Abstract
Purpose
Lymphedema following breast cancer surgery remains a common and feared treatment complication. Accurate information on health-related quality of life (HRQOL) outcomes among patients with lymphedema is critically needed to inform shared medical decision making and evidence-based practice in oncologic breast surgery. Our systematic review aimed to (1) identify studies describing HRQOL outcomes in breast cancer-related lymphedema (BCRL) patients, (2) assess the quality of these studies, and (3) assess the quality and appropriateness of the patient-reported outcome (PRO) instruments used.
Methods
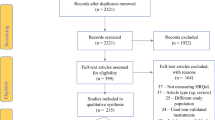
Using the PRISMA statement, we performed a systematic review including studies describing HRQOL outcomes among BCRL patients. Studies were classified by levels of evidence and fulfillment of the Efficace criteria. PRO instruments were assessed using the COSMIN criteria.
Results
Thirty-nine studies met inclusion criteria, including 8 level I and 14 level II studies. Sixteen of 39 studies were compliant with the Efficace criteria. Seventeen HRQOL instruments were used, two specific to lymphedema patients. Exercise and complex decongestive therapy treatment interventions were associated with improved HRQOL.
Conclusions
High-quality data on HRQOL outcomes is required to inform surgical decisions for breast cancer management and survivors. Of the lymphedema-specific PRO instruments, the Upper Limb Lymphedema 27 (ULL-27) was found to have strong psychometric properties. Future studies should strive to use high-quality condition- specific PRO instruments, follow existing guidelines for HRQOL measurement and to consider economic burdens of BCRL.
Implications for Cancer Survivors
As lymphedema may develop many years after breast cancer surgery, the ULL-27 may offer greater content validity for use in survivorship research.

Similar content being viewed by others
References
Cancer Facts & Figures 2011. American Cancer Society. Atlanta, 2011.
Ahmed RL, Prizment A, Lazovich D, Schmitz KH, Folsom AR. Lymphedema and quality of life in breast cancer survivors: the Iowa Women’s Health Study. J Clin Oncol. 2008;26(35):5689–96.
Rockson SG. Lymphedema. Am J Med. 2001;110(4):288–95.
Hormes JM, Bryan C, Lytle LA, et al. Impact of lymphedema and arm symptoms on quality of life in breast cancer survivors. Lymphology. 2010;43(1):1–13.
Winters ZE, Benson JR, Pusic AL. A systematic review of the clinical evidence to guide treatment recommendations in breast reconstruction based on patient- reported outcome measures and health-related quality of life. Ann Surg. 2010;252(6):929–42.
Pusic A, Liu JC, Chen CM, et al. A systematic review of patient-reported outcome measures in head and neck cancer surgery. Otolaryngol Head Neck Surg. 2007;136(4):525–35.
Efficace F, Bottomley A, Osoba D, et al. Beyond the development of health-related quality-of-life (HRQOL) measures: a checklist for evaluating HRQOL outcomes in cancer clinical trials—does HRQOL evaluation in prostate cancer research inform clinical decision making? J Clin Oncol. 2003;21(18):3502–11.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–e34.
Making health care safer: a critical analysis of patient safety practices. Evidence-based review methodology (Chap. 3). http://www.ahrq.gov/clinic/ptsafety/chap3.htm. Accessed 23 Oct 2012.
Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN checklist for assessing the methodological quality of studies on measurement properties of health status measurement instruments: an international Delphi study. Qual Life Res. 2010;19(4):539–49.
Speck RM, Gross CR, Hormes JM, et al. Changes in the Body Image and Relationship Scale following a one-year strength training trial for breast cancer survivors with or at risk for lymphedema. Breast Cancer Res Treat. 2010;2:421–30.
Kim do S, Sim YJ, Jeong HJ, Kim GC. Effect of active resistive exercise on breast cancer-related lymphedema: a randomized controlled trial. Arch Phys Med Rehabil. 2010;91(12):1844–8.
Tidhar D, Katz-Leurer M. Aqua lymphatic therapy in women who suffer from breast cancer treatment-related lymphedema: a randomized controlled study. Supportive Care in Cancer. 2010;18(3):383–92.
