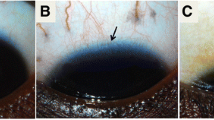
Abstract
Minocycline has a unique solubility in lipids and may reach therapeutic concentrations in tears and saliva. In two consecutive prospective double-masked clinical trials that were carried out in two villages in Saudi Arabia, we assessed the effects of oral minocycline in the treatment of trachoma and compared its effects with those of topical tetracycline ointment in the first study and to tetracycline ointment and placebo in the second study. A total of 178 eyes in 96 patients were included. The age range was 7 to 14 years, with a mean age of 9 years. All patients underwent complete ophthalmologic evaluation. School children were divided into two groups in a double-masked fashion. The first group received either oral minocycline or topical tetracycline 1% ointment and the second group was divided into three subgroups, each receiving one of the following therapeutic modalities: oral minocycline, topical tetracycline ointment, or placebo ointment. All patients were evaluated before initiation of therapy, at three weeks and at 12 months following treatment. Therapy was continued for a period of five weeks. These two double-masked field-based clinical trials have shown both minocycline given orally and tetracycline ointment given topically were effective in decreasing the intensity of inflammation due to trachoma. Oral minocycline was found to be equally effective as topical tetracycline ointment in the treatment of trachoma at three weeks. Minocycline, however, was found to be superior to topical tetracycline when patients were evaluated one year after therapy (p<0.05). These clinical trials have shown that long acting antibiotic agents such as minocycline is an effective therapeutic modality in the treatment of trachoma. Once a day minocycline administration assures compliance and helps in the eradication of the disease.
Similar content being viewed by others
References
Bowie WR, Lee CK, Alexander ER. Production of efficacy of antimicrobial agents in treatment of infections due to Chlamydia trachomatis. J Inf Dis 1978; 138: 655–59.
Dawson CF, Daghfous T, Messadi M et al. Severe endemic trachoma in Tunisia II. A controlled therapy trial of topically applied chlortetracycline and erythromycin. Archiv Ophthalmol 1974; 92: 198–203.
Dawson CR, Jones BR, Tarizzo ML. Guide to trachoma control in programs for the prevention of blindness. World Health Organization, Geneva, 1981.
Hoeprich PD, Warshauer DM. Entry of four tetracyclines into saliva and tears. Antimicrob Agents Chemother 1974; 5: 330–36.
Jones BR. The prevention of blindness from trachoma. Trans Ophthalmol Soc UK 1975; 95: 16–33.
Kupfer C. Worldwide prevention of blindness. Am J Ophthalmol 1983; 96: 543–45.
Schachter J, Dawson CR. Is trachoma an ocular component of a more generalized chlamydia infection? Lancet 1979: 702-03.
Tabbara KF, Ross-Degnan D. Blindness in Saudi Arabia. JAMA 1986; 255: 3378–84.
Tabbara KF. Tetracyclines. In: Smolin G, Okomoto M, eds. Antimicrobial agents in ophthalmology. New York: Masson Publishing Co, 1983: 65–68.
Tabbara KF, Ghosheh R, O'Connor GR. Ocular tissue absorption of minocycline in the rabbit. Archiv Ophthalmol 1983; 101: 1426–28.
Tarizzo ML. Chemotherapy of trachoma. WHO Chronicle 1972; 26: 99–101.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tabbara, K.F., Summanen, P., Taylor, P.B. et al. Minocycline effects in patients with active trachoma. Int Ophthalmol 12, 59–63 (1988). https://doi.org/10.1007/BF00133783
Issue Date:
DOI: https://doi.org/10.1007/BF00133783