Abstract
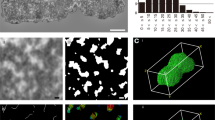
A study of ultrathin sections of normal Chinese hamster cells and cells treated with decreasing concentrations of bivalent cations (Ca2+ and Mg2+) in situ revealed several discrete levels of compaction of DNA-nucleoprotein (DNP) fibrils in mitotic chromosomes and the chromatin of interphase nuclei. At concentrations ranging from 3 mM CaCl2 and 1 mM MgCl2 to ten times less, the chromosomes are found to contain fibrous elements (chromonemata) about 100 nm in diameter. As Ca2+ concentration is gradually decreased to 0.2–0.1 mM, the chromosomes decondense into a number of discrete chromatin structures, the chromomeres. As decondensation proceeds, these chromomeres acquire a rosettelike structure with DNP fibrils radiating from an electron-dense core. Upon complete decondensation of chromosomes, individual chromomeres persist only in the centromeric regions. The following levels of DNP compaction in mitotic chromosomes are suggested: a 10-nm nucleosomal fibril, a 25-nm nucleomeric fibril, and the chromonema, a fibrous structure, about 100 nm in diameter, composed of chromomeres. Interphase nuclei also contain structures which are morphologically similar to the chromomeres of mitotic chromosomes.
Similar content being viewed by others
References
Bak P, Bak A, Zeuthen J (1979) Characterization of human chromosomal unit fibers. Chromosoma 73:301–315
Comings DE (1978) Mechanisms of chromosome banding and implications for chromosoma structure. Ann Rev Genet 12:25–46
Comings DE, Okada TA (1976) Nuclear proteins. III. The fibrillar nature of nuclear matrix. Exp Cell Res 103:341–360
Du Praw EJ (1965) Macromolecular organization of nuclei and chromosomes: a folded fibre model based on wholemount electron microscopy. Nature 206:338–343
Finch JT, Klug A (1976) Solenoidal model for superstructure in chromatin. Proc Natl Acad Sci USA 73:1897–1901
Hozier J, Renz M, Nehls P (1977) The chromosome fiber: evidence for an ordered superstructure of nucleosomes. Chromosoma 62:301–317
Igó-Kemenes T, Zachau HG (1978) Domains in chromatin structure. Cold Spring Harbor Sympos Quant Biol 42:109–118
Kiryanov GI, Manamsjan TA, Polyakov VYu, Chentsov YuS (1976) Levels of granular organization of chromatin fibers. FEBS Lett 67:323–327
Kiryanov GI, Smirnova TA, Polyakov VYu (1982) Nucleomeric organization of chromatin. Eur J Biochem 124:331–338
Lima-de-Faria A (1975) The relation between chromomeres, replicons, operons, transcription units, genes, viruses and palindromes. Hereditas 81:249–284
Mace ML, Daskal Ye, Busch H, Wray VP, Wray W (1978) Isolated metaphase chromosomes: scanning electron microscopic appearance of salt-extracted chromosomes. Cytobios 19:27–40
Marsden MPF, Laemmli UK (1979) Metaphase chromosome structure: evidence for a radial loop model. Cell 17:849–858
Okada TA, Comings DE (1979) Higher order structure of chromosomes. Chromosoma 72:1–14
Olins AL, Olins DE (1974) Spheroid chromatin units (v-bodies). Science 183:330–331
Oudet P, Gross-Bellard M, Chambon P (1975) Electron microscopic and biochemical evidence that chromatin structure is a repeating unit. Cell 4:281–300
Paulson JR, Laemmli UK (1977) The structure of histone-depleted metaphase chromosomes. Cell 12:817–828
Ris H, Korenberg Y (1979) Chromosome structure and levels of chromosome organization. Cell Biol 2:268–361
Serra JA (1947) Contribution to a physiological interpretation of mitosis and meiosis. II. The prophasic appearing of the spiralization. Portugaliae Acta Biol 2:45–90
Sparvoli E, Gay H, Kaufmann BP (1965) Number and pattern of association of chromonemata in the chromosomes of Tradescantia. Chromosoma 16:415–435
Wilson EB (1925) The cell. MacMillan. N.Y.
Wolfe SL, Martin PG (1968) The ultrastructure and strandedness of chromosomes from two species of Vicia. Exp Cell Res 50:140–150
Zatsepina OV, Polyakov VYu, Chentsov YuS (1982a) Nuclear envelope formation around metaphase chromosomes: Chromosome decondensation and nuclear envelope reconstitution during mitosis. Europ Cell Biol 26:277–283
Zatsepina OV, Polyakov VYu, Chentsov YuS (1982b) Higher levels of DNP organization. In: Ann second Soviet-Italian Symp. Macromolecules in the function cell, Moscow, p 103, Nauka
Zelenin MG, Zakharov AF, Zatsepina OV, Polyakov VYu, Chentsov YuS (1982) Reversible differential decondensation of unfixed Chinese hamster chromosomes induced by change in calciumion concentration of the medium. Chromosoma 84:729–736
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zatsepina, O.V., Polyakov, V.Y. & Chentsov, Y.S. Chromonema and chromomere. Chromosoma 88, 91–97 (1983). https://doi.org/10.1007/BF00327327
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00327327