McClure MK, McClure RJ, Day R, Brufsky AM. Randomized controlled trial of the breast cancer recovery program for women with breast cancer-related lymphedema. Am J Occup Ther. 2010;64(1):59–72.
Tsai HJ, Hung HC, Yang JL, Huang CS, Tsauo JY. Could Kinesio tape replace the bandage in decongestive lymphatic therapy for breast-cancer-related lymphedema? A pilot study. Support Care Cancer. 2009;11:1353–60.
McKenzie DC, Kalda AL. Effect of upper extremity exercise on secondary lymphedema in breast cancer patients: a pilot study. J Clin Oncol. 2003;21(3):463–6.
Williams AF, Vadgama A, Franks PJ, Mortimer PS. A randomized controlled crossover study of manual lymphatic drainage therapy in women with breast cancer-related lymphoedema. Eur J Cancer Care (Engl). 2002;11(4):254–61.
Vassard D, Olsen MH, Zinckernagel L, Vibe-Petersen J, Dalton SO, Johansen C. Psychological consequences of lymphoedema associated with breast cancer: a prospective cohort study. Eur J Cancer. 2010;46(18):3211–8.
Sagen A, Karesen R, Sandvik L, Risberg M. Changes in arm morbidities and health-related quality of life after breast cancer surgery: a five-year follow-up study. J Clin Oncol. 2009;27(15):e20602.
Paskett ED, Naughton MJ, McCoy TP, Case LD, Abbott JM. The epidemiology of arm and hand swelling in premenopausal breast cancer survivors. Cancer Epidemiol Biomarkers Prev. 2007;16(4):775–82.
Kim SJ, Yi CH, Kwon OY. Effect of complex decongestive therapy on edema and the quality of life in breast cancer patients with unilateral leymphedema. Lymphology. 2007;40(3):143–51.
Cheema BS, Gaul CA. Full-body exercise training improves fitness and quality of life in survivors of breast cancer. J Strength Cond Res. 2006;20(1):14–21.
O’Neill J, Beatus J. The effects of complete decongestive physical therapy treatment on edema reduction, quality of life, and functional ability of persons with upper extremity lymphedema. J Women’s Health Phys Therapy. 2006;30(1):5–10.
Brorson H, Ohlin K, Olsson G, Langstrom G, Wiklund I, Svensson H. Quality of life following liposuction and conservative treatment of arm lymphedema. Lymphology. 2006;39(1):8–25.
Wilburn O, Wilburn P, Rockson SG. A pilot, prospective evaluation of a novel alternative for maintenance therapy of breast cancer-associated lymphedema. BMC Cancer. 2006;6:84.
Strauss-Blasche G, Gnad E, Ekmekcioglu C, Hladschik B, Marktl W. Combined inpatient rehabilitation and spa therapy for breast cancer patients: effects on quality of life and CA 15–3. Cancer Nurs. 2005;28(5):390–8.
Mondry TE, Riffenburgh RH, Johnstone PA. Prospective trial of complete decongestive therapy for upper extremity lymphedema after breast cancer therapy. Cancer J. 2004;10(1):42–8.
Turner J, Hayes S, Reul-Hirche H. Improving the physical status and quality of life of women treated for breast cancer: a pilot study of a structured exercise intervention. J Surg Oncol. 2004;86(3):141–6.
Coster S, Poole K, Fallowfield LJ. The validation of a quality of life scale to assess the impact of arm morbidity in breast cancer patients post-operatively. Breast Cancer Res Treat. 2001;68(3):273–82.
Velanovich V, Szymanski W. Quality of life of breast cancer patients with lymphedema. Am J Surg. 1999;177(3):184–7.
Mirolo BR, Bunce IH, Chapman M, et al. Psychosocial benefits of postmastectomy lymphedema therapy. Cancer Nurs. 1995;18(3):197–205.
Beaulac SM, McNair LA, Scott TE, LaMorte WW, Kavanah MT. Lymphedema and quality of life in survivors of early-stage breast cancer. Arch Surg. 2002;137(11):1253–7.
