Abstract
Late blight of potato (Solanum tuberosum) caused by Phytophthora infestans significantly reduces the productivity of potato around the world. Resistance to late blight in potato is either qualitative or quantitative. Qualitative resistance governed by race-specific single R genes is well characterized and gives complete resistance, but is not durable. Quantitative resistance governed by polygenes gives partial resistance, but is durable in nature. However, the quantitative resistance mechanisms are poorly studied and are not efficiently exploited in potato breeding. A nontargeted metabolic profiling of resistant (F06037) and susceptible (Shepody) potato cultivars, using high-resolution liquid chromatography–mass spectrometry, was applied to elucidate the quantitative resistance mechanisms against P. infestans (US-8 genotype). The hydroxycinnamic acid amides (HCAAs) of the shunt phenylpropanoid pathway were highly induced following pathogen inoculation in F06037. In parallel, the transcript abundances of genes that catalyze the biosynthesis of these metabolites, such as 4-coumarate:CoA ligase, tyrosine decarboxylase, ornithine decarboxylase, tyramine hydroxycinnamoyl transferase, and putrescine hydroxycinnamoyl transferase, were also higher in the resistant genotype. Sequencing of the coding genes of these enzymes revealed single-nucleotide polymorphisms between resistant and susceptible genotypes, and the amino acid changes caused missense mutations altering protein functions. HCAAs deposited at host cell walls inhibit pathogen colonization, thus reducing lesion expansion. In addition, these also act as phytoalexins, leading to the reduced biomass of the pathogen. Following validation, the HCAAs can be used as biomarker metabolites for late blight resistance. The putative candidate genes can be either used to develop allele-specific markers for marker-assisted breeding programs or suitably stacked into elite cultivars through cisgenic approaches, following validation.





Similar content being viewed by others
References
Abu-Nada KAC (2010) Metabolic profiling to phenotype potato cultivars varying in horizontal resistance to leaf infection by Phytophthora infestans. Am J Plant Sci Biotechnol 4(2):55–64
Agati G, Azzarello E, Pollastri S, Tattini M (2012) Flavonoids as antioxidants in plants: location and functional significance. Plant Sci 196:67–76
Andrivon D, Corbière R, Lucas JM, Pasco C, Gravoueille JM, Pellé R, Dantec JP, Ellissèche D (2003) Resistance to late blight and soft rot in six potato progenies and glycoalkaloid contents in the tubers. Am J Potato Res 80(2):125–134
Arman M (2011) LC-ESI-MS characterisation of phytoalexins induced in chickpea and pea tissues in response to a biotic elicitor of Hypnea musciformis (red algae). Nat Prod Res 25(14):1352–1360
Ashkenazy H, Erez E, Martz E, Pupko T, Ben-Tal N (2010) ConSurf 2010: calculating evolutionary conservation in sequence and structure of proteins and nucleic acids. Nucleic Acids Res 38(suppl 2):W529–W533
Bassard JE, Ullmann P, Bernier F, Werck-Reichhart D (2010) Phenolamides: bridging polyamines to the phenolic metabolism. Phytochemistry 71(16):1808–1824
Bernards MA, Lopez ML, Zajicek J, Lewis NG (1995) Hydroxycinnamic acid-derived polymers constitute the polyaromatic domain of suberin. J Biol Chem 270(13):7382–7386
Birch PRJ, Bryan G, Fenton B, Gilroy EM, Hein I, Jones JT, Prashar A, Taylor MA, Torrance L, Toth IK (2012) Crops that feed the world 8: Potato: are the trends of increased global production sustainable? Food Security 4:477–508
Blount JW, Dixon RA, Paiva NL (1992) Stress responses in alfalfa (Medicago sativa L.) XVI. Antifungal activity of medicarpin and its biosynthetic precursors; implications for the genetic manipulation of stress metabolites. Physiol Mol Plant Pathol 41(5):333–349
Bollina V, Kumaraswamy GK, Kushalappa AC, Choo TM, Dion Y, Rioux S, Faubert D, Hamzehzarghani H (2010) Mass spectrometry–based metabolomics application to identify quantitative resistance–related metabolites in barley against Fusarium head blight. Mol Plant Pathol 11(6):769–782
Bryant D, Cummins I, Dixon DP, Edwards R (2006) Cloning and characterization of a theta class glutathione transferase from the potato pathogen Phytophthora infestans. Phytochemistry 67(14):1427–1434
Cho K, Kim Y, Wi SJ, Seo JB, Kwon J, Chung JH, Park KY, Nam MH (2012) Nontargeted metabolite profiling in compatible pathogen-inoculated tobacco (Nicotiana tabacum L. cv. Wisconsin 38) using UPLC-Q-TOF/MS. J Agric Food Chem 60(44):11015–11028
