Abstract
Background and aims
Soils can act as agents of natural selection, causing differential fitness among genotypes and/or families of the same plant species, especially when soils have extreme physical or chemical properties. More subtle changes in soils, such as variation in microbial communities, may also act as agents of selection. We hypothesized that variation in soil properties within a single river drainage can be a selective gradient, driving local adaptation in plants.
Methods
Using seeds collected from individual genotypes of Populus angustifolia James and soils collected from underneath the same trees, we use a reciprocal transplant design