Abstract
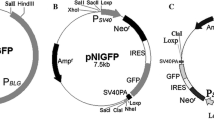
Transgenic cattle expressing high levels of recombinant human serum albumin (HSA) in their milk may as an alternative source for commercial production. Our objective was to produce transgenic cattle highly expressing HSA in milk by using phiC31 integrase system and somatic cell nuclear transfer (SCNT). The mammary-specific expression plasmid pIACH(−), containing the attB recognition site for phiC31 integrase, were co-transfected with integrase expression plasmid pCMVInt into bovine fetal fibroblast cells (BFFs). PhiC31 integrase-mediated integrations in genome of BFFs were screened by nested inverse PCR. After analysis of sequence of the PCR products, 46.0 % (23/50) of the both attB-genome junction sites (attL and attR) were confirmed, and four pseudo attP sites were identified. The integration rates in BF3, BF11, BF19 and BF4 sites were 4.0 % (2/50), 6.0 % (3/50), 16.0 % (8/50) and 20.0 % (10/50), respectively. BF3 is located in the bovine chromosome 3 collagen alpha-3 (VI) chain isomer 2 gene, while the other three sites are located in the non-coding region. The transgenic cell lines from BF11, BF19 and BF4 sites were used as donors for SCNT. Two calves from transgenic cells BF19 were born, one died within a few hours after birth, and another calf survived healthy. PCR and Southern blot analysis revealed integration of the transgene in the genome of cloned calves. The nested reverse PCR confirmed that the integration site in cloned calves was identical to the donor cells. The western blotting assessment indicated that recombinant HSA was expressed in the milk of transgenic cattle and the expression level was about 4–8 mg/mL. The present study demonstrated that phiC31 integrase system was an efficient and safety gene delivery tool for producing HSA transgenic cattle. The production of recombinant HSA in the milk of cattle may provide a large-scale and cost-effective resource.




Similar content being viewed by others
References
Aneja MK, Imker R, Rudolph C (2007) Phage phiC31 integrase-mediated genomic integration and long-term gene expression in the lung after nonviral gene delivery. J Gene Med 9:967–975
Bi Y, Liu X, Zhang L, Shao C, Ma Z, Hua Z, Zhang L, Li L, Hua W, Xiao H, Wei Q, Zheng X (2013) Pseudo attP sites in favor of transgene integration and expression in cultured porcine cells identified by Streptomyces phage phiC31 integrase. BMC Mol Biol 14:20. doi:10.1186/1471-2199-14-20
Chalberg TW, Genise HL, Vollrath D, Calos MP (2005) phiC31 integrase confers genomic integration and long-term transgene expression in rat retina. Invest Ophthalmol Vis Sci 46:2140–2146. doi:10.1167/iovs.04-1252
Chalberg TW, Portlock JL, Olivares EC, Thyagarajan B, Kirby PJ, Hillman RT, Hoelters J, Calos MP (2006) Integration specificity of phage phiC31 integrase in the human genome. J Mol Biol 357:28–48. doi:10.1016/j.jmb.2005.11.098
Chen Z, He Y, Shi B, Yang DC (2013) Human serum albumin from recombinant DNA technology: challenges and strategies. BBA-Gen Subj. 1830:5515–5525. doi:10.1016/j.bbagen.2013.04.037
Echelard Y, Williams JL, Destrempes MM et al (2009) Production of recombinant albumin by a herd of cloned transgenic cattle. Transgenic Res 18:361–376. doi:10.1007/s11248-008-9229-9
Fanali G, di Masi A, Trezza V, Marino M, Fasano M, Ascenzi P (2012) Human serum albumin: from bench to bedside. Mol Aspects Med 33:209–290. doi:10.1016/j.mam.2011.12.002
He Y, Ning T, Xie T et al (2011) Large-scale production of functional human serum albumin from transgenic rice seeds. Proc Natl Acad Sci USA 108:19078–19083. doi:10.1073/pnas.1109736108
Ishikawa Y, Tanaka N, Murakami K et al (2006) Phage phiC31 integrase-mediated genomic integration of the common cytokine receptor gamma chain in human T-cell lines. J Gene Med 8:646–653. doi:10.1002/jgm.891