Clarke D (1982) The accumulation of cinnamic acid amides in the cell walls of potato tissue as an early response to fungal attack. In: Wood RKS (ed) Active defense mechanisms in plants. Plenum, London, 321 pp
Cowley T, Walters D (2002) Polyamine metabolism in barley reacting hypersensitively to the powdery mildew fungus Blumeria graminis f. sp. hordei. Plant Cell Environ 25(3):461–468
El-Bebany AF, Adam LR, Daayf F (2013) Differential accumulation of phenolic compounds in potato in response to weakly and highly aggressive isolates of Verticillium dahliae. Can J Plant Pathol 35(2):232–240
Fewell AM, Roddick JG (1997) Potato glycoalkaloid impairment of fungal development. Mycol Res 101(5):597–603
Fiehn O (2002) Metabolomics—the link between genotypes and phenotypes. Plant Mol Biol 48(1–2):155–171
Fleurence J, Negrel J (1989) Partial purification of tyramine feruloyl transferase from TMV inoculated tobacco leaves. Phytochemistry 28(3):733–736
Friedman M (2006) Potato glycoalkaloids and metabolites: roles in the plant and in the diet. J Agric Food Chem 54(23):8655–8681
Fry W (2008) Phytophthora infestans: the plant (and R gene) destroyer. Mol Plant Pathol 9(3):385–402
Fry WE, Goodwin SB (1997) Resurgence of the Irish potato famine fungus. BioScience 47:363–371
Gebhardt C (2012) Bridging the gap between genome analysis and precision breeding in potato. Trends Genet 29:248–256
González-Coloma A, López-Balboa C, Santana O, Reina M, Fraga BM (2011) Triterpene-based plant defenses. Phytochem Rev 10(2):245–260
Gunnaiah R, Kushalappa AC, Duggavathi R, Fox S, Somers DJ (2012) Integrated metabolo-proteomic approach to decipher the mechanisms by which wheat QTL (Fhb1) contributes to resistance against Fusarium graminearum. PLoS ONE 7(7):e40695
Hagel JM, Facchini PJ (2005) Elevated tyrosine decarboxylase and tyramine hydroxycinnamoyltransferase levels increase wound-induced tyramine-derived hydroxycinnamic acid amide accumulation in transgenic tobacco leaves. Planta 221(6):904–914
Haverkort A, Struik P, Visser RGF, Jacobsen E (2009) Applied biotechnology to combat late blight in potato caused by Phytophthora infestans. Potato Res 52(3):249–264
Katagiri Y, IBRAHIM RK, TAHARA S (2000) HPLC analysis of white lupin isoflavonoids. Biosci Biotechnol Biochem 64(6):1118–1125
Kaur H, Heinzel N, Schöttner M, Baldwin IT, Gális I (2010) R2R3-NaMYB8 regulates the accumulation of phenylpropanoid-polyamine conjugates, which are essential for local and systemic defense against insect herbivores in Nicotiana attenuata. Plant Physiol 152(3):1731–1747
Keller H, Hohlfeld H, Wray V, Hahlbrock K, Scheel D, Strack D (1996) Changes in the accumulation of soluble and cell wall-bound phenolics in elicitor-treated cell suspension cultures and fungus-infected leaves of Solanum tuberosum. Phytochemistry 42(2):389–396
Kolattukudy P (1981) Structure, biosynthesis, and biodegradation of cutin and suberin. Annu Rev Plant Physiol 32(1):539–567
Kolattukudy P (1984) Biochemistry and function of cutin and suberin. Can J Bot 62(12):2918–2933
Kou Y, Wang S (2010) Broad-spectrum and durability: understanding of quantitative disease resistance. Curr Opin Plant Biol 13(2):181–185
Kumar P, Henikoff S, Ng PC (2009) Predicting the effects of coding non-synonymous variants on protein function using the SIFT algorithm. Nat Protocol 4(7):1073–1081
Kumaraswamy GK, Bollina V, Kushalappa AC, Choo TM, Dion Y, Rioux S, Mamer O, Faubert D (2011) Metabolomics technology to phenotype resistance in barley against Gibberella zeae. Eur J Plant Pathol 130(1):29–43
Kushalappa AC, Gunnaiah R (2013) Metabolo-proteomics to discover plant biotic stress resistance genes. Trends Plant Sci 18:522–531
Langebartels C, Kerner K, Leonardi S, Schraudner M, Trost M, Heller W, Sandermann H (1991) Biochemical plant responses to ozone I. Differential induction of polyamine and ethylene biosynthesis in tobacco. Plant Physiol 95(3):882–889
Menden B, Kohlhoff M, Moerschbacher BM (2007) Wheat cells accumulate a syringyl-rich lignin during the hypersensitive resistance response. Phytochemistry 68(4):513–520
Mustafa NR, Verpoorte R (2007) Phenolic compounds in Catharanthus roseus. Phytochem Rev 6(2):243–258
Naoumkina MA, Zhao Q, Gallego-giraldo L, Dai X, Zhao PX, Dixon RA (2010) Genome-wide analysis of phenylpropanoid defence pathways. Mol Plant Pathol 11(6):829–846
Negrel J, Pollet B, Lapierre C (1996) Ether-linked ferulic acid amides in natural and wound periderms of potato tuber. Phytochemistry 43(6):1195–1199
Ng PC, Henikoff S (2003) SIFT: predicting amino acid changes that affect protein function. Nucleic Acids Res 31(13):3812–3814
