Abstract
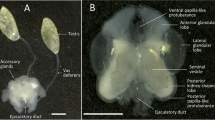
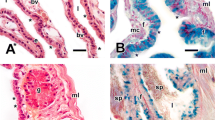
Male red frog crabs, Ranina ranina, were collected year round in 1990 and 1991 off Hachijojima for histological study of the reproductive system and cycle. The testis containing the lobules and seminiferous ducts is surrounded by connective tissue. The seminiferous duct connects to the anterior end of the vas deferens, which can be histologically divided into three portions similar to one another in appearance. It was surrounded by fibrous connective tissue, muscle fibrils and columnar epithelium. Muscle fibrils were absent in the anterior portion. Multiple sperm masses were not formed in the vas deferens and ejaculatory duct, but the sperm mass was covered with a capsule composed of two layers. The outer layer of the capsule was periodic acid Schiff (PAS)-positive, but the inner layer was negative. Both layers were Alcian Blue negative, except the vacuoles in the outer layer that were stained blue. The small round androgenic gland was attached to the posterior end of the vas deferens of the coxa of the eighth thoracic appendage. The ejaculatory duct was distinguishable from the vas deferens by the absence of columnar epithelium and the presence of thick longitudinal muscle fibers. Spermatogenesis was histochemically examined. The acrosomal vesicle appeared to be derived from PAS-positive vesicles in the cytoplasm of the spermatid at the early stage of spermiogenesis. The arms were positive to the Feulgen reaction and the subacrosomal region was negative to PAS. Seasonal changes in reproductive cycle were inconspicuous histologically and microscopically. Sperm were always present in the testis and vas deferens throughout the year and occupied 5.1 to 19.6% of testis observed in cross sections. The minimum size of maturity is less than 39 mm carapace length, but the minimum size capable of successful mating was estimated to be ca. 55 mm.
Similar content being viewed by others
References
Barnard, K. H. (1950). Descriptive catalogue of South African decapod crustacea. Ann. S. Afr. Mus. 38: 396–399
Batoy, C. B., Sarmago, J. F., Pilapil, B. C. (1987) Breeding season, sexual maturity and fecundity of the blue crab, Portunus pelagicus (L.) in selected coastal waters in Leyte and vicinity, Philippines. Ann. trop. Res. 9: 157–177
Beninger, P. G., Elner, R. W., Foyle, T. P., Odense, P. H. (1988). Functional anatomy of the male reproductive system and the female spermatheca in the snow crab Chionoecetes opilio (O. Fabricius) (Decapoda: Majidae) and a hypothesis for fertilization. J. Crust. Biol. 8(3): 322–332
Brown, I. W. (1986). Population biology of the spanner crab in south-east Queensland. Final project report to Fishing Industry Research Committee. Queensland Department of Primary Industries, Queensland, p. 1–106
Charniaux-Cotton, H., Payen, G. (1985). Sex differentiation. In: Bliss, D. E., Mantel, L. H. (eds.). The biology of crustacea, Vol. 9. Integument, pigments, and hormonal processes. Academic Press, New York, p. 217–299
Chiba, A., Honma, Y. (1972). Studies on gonad maturity in some marine invertebrates-VI. Seasonal changes in the testis of the lined shore crab. Nippon Suisan Gakk. 38(4): 317–322
Cronin, L. E. (1947). Anatomy and histology of the male reproductive system of Callinectes sapidus Rathbun. J. Morph. 81: 209–239