Karow M, Chavez CL, Farruggio AP et al (2011) Site-specific recombinase strategy to create induced pluripotent stem cells efficiently with plasmid DNA. Stem Cells 29:1696–1704. doi:10.1002/stem.730
Kim H, Kim JS (2014) A guide to genome engineering with programmable nucleases. Nat Rev Genet 15:321–334. doi:10.1038/nrg3686
Kudriavtseva EA, Yun Yang L, Voeikova TA, Lomovskaia ND (1994) Use of a plasmid with integrative function of phage phiC31 for transfer of cloned genes into Streptomyces strains. Genetika 30:886–897
Luo Y, Liu J, Liu Q, Zheng L et al (2013) Chicken hypersensitive site-4 insulator increases human serum albumin expression in bovine mammary epithelial cells modified with phiC31 integrase. Biotechnol Lett 35:529–537. doi:10.1007/s10529-012-1125-y
Ma H, Ma Q, Lu Y et al (2014) PhiC31 integrase induces efficient site-specific recombination in the Capra hircus genome. DNA Cell Biol 33:484–491. doi:10.1089/dna.2013.2124
MacLennan S, Barbara JAJ (2006) Risks and side effects of therapy with plasma and plasma fractions. Best Pract Res Clin Haematol 19:169–189. doi:10.1016/j.beha.2005.01.033
Moghaddassi S, Eyestone W, Bishop CE (2014) TALEN-mediated modification of the bovine genome for large-scale production of human serum albumin. PLoS One 9:e89631. doi:10.1371/journal.pone.0089631
Ni W, Hu S, Qiao J et al (2012) PhiC31 integrase mediates efficient site-specific integration in sheep fibroblasts. Biosci Biotechnol Biochem 76:2093–2095. doi:10.1271/bbb.120439
Ou HL, Huang Y, Ma QW, Ren ZR, Huang SZ, Zeng FY, Zeng YT (2013) A highly efficient site-specific integration strategy using combination of homologous recombination and the phi C31 integrase. J Biotechnol 167:427–432. doi:10.1016/j.jbiotec.2013.08.001
Papapetrou EP, Lee G, Malani N et al (2011) Genomic safe harbors permit high beta-globin transgene expression in thalassemia induced pluripotent stem cells. Nat Biotechnol 29:73–78. doi:10.1038/nbt.1717
Qu LJ, Ma QW, Zhou ZW et al (2012) A profile of native integration sites used by phi C31 integrase in the bovine genome. J Genet Genomics 39:217–224. doi:10.1016/j.jgg.2012.03.004
Shani M, Barash I, Nathan M et al (1992) Expression of human serum albumin in the milk of transgenic mice. Transgenic Res 1:195–208
Thorpe HM, Smith MCM (1998) In vitro site-specific integration of bacteriophage DNA catalyzed by a recombinase of the resolvase/invertase family. Proc Natl Acad Sci USA 95:5505–5510. doi:10.1073/pnas.95.10.5505
Thyagarajan B, Olivares EC, Hollis RP, Ginsburg DS, Calos MP (2001) Site-specific genomic integration in mammalian cells mediated by phage phiC31 integrase. Mol Cell Biol 21:3926–3934. doi:10.1128/MCB.21.12.3926-3934.2001
Wang Y, Su J, Wang L, Xu W, Quan F, Liu J, Zhang Y (2011) The effects of 5-aza-2′-deoxycytidine and trichostatin A on gene expression and DNA methylation status in cloned bovine blastocysts. Cell Reprogram 13:297–306. doi:10.1089/cell.2010.0098
Yu Y, Wang Y, Tong Q, Liu X, Su F, Quan F, Guo Z, Zhang Y (2013) A site-specific recombinase-based method to produce antibiotic selectable marker free transgenic cattle. PLoS One 8:e62457
Acknowledgments
This work was supported by the National High Technology Research and Development Program of China (863 Program) (No. 2011AA100303).
Author information
Authors and Affiliations
Corresponding author
Additional information
Yan Luo and Yongsheng Wang have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Luo, Y., Wang, Y., Liu, J. et al. Production of transgenic cattle highly expressing human serum albumin in milk by phiC31 integrase-mediated gene delivery. Transgenic Res 24, 875–883 (2015). https://doi.org/10.1007/s11248-015-9898-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-015-9898-0