Nick A, Wright AD, Rali T, Sticher O (1995) Antibacterial triterpenoids from Dillenia papuana and their structure–activity relationships. Phytochemistry 40(6):1691–1695
Nicot N, Hausman J-F, Hoffmann L, Evers D (2005) Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. J Exp Bot 56(421):2907–2914
Osbourn AE (1996) Preformed antimicrobial compounds and plant defense against fungal attack. Plant Cell 8(10):1821
Picman AK (1986) Biological activities of sesquiterpene lactones. Biochem Syst Ecol 14(3):255–281
Pluskal T, Castillo S, Villar-Briones A, Orešič M (2010) MZmine 2: modular framework for processing, visualizing, and analyzing mass spectrometry-based molecular profile data. BMC Bioinforma 11(1):395
Ponts N, Pinson-Gadais L, Boutigny A-L, Barreau C, Richard-Forget F (2011) Cinnamic-derived acids significantly affect Fusarium graminearum growth and in vitro synthesis of type B trichothecenes. Phytopathology 101(8):929–934
Prost I, Dhondt S, Rothe G, Vicente J, Rodriguez MJ, Kift N, Carbonne F, Griffiths G, Esquerré-Tugayé M-T, Rosahl S (2005) Evaluation of the antimicrobial activities of plant oxylipins supports their involvement in defense against pathogens. Plant Physiol 139(4):1902–1913
Ranathunge K, Schreiber L, Franke R (2011) Suberin research in the genomics era—new interest for an old polymer. Plant Sci 180(3):399–413
Reina-Pinto JJ, Yephremov A (2009) Surface lipids and plant defenses. Plant Physiol Biochem 47(6):540–549
Riley RG, Kolattukudy PE (1975) Evidence for covalently attached p-coumaric acid and ferulic acid in cutins and suberins. Plant Physiol 56(5):650–654
Roy A, Kucukural A, Zhang Y (2010) I-TASSER: a unified platform for automated protein structure and function prediction. Nat Protocol 5(4):725–738
Schauer N, Fernie AR (2006) Plant metabolomics: towards biological function and mechanism. Trend Plant Sci 11(10):508–516
Secor GA, Gudmestad NC (1999) Managing fungal diseases of potato. Can J Plant Pathol 21(3):213–221
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13(11):2498–2504
Simko I (2002) Comparative analysis of quantitative trait loci for foliage resistance to Phytophthora infestans in tuber-bearing Solanum species. Am J Potato Res 79(2):125–132
Stewart H, Bradshaw J, Pande B (2003) The effect of the presence of R-genes for resistance to late blight (Phytophthora infestans) of potato (Solanum tuberosum) on the underlying level of field resistance. Plant Pathol 52(2):193–198
Thomas R, Fang X, Ranathunge K, Anderson TR, Peterson CA, Bernards MA (2007) Soybean root suberin: anatomical distribution, chemical composition, and relationship to partial resistance to Phytophthora sojae. Plant Physiol 144(1):299–311
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acid Res 25(24):4876–4882
Vanholme R, Demedts B, Morreel K, Ralph J, Boerjan W (2010) Lignin biosynthesis and structure. Plant Physiol 153(3):895–905
Walters DR (2003) Polyamines and plant disease. Phytochemistry 64(1):97–107
Acknowledgments
This work was carried out with the aid of a grant from the International Development Research Center, Ottawa, Canada and with the financial support of the Foreign Affairs, Trade and Development Canada.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Fig. S1
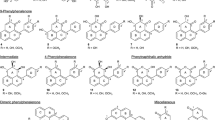
MS/MS spectra and in silico fragmentation of metabolites detected in potato cultivars with contrasting levels of late blight resistance following P. infestans or mock inoculation. a) p-Coumaroyltyramine, b) sinapoyltyramine. (JPEG 103 kb)
High resolution image
(TIFF 259 kb)
Fig. S2
Resistant related metabolites are clustered in to two groups mainly RRC and RRI. Their chemical groups and fold change are indicated as node shape and color. RRC is resistant related constitutive metabolites, RRI is resistant related induced metabolites and NI and NS is not identified in our study and non-significant at (P < 0.05). (JPEG 57 kb)
High resolution image
(TIFF 15516 kb)
Table S1
Late blight resistance related constitutive metabolites identified in potato cultivars following P. infestans or mock inoculation. (XLS 40 kb)
Table S2
Late blight resistance related induced metabolites identified in potato cultivars following P. infestans or mock inoculation. (XLS 36 kb)
Rights and permissions
About this article
Cite this article
Pushpa, D., Yogendra, K.N., Gunnaiah, R. et al. Identification of Late Blight Resistance-Related Metabolites and Genes in Potato through Nontargeted Metabolomics. Plant Mol Biol Rep 32, 584–595 (2014). https://doi.org/10.1007/s11105-013-0665-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-013-0665-1