Abstract
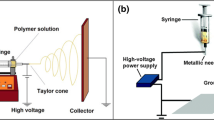
We report the fabrication and characterization of electrospun nanofibres from Gonometa postica and Gonometa rufobrunnae silk fibroin indigenous to Southern Africa. Nanofibres were electrospun from regenerated silk fibroin (RSF) powders obtained by freeze drying or via rotary evaporation using methanol as a desiccant. Optimal electrospinning conditions employed trifluoroacetic acid as a solvent. The solution of RSF powder prepared by rotary evaporation was electrospun at a lower concentration (27% w/v) than that of freeze dried RSF powder (40% w/v). Concentration of the fibroin solution had the most influence on electrospinnability whereas voltage and flow rate mainly affected the fibre morphology. Scanning electron microscopy (SEM) showed that nanofibres from freeze dried and rotary evaporator dried RSF powders had diameters ranging from 300–760 and 400–1,000 nm respectively. Fourier transform infrared (FTIR) analysis revealed that the RSF powders were mainly composed of β-sheets, similar to degummed silk fibroin. The nanofibres, however, exhibited predominantly random coil/α-helical structure showing degradation of the native silk structure. β-sheet structure in the nanofibres was restored upon solvent treatment resulting in improved water stability. The extent of structural transformation was dependent on the type of solvent used. This study confirms the feasibility of fabricating Gonometa fibroin into stable nanofibrous structures that could be used as scaffolds in biotechnological and biomedical applications.

Similar content being viewed by others
References
X. Zhang, Adv. Drug Deliver. Rev., 61, 988 (2009).
W. Friess, Eur. J. Pharm. Biopharm., 45, 113 (1998).
G. H. Altman, F. Diaz, C. Jakuba, T. Calabro, R. L. Horan, J. Chen, H. Lu, J. Richmond, and D. L. Kaplan, Biomaterials, 24, 401 (2003).
C. Vepari and D. L. Kaplan, Prog. Polym. Sci., 32, 991 (2007).
Z. M. Huang, Y. Z. Zhang, M. Kotaki, and S. Ramakrishna, Compos. Sci. Technol., 63, 2223 (2003).
J. D. Schiffman and C. L. Schauer, Polym. Rev., 48, 317 (2008).
J. K. Jayaraman, M. Kotaki, Y. Zhang, X. Mo, and S. Ramakrishna, J. Nanosci. Nanotechnol., 4, 52 (2004).
Z. Ma, M. Kotaki, R. Inai, and S. Ramakrishna, Tissue Eng., 11, 101 (2005).
Y. Tamada, Biomacromolecules, 6, 3100 (2005).
E. Wenk, A. J. Meinel, S. Wildy, H. P. Merkle, and L. Meinel, Biomaterials, 30, 2571 (2009).
B. M. Min, G. Lee, S. H. Kim, Y. S. Nam, T. S. Lee, and W. H. Park, Biomaterials, 25, 1289 (2004).
L. Soffer, X. Wang, X. Zhang, J. Kluge, L. Dorfmann, D. L. Kaplan, and G. Leisk, J. Biomater. Sci. Polym. Ed., 19, 653 (2008).
K. H. Kim, L. Jeong, H. N. Park, S. Y. Shin, W. H. Park, S. C. Lee, T. I. Kim, Y. J. Park, Y. J. Seol, Y. M. Lee, Y. Ku, I. C. Rhyu, S. B. Han, and C. P. Chung, J. Biotechnol., 120, 327 (2005).
Y. Srisuwan and P. Srihanam, J. Appl. Sci., 9, 978 (2009).
K. Ohgo, C. Zhao, M. Kobayashi, and T. Asakura, Polymer, 44, 841 (2003).
F. Zhang, B. Q. Zuo, H. X. Zhang, and L. Bai, Polymer, 50, 279 (2009).
C. Meechaisue, P. Wutticharoenmongkol, R. Waraput, T. Huangjing, N. Ketbumrung, P. Pavasant, and P. Supaphol, Biomed. Mater., 2, 181 (2007).
V. Mhuka, S. Dube, and M. M. Nindi, Int. J. Biol. Macromol., DOI: 10.1016/j.ijbiomac.2012.09.010 (2012).
G. Freddi, A. B. Svilokos, H. Ishikawa, and M. Tsukada, J. Appl. Polym. Sci., 48, 99 (1993).
A. De’r, L. Kelemen, L. Fábián, S. G. Taneva, E. Fodor, T. Páli, A. Cupane, M. G. Cacace, and J. J. Ramsden, J. Phys. Chem. B, 111, 5344 (2007).
K. D. Collins, Methods, 34, 300 (2004).
W. N. L. Zhang, Y. Wenhua, and X. Shiying, J. Chin. Inst. Food Sci. Technol., 1, 56 (2001).
Y. Miyaguchi and J. Hu, Food Sci. Technol. Res., 11, 3742 (2005).
A. S. Bommarius and B. R. Riebel, in Biocatalysis: Fundamentals and Applications, Wiley, New York, 2004, p 228.
D. H. Reneker and I Chun, Nanotechnology, 7, 36 (1996).
J. M. Deitzel, J. Kleinmeyer, D. Harris, and N. C. Beck Tan, Polymer, 42, 261 (2001).
B. Dhandayuthapani, Y. Yoshida, T. Maekawa, and D. S. Kumar, Mater. Res., 14, 317 (2011).
Z W. Zhou, J. He, S. Cui, and W. Gao, Open Mater. Sci. J., 5, 51 (2011).
S. H. Tan, R. Inai, M. Kotaki, and S. Ramakrishna, Polymer, 46, 6128 (2005).
X. H. Zong, K. Kim, B. S. Hsiao, and S. F. Ran, Polymer, 43, 4403 (2002).
E. R. Kenawy, J. M. Layman, J. R. Watkins, G. L. Bowlin, J. A Matthews, and D. G. Simpson, Biomaterials, 24, 907 (2003).
M. Tsukada, G. Freddi, Y. Gotoh, and N. Kasai, J. Polym. Sci. Part B: Polym. Phys., 32, 1407 (1994).
A. L. Andrady, in Science and Technology of Polymer Nanofibers, John Wiley & Sons, Hoboken, 2007, p 105.
B. Marelli, A. Alessandrino, S. Farè, G. Freddi, D. Mantovani, and M. C. Tanzi, Acta Biomater., 6, 4019 (2010).
M. Min, L. Jeong, K.Y. Lee, and W. H. Park, Macromol. Biosci., 6, 285 (2006).
H. Kweon and Y. H. Park, J. Appl. Polym. Sci., 82, 750 (2001).
P. Wadbua, B. Promdonkoy, S. Maensiri, and S. Siri, Int. J. Biol. Macromol., 46, 493 (2010).
W. Wei, Y. Zhang, H. Shao, and X. Hu, J. Mater. Res., 26, 1100 (2011).
W. Tao, M. Li, and C. Zhao, Int. J. Biol. Macromol., 10, 472 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mhuka, V., Dube, S., Nindi, M.M. et al. Fabrication and structural characterization of electrospun nanofibres from Gonometa Postica and Gonometa Rufobrunnae regenerated silk fibroin. Macromol. Res. 21, 995–1003 (2013). https://doi.org/10.1007/s13233-013-1127-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-013-1127-1