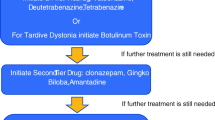
Opinion statement
Tardive dyskinesia (TD) is iatrogenic (drug-induced); hence the best strategy is prevention. Try to limit exposure to any dopamine receptor blocking agents (DRBAs) if possible. These agents may be unavoidable in some psychiatric conditions such as schizophrenia, but alternative therapies can be used in many situations, such as in the treatment of depression, anxiety, gastrointestinal conditions, and other neurologic conditions, including migraines and sleep disorders. When DRBAs are necessary, physicians should prescribe the smallest possible dose and try to taper and stop the drug at the earliest signs of TD. Abrupt cessation should be avoided, as this can worsen symptoms of TD. Always discuss and document the possibility of TD as an adverse effect when starting patients on DRBAs. If TD is mild and tolerable, the withdrawal of the offending agent is possible, and exposure to DRBAs was short, physicians should consider avoiding treatment and waiting for spontaneous recovery. When treatment is necessary, tetrabenazine (TBZ) is considered a potential first-line agent and is known to be one of the most effective drugs in treating TD, but it is expensive and adverse effects such as depression, akathisia and parkinsonism frequently occur. Therefore, second-line agents with better tolerability profiles are often tried first in practice. These include amantadine, benzodiazepines, beta-blockers, and levetiracetam. When using TBZ, adverse effects should be aggressively monitored. (Depression often can be managed with antidepressants, for instance). In patients with psychosis, withdrawal of the antipsychotic may not be possible. Switching to clozapine or quetiapine is one option to minimize TD. When these agents are contraindicated and the patient must continue using other atypical antipsychotic drugs, try to add dopamine-depleting agents such as TBZ or reserpine, but watch for the development of parkinsonism. When the symptoms are focal, such as tongue protrusion or blepharospasm, botulinum toxin injections can be very effective if spontaneous recovery does not occur. As a last resort, when disabling, life-threatening symptoms of TD persist despite all of the above-mentioned methods, some advocate resuming treatment with the DRBA to suppress symptoms of TD. This has the potential to worsen TD in the long run.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance
Kruse W. Persistent muscular restlessness after phenothiazine treatment: report of 3 cases. Am J Psychiatry. 1960;117:152–3.
Correll CU, Schenk EM. Tardive dyskinesia and new antipsychotics. Curr Opin Psychiatry. 2008;21(2):151–6.
Rapaport A, et al. Botulinum toxin for the treatment of oro-facial-lingual-masticatory tardive dyskinesia. Mov Disord. 2000;15(2):352–5.
Bateman DN, Rawlins MD, Simpson JM. Extrapyramidal reactions with metoclopramide. Br Med J (Clin Res Ed). 1985;291(6500):930–2.
Wiholm BE, et al. Tardive dyskinesia associated with metoclopramide. Br Med J (Clin Res Ed). 1984;288(6416):545–7.
Jeste DV, Wyatt RJ. Therapeutic strategies against tardive dyskinesia. Two decades of experience. Arch Gen Psychiatry. 1982;39(7):803–16.
Kane JM, Woerner M, Lieberman J. Tardive dyskinesia: prevalence, incidence, and risk factors. J Clin Psychopharmacol. 1988;8(4 Suppl):52S–6S.
Marsden CD, MRHS, Baldessarini RJ. Extrapyramidal movement disorders produced by antipsychotic drugs. In: Hirsch SR, Bradley PB, editors. Pharmacology and Treatment of Schizophrenia. London: Oxford; 1986.
Woerner MG, et al. Diabetes and development of tardive dyskinesia. Am J Psychiatry. 1993;150(6):966–8.
Yassa R, Jeste DV. Gender differences in tardive dyskinesia: a critical review of the literature. Schizophr Bull. 1992;18(4):701–15.
FitzGerald PM, Jankovic J. Tardive oculogyric crises. Neurology. 1989;39(11):1434–7.
Fahn S, Jankovic J. Principles and practice of movement disorders. Philadelphia: Churchill Livingstone; 2007.
Lang AE. Clinical differences between metoclopramide- and antipsychotic-induced tardive dyskinesias. Can J Neurol Sci. 1990;17(2):137–9.
Samie MR, Dannenhoffer MA, Rozek S. Life-threatening tardive dyskinesia caused by metoclopramide. Mov Disord. 1987;2(2):125–9.
Horiguchi J, et al. Antipsychotic-induced life-threatening ‘esophageal dyskinesia’. Int Clin Psychopharmacol. 1999;14(2):123–7.
