Abstract
Background
The purpose of the study is to review the CT findings associated with ventriculostomy placement in regards to the safety of an EVD plus recombinant tissue plasminogen activator (rt-PA) for IVH.
Methods
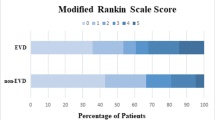
A retrospective review was conducted for patients receiving intraventricular rt-PA for IVH from January 2004 to September 2009. Safety was assessed by the presence of EVD tract hemorrhage by CT at baseline after EVD placement, worsening hemorrhage after rt-PA, and CSF infection. IVH volumetrics were assessed by the Le Roux score and outcomes by Glasgow Outcome Scale and modified Rankin Scale.
Results
Twenty-seven patients received rt-PA for IVH. Median dose was 2 mg (range 0.3–8) and a median of two doses (range 1–17) were given. Worsening EVD catheter tract hemorrhage after rt-PA was 46.7 %, with a significantly higher incidence of worsening tract hemorrhage seen with incorrectly placed EVDs (p = 0.04). IVH hematoma burden decreased by a median Le Roux score of 10 (range 3–16) prior to rt-PA to 4 (range 0–16) after rt-PA. There were no central nervous system bacterial infections.
Conclusion
Intraventricular rt-PA appears to be relatively safe especially when all EVD fenestrations are within the ventricle and reduces IVH burden similar to other studies. We describe a CT-based EVD tract hemorrhage grading scale to evaluate EVD tract hemorrhage before and after thrombolysis, and a bone-window technique to evaluate EVD fenestrations prior to IVH thrombolysis. Further research is needed evaluating these imaging techniques in regard to intraventricular thrombolytic safety and EVD tract hemorrhage.



Similar content being viewed by others
References
Little JR, Blomquist GA Jr, Ethier R. Intraventricular hemorrhage in adults. Surg Neurol. 1977;8:143–9.
Coplin WM, Vinas FC, Agris JM, et al. A cohort study of the safety and feasibility of intraventricular urokinase for nonaneurysmal spontaneous intraventricular hemorrhage. Stroke. 1998;29:1573–9.
Steiner T, Diringer MN, Schneider D, et al. Dynamics of intraventricular hemorrhage in patients with spontaneous intracerebral hemorrhage: risk factors, clinical impact, and effect of hemostatic therapy with recombinant activated factor VII. Neurosurgery. 2006;59(4):767–73. Discussion 773–764.
Tuhrim S, Horowitz DR, Sacher M, Godbold JH. Volume of ventricular blood is an important determinate of outcome in supratentorial intracerebral hemorrhage. Crit Care Med. 1999;27:617–21.
Jayakumar PN, Taly AB, Rao BV, Arya BT, Nagaraja D. Prognosis in solitary intraventricular hemorrhage: clinical and computed tomographic observations. Acta Neurol Stand. 1989;80:l–5.
Vereecken KK, Van Havenbergh T, De Beuckelaar W, Parizel PM, Jorens PG. Treatment of intraventricular hemorrhage with intraventricular administration of recombinant tissue plasminogen activator A clinical study of 18 cases. Clin Neurol Neurosurg. 2006;108:451–5.
Steinke W, Sacco RL, Mohr JP, et al. Thalamic stroke. Presentation and prognosis of infarcts and hemorrhages. Arch Neurol. 1992;49:703–10.
Lee KR, Betz AL, Kim S, Keep RF, Hoff JT. The role of the coagulation cascade in brain edema formation after intracerebral hemorrhage. Acta Neurochir (Wien). 1996;138:396–400; discussion 400–401.
Diringer MN, Edwards DF, Zazulia AR. Hydrocephalus: a previously unrecognized predictor of poor outcome from supratentorial intracerebral hemorrhage. Stroke. 1998;29:1352–7.
Ehtisham A, Taylor S, Bayless L, Klein MW, Janzen JM. Placement of external ventricular drains and intracranial pressure monitors by neurointensivists. Neurocrit Care. 2009;10:241–7.
Guyot LL, Dowling C, Diaz FG, Michael DB. Cerebral monitoring devices: analysis of complications. Acta Neurochir Suppl. 1998;71:47–9.
Wiesmann M, Mayer TE. Intracranial bleeding rates associated with two methods of external ventricular drainage. J Clin Neurosci. 2001;8:126–8.
Hanley DF, Naff NJ, Harris DM. Intraventricular hemorrhage: presentation and management options. Semin Cerebrovasc Dis Stroke. 2005;5:209–16.
Hanley DF. Clot Lysis: Evaluating Accelerated Resolution of Intraventricular Hemorrhage Phase III (Clear III Trial) Clinical Trials.gov. NCT00784134.
Naff N, Williams MA, Keyl PM, et al. Low-dose recombinant tissue-type plasminogen activator enhances clot resolution in brain hemorrhage: the intraventricular hemorrhage thrombolysis trial. Stroke. 2011;42(11):3009–16.
Naff NJ, Hanley DF, Keyl PM, et al. Intraventricular thrombolysis speeds blood clot resolution: results of a pilot, prospective, randomized, double-blind, controlled trial. Neurosurgery. 2004;54:577–83; discussion 583–584.
LeRoux PD, Haglund MM, Newell DW, Grady MS, Winn HR. Intraventricular hemorrhage in blunt head trauma: an analysis of 43 cases. Neurosurgery. 1992;31:678–84; discussion 684–685.
Rankin J. Cerebral vascular accidents in patients over the age of 60. II. Prognosis. Scott Med J. 1957;2:200–15.
van Swieten JC, Koudstaal PJ, Visser MC, Schouten HJ, van Gijn J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke. 1988;19:604–7.
Jennett B, Bond M. Assessment of outcome after severe brain damage. Lancet. 1975;1:480–4.
Alteplase (rt-PA) [Activase®]. Package Insert. San Francisco, CA: Genetech Inc.; 2011.
Acknowledgments
The authors would like to acknowledge the contributions of the following colleagues: Paula Fuqua, PharmD, CCRC; Jamila Russeau PharmD, BCPS; Leah Ward PharmD; and Amy Swan M.S., PharmD, for guidance and assistance in study protocol development and manuscript review. Michael Heckman, MS for general advice on using appropriate statistics for this study. Tara Brigham, MLIS and Victoria Jackson, MLIS for reviewing and formatting the final manuscript.
Disclosure
Dr. Jackson reports no disclosures. Dr. Patel reports no disclosures. Dr. Darracott reports no disclosures. Dr. Hanel serves as a scientific advisor for NeuroVasx, Inc. although it is unrelated to this study. Dr. Freeman receives research support for his time involved as a site investigator in the CLEAR IVH III trial, NIH grant # U01 NS062851, which is a NINDS-Genentech funded study with Dr. Daniel F. Hanley as the principle investigator. Dr. Hanley receives research support for his time involved as principle investigator in the following clinical trials: CLEAR IVH III (NIH grant # U01 NS062851) and MISTIE ICH (NIH grant # U01 PAR 10198), which are NIH-NINDS funded studies; and as Jeffrey & Harriet Legum Professor at Johns Hopkins University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jackson, D.A., Patel, A.V., Darracott, R.M. et al. Safety of Intraventricular Hemorrhage (IVH) Thrombolysis Based on CT Localization of External Ventricular Drain (EVD) Fenestrations and Analysis of EVD Tract Hemorrhage. Neurocrit Care 19, 103–110 (2013). https://doi.org/10.1007/s12028-012-9713-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-012-9713-1