Nesvold IL, Foss SD, Holm I, Naume B, Dahl AA. Arm/shoulder problems in breast cancer survivors are associated with reduced health and poorer physical quality of life. Acta Oncol. 2010;49(3):347–53.
Nesvold IL, Reinertsen KV, Fossa SD, Dahl AA. The relation between arm/shoulder problems and quality of life in breast cancer survivors: a cross-sectional and longitudinal study. J Cancer Surviv. 2010;5(1):62–72.
Chachaj A, Malyszczak K, Pyszel K, et al. Physical and psychological impairments of women with upper limb lymphedema following breast cancer treatment. Psychooncology. 2010;19(3):299–305.
Mak SS, Mo KF, Suen JJ, Chan SL, Ma WL, Yeo W. Lymphedema and quality of life in Chinese women after treatment for breast cancer. Eur J Oncol Nurs. 2009;13(2):110–5.
Paim CR, de Paula Lima ED, Fu MR, de Paula Lima A, Cassali GD. Post lymphadenectomy complications and quality of life among breast cancer patients in Brazil. Cancer Nurs. 2008;31(4):302–9.
Pyszel A, Malyszczak K, Pyszel K, Andrzejak R, Szuba A. Disability, psychological distress and quality of life in breast cancer survivors with arm lymphedema. Lymphology. 2006;39(4):185–92.
Ridner SH, Dietrich MS, Kidd N. Breast cancer treatment-related lymphedema self-care: education, practices, symptoms, and quality of life. Support Care Cancer. 2011;19(5):631–7.
Karadibak D, Yavuzsen T, Saydam S. Prospective trial of intensive decongestive physiotherapy for upper extremity lymphedema. J Surg Oncol. 2008;97(7):572–7.
Dawes DJ, Meterissian S, Goldberg M, Mayo NE. Impact of lymphoedema on arm function and health-related quality of life in women following breast cancer surgery. J Rehabil Med. 2008;40(8):651–8.
Heiney SP, McWayne J, Cunningham JE, et al. Quality of life and lymphedema following breast cancer. Lymphology. 2007;40(4):177–84.
Ridner SH. Quality of life and a symptom cluster associated with breast cancer treatment-related lymphedema. Support Care Cancer. 2005;13(11):904–11.
Wilson RW, Hutson LM, Vanstry D. Comparison of 2 quality-of-life questionnaires in women treated for breast cancer: the RAND 36-Item Health Survey and the Functional Living Index-Cancer. Phys Ther. 2005;85(9):851–60.
Pain SJ, Vowler SL, Purushotham AD. Is physical function a more appropriate measure than volume excess in the assessment of breast cancer-related lymphoedema (BCRL)? Eur J Cancer. 2003;39(15):2168–72.
Kwan W, Jackson J, Weir LM, Dingee C, McGregor G, Olivotto IA. Chronic arm morbidity after curative breast cancer treatment: prevalence and impact on quality of life. J Clin Oncol. 2002;20(20):4242–8.
Kirshbaum M. The development, implementation and evaluation of guidelines for the management of breast cancer related lymphoedema. Eur J Cancer Care (Engl). 1996;5(4):246–51.
Launois R, Megnigbeto AC, Pocquet K, Alliot F. A specific quality of life scale in upper limb lymphoedema: the ULL-27 questionnaire. Lymphology. 2002;35(1–760):181–7.
Schipper H, Clinch J, McMurray A, Levitt M. Measuring the quality of life of cancer patients: the Functional Living Index-Cancer: development and validation. J Clin Oncol. 1984;2(5):472–83.
Cemal Y, Pusic A, Mehrara BJ. Preventative measures for lymphedema: separating fact from fiction. J Am Coll Surg. 2011;213(4):543–51.
Conflict of interest
The authors declare that they have no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pusic, A.L., Cemal, Y., Albornoz, C. et al. Quality of life among breast cancer patients with lymphedema: a systematic review of patient-reported outcome instruments and outcomes. J Cancer Surviv 7, 83–92 (2013). https://doi.org/10.1007/s11764-012-0247-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-012-0247-5