1988) Properties of single gap junction channels between isolated neonatal rat heart cells. Am J Physiol 255:H 770-H 782
Rüdisüli A, Weingart R (1989) Electrical properties of gap junction channels in guinea-pig ventricular cell pairs revealed by exposure to heptanol. Pflügers Arch 415:12–21
Schmilinsky Fluri G, Rüdisüli A, Willi M, Weingart R (1990) Effects of arachidonic acid on the gap junctions of neonatal rat heart cells. Pflügers Arch 417:149–156
Sugiura H, Toyama J, Tsuboi N, Kamiya K, Kodama I (1990) ATP directly affects junctional conductance between paired ventricular myocytes isolated from guinea pig heart. Circ Res 66:1095–1102
Szekeres L, Papp GJ (1971) Experimental cardiac arrhythmias and antiarrhythmic drugs. Akadémiai Kiadó, Budapest, pp 93–151
Trautwein W, Gottstein U, Federschmidt K (1953) Der Einfluß der Temperatur auf den Aktionsstrom des excidierten Purkinje-Fadens, gemessen mit einer intrazellulären Elektrode. Pflügers Arch 258:243–260
Veenstra RD (1990) Voltage-dependent gating of gap junction channels in embryonic chick ventricular cell pairs. Am J Physiol 258:C662-C672
Verselis V, Brink PR (1986) The gap junction channel. Its aqueous nature as indicated by deuterium oxide effects. Biophys 150:1003–1007
Weingart R (1986) Electrical properties of the nexal membrane studied in rat ventricular cell pairs. J Physiol (Lond) 370:267–284
Weingart R, Rüdisüli A, Maurer P (1990) Cell to cell communication. In: Zipes DP, Jalife J (eds) Cardiac electrophysiology. Saunders, Philadelphia, pp 122–127
White RL, Spray DC, Campos de Carvalho AC, Wittenberg BA, Bennett MVL (1985) Some electrical and pharmacological properties of gap junctions between adult ventricular myocytes. Am J Physiol 249:C 447-C 455
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bukauskas, F.F., Weingart, R. Temperature dependence of gap junction properties in neonatal rat heart cells. Pflugers Arch. 423, 133–139 (1993). https://doi.org/10.1007/BF00374970
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374970