Dhawan, R. M., Dwivedi, S. N., Rajamanickam, G. V. (1976). Ecology of the blue crab Portunus pelagicus (L.) and its potential fishery in Zuari Estuary. Indian J. Fish. 23(1–2): 57–64
Diesel, R. (1989). Structure and function of the reproductive system of the symbiotic spider crab Inachus phalangium (Decapoda: Majidae): observations on sperm transfer, sperm storage, and spawning. J. Crustacean Biol. (Lawrence, Kansas). 9(2): 266–277
El-Serief, S. S. (1988). Histological study of the secretions of the male reproductive system of Portunus pelagicus (L. Crustacea-Brachyura). Folia Morph. (Prague) 36: 95–106
Fielding, A., Haley, S. R. (1976). Sex ratio, size at reproductive maturity, and reproduction of the Hawaiian Kona crab, Ranina ranina (Linnaeus) (Brachyura, Gymnopleura, Raninidae). Pacific Sci. 30(2): 131–145
George, M. J. (1963). The anatomy of the crab Neptunus sanguinolentus Herbst. J. Madras Univ. (Sect. B) 33(3): 289–304
Gordon, I. (1963). On the relationship of Dromiacea, Tymolinae and Raninidae to the Brachyura. In: Whittington, H. B., Rolfe, W. D. I. (eds.). Phylogeny and evolution of crustacea. Museum of Comparative Zoology, Special Publication, Cambridge, p. 51–57
Hartnoll, R. G. (1979). The phyletic implications of spermathecal structure in the Raninidae (Decapoda: Brachyura). J. Zool. 187: 75–83
Harutsugu, M. (1960). Electron microscope studies on the spermatogenesis of Crustacea. J. Nara Med. Assoc. 11: 82–93
Hinsch, G. W. (1969). Microtubules in the sperm of the spider crab, Libinia emarginata L. J. Ultrastruct. Res. 29: 525–534
Hinsch, G. W. (1988a). Morphology of the reproductive tract and seasonality of reproduction in the golden crab Geryon fenneri from the eastern Gulf of Mexico. J. Crustacean Biol. (Lawrence, Kansas) 8(2): 254–261
Hinsch, G. W. (1988b). Ultrastructure of the sperm and spermatophores of the golden crab Geryon fenneri and a closely related species, the red crab G. quinquedens, from the eastern Gulf of Mexico. J. Crustacean Biol. (Lawrence, Kansas) 8: 340–345
Hinsch, G. W. (1991a). Structure and chemical content of the spermatophores and seminal fluid of reptantian decapods. In: Bauer, R. T., Martin, J. W. (eds). Crustacean sexual biology. Columbia University Press, New York, p. 290–307
Hinsch, G. W. (1991b). Ultrastructure of the sperm and spermatophores of the anomuran crab Pleuroncodes planipes. J. Crustacean Biol. (Lawrence, Kansas) 11(1): 17–22
Iwata, Y., Sugita, H., Kobayashi, T., Deguchi, Y., Kamemoto, F. I. (1987). Mating, spawning and molting of the Asahi-gani, Ranina ranina, in aquaria. Bull. Coll. Agric. Vet. Med., Nihon Univ. 44: 165–168
Jamieson, B. G. M. (1989). Ultrastructural comparison of the spermatozoa of Ranina ranina (Oxystomata) and of other crabs exemplified by Portunus pelagicus (Brachygnatha) (Crustacea, Brachyura). Zoomorph. 109: 103–111
Johnson, P. T. (1980). Histology of the Blue Crab, Callinectes sapidus. Praeger Publishers, New York. p. 3–15, 328–346
Joshi, P. C., Khanna, S. S. (1987). Studies on the androgenic gland of the freshwater crab, Potamon koolooense (Rathbun). Z. mikrosk.-anat. Forsch 101(4): 699–713
Kiriyama, M. (1958). Electron microscope study on spermatogenesis of Crustacea. J. Nara Med. Assoc. 9: 269–289
Kon, T., Honma, Y. (1970). Studies on the maturity of the gonad in some marine invertebrates-IV. Seasonal changes in the testis of the tanner crab. Nippon Suisan Gakk. 36(10): 1028–1033
Melville-Smith, R. (1987). The reproductive biology of Geryon maritae (Decapoda, Brachyura) off south west African/Nambia. Crustaceana 53(3): 259–275