Labbate LA, et al. Tardive dyskinesia in older out-patients: a follow-up study. Acta Psychiatr Scand. 1997;96(3):195–8.
• Fasano A, Bentivoglio AR. Tetrabenazine. Expert Opin Pharmacother. 2009;10(17):2883–96.
Huntington Study Group. Tetrabenazine as antichorea therapy in Huntington disease: a randomized controlled trial. Neurology. 2006;66(3):366–72.
Ondo WG, Hanna PA, Jankovic J. Tetrabenazine treatment for tardive dyskinesia: assessment by randomized videotape protocol. Am J Psychiatry. 1999;156(8):1279–81.
Kenney C, Hunter C, Jankovic J. Long-term tolerability of tetrabenazine in the treatment of hyperkinetic movement disorders. Mov Disord. 2007;22(2):193–7.
Huang CC, et al. Evaluation of reserpine and alpha-methyldopa in the treatment of tardive dyskinesia. Psychopharmacol Bull. 1980;16(3):41–3.
Huang CC, et al. Reserpine and alpha-methyldopa in the treatment of tardive dyskinesia. Psychopharmacol Berl. 1981;73(4):359–62.
Fahn S. A therapeutic approach to tardive dyskinesia. J Clin Psychiatry. 1985;46(4 Pt 2):19–24.
Lieberman JA, et al. The effects of clozapine on tardive dyskinesia. Br J Psychiatry. 1991;158:503–10.
Bacher NA, Lewis HA. Propranolol, anxiety, and tardive dyskinesia. J Clin Psychiatry. 1983;44(5):196.
Perenyi A, Farkas A. Propranolol in the treatment of tardive-dyskinesia. Biol Psychiatry. 1983;18(3):391–4.
Pitts FN. Treatment of tardive-dyskinesia with propranolol. J Clin Psychiatry. 1982;43(8):304–4.
Schrodt GR, et al. Treatment of tardive-dyskinesia with propranolol. J Clin Psychiatry. 1982;43(8):328–31.
Soares KV, McGrath JJ. Vitamin E for neuroleptic-induced tardive dyskinesia. Cochrane Database Syst Rev. 2000;(2):CD000209.
Adler LA, et al. Vitamin E treatment of tardive dyskinesia. Am J Psychiatry. 1993;150(9):1405–7.
Adler LA, et al. Vitamin E treatment for tardive dyskinesia. Veterans Affairs Cooperative Study #394 Study Group. Arch Gen Psychiatry. 1999;56(9):836–41.
Thaker GK, et al. Clonazepam treatment of tardive dyskinesia: a practical GABAmimetic strategy. Am J Psychiatry. 1990;147(4):445–51.
Bobruff A, et al. Clonazepam and phenobarbital in tardive dyskinesia. Am J Psychiatry. 1981;138(2):189–93.
• Pappa S, et al. Effects of amantadine on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. Clin Neuropharmacol. 2010;33(6):271–5.
Angus S, et al. A controlled trial of amantadine hydrochloride and neuroleptics in the treatment of tardive dyskinesia. J Clin Psychopharmacol. 1997;17(2):88–91.
Woods SW, et al. Effects of levetiracetam on tardive dyskinesia: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2008;69(4):546–54.
Slotema CW, et al. Botulinum toxin in the treatment of orofacial tardive dyskinesia: a single blind study. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(2):507–9.
Esper CD, Freeman A, Factor SA. Lingual protrusion dystonia: frequency, etiology and botulinum toxin therapy. Parkinsonism Relat Disord. 2010;16(7):438–41.
Hennings JM, Bötzel KE, Wetter TC. Successful treatment of tardive lingual dystonia with botulinum toxin: case report and review of the literature. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32(5):1167–71.
Welter ML, Grabli D, Vidailhet M. Deep brain stimulation for hyperkinetics disorders: dystonia, tardive dyskinesia, and tics. Curr Opin Neurol. 2010;23(4):420–5.
Disclosure
Conflicts of Interest: P. Aia: none; G. Revuelta: none; L. Cloud: Participant in scientific advisory board for UCB Pharma; S. Factor: Expert testimony on behalf of Boehringer Ingelheim.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aia, P.G., Revuelta, G.J., Cloud, L.J. et al. Tardive Dyskinesia. Curr Treat Options Neurol 13, 231–241 (2011). https://doi.org/10.1007/s11940-011-0117-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11940-011-0117-x