Minagawa, M., Chiu, J.-R., Kudo, M., Ito, F., Takashima, F. (1993). Female reproductive biology and oocyte development of the red frog crab, Ranina ranina, off Hachijojima, Izu Islands, Japan. Mar. Biol. 115(4): 613–623
Onizuka, E. W. (1972). Management and development investigations of the Kona crab, Ranina ranina (Linnaeus). Final report. Department of Island and Natural Resources, Division of Fish and Game, State of Hawaii, Honolulu, Hawaii
Pillay, K. K., Nair, N. B. (1971). The annual reproductive cycles of Uca annulipes, Portunus pelagicus and Metapenaeus affinis (Decapoda: Crustacea) from the southwest coast of India. Mar. Biol. 11: 152–166
Pochon-Masson, J. (1983). Arthropoda-Crustacea. In: Adiyodi, K. G., Adiyodi, R. G. (eds.). Reproductive biology of invertebrates, Vol. 2. Spermatogenesis and sperm function. John Wiley & Sons, New York, p. 407–449
Radha, T., Subramoniam, T. (1985). Origin and nature of spermatophoric mass of the spiny lobster Panulirus homarus. Mar. Biol. 86: 13–19
Ryan, E. P. (1967). Structure and function of the reproductive system of the crab Portunus sanguinolentus (Herbst) (Brachyura: Portunidae) I. The male system. Proc. Symp. Crustacea, Ernakulami Coct. in (Part II): 506–521 (Mar. biol. Ass. India)
Sakai, T. (1976). Crabs of Japan and the adjacent seas. Kodansha, Tokyo, p. 49
Sasikala, S. L., Subramoniam, T. (1987). On the occurrence of acid mucopolysaccharides in the spermatophores of two marine prawns, Penaeus indicus (Milne-Edwards) and Metapenaeus monoceros (Fabricius) (Crustacea: Macrura). J. exp. mar. Biol. Ecol. 113: 145–153
Skinner, D. G., Hill, B. J. (1987). Feeding and reproductive behaviour and their effect on catchability of the spanner crab Ranina ranina. Mar. Biol. 94: 211–218
Spalding, J. F. (1942). The nature and formation of the spermatophore and sperm plug in Carcinus maenas. Quart. Jl. micross. Sci. 83(3/4): 399–422
Subramoniam, T. (1984). Spermatophore formation in two intertidal crabs Albunea symnista and Emertia asiatica (Decapoda: Anomura). Biol. Bull. mar. biol. Lab., Woods Hole 166: 78–95
Subramoniam, T. (1991). Chemical composition of spermatophores in decapod crustaceans. In: Bauer, R. T., Martin, J. W. (eds.). Crustacean sexual biology. Columbia University Press, New York, p. 308–321
Tahil, A. S. (1983). Reproductive period and exploitation of the red frog crab, Ranina ranina (Linnaeus 1758) in Central Tawi-Tawi, Philippines. Philipp. Scient. San Carlos Univ. Cebu City 20: 57–72
Thampy, D. M., John, P. A. (1970). On the androgenic gland of the ghost crab Ocypoda platytarsis M. Edwards (Crustacea: Brachyura). Acta zool., Stockh. 51: 203–210
Uma, K., Subramoniam, T. (1979). Histochemical characteristics of spermatophore layers of Scylla serrata (Forskal) (Decapoda: Portunidae). Int. J. Invert. Reprod. 1: 31–40
Yasuzumi, M. D. (1960). Spermatogenesis in animals as revealed by electron microscopy VII. Spermatid differentiation in the crab Eriocheir japonicus. J. biophysic. biochem. Cytol. 7(1): 73–78
Author information
Authors and Affiliations
Additional information
Communicated by T. Ikeda
Rights and permissions
About this article
Cite this article
Minagawa, M., Chiu, J.R., Kudo, M. et al. Male reproductive biology of the red frog crab, Ranina ranina, off Hachijojima, Izu Islands, Japan. Marine Biology 118, 393–401 (1994). https://doi.org/10.1007/BF00350296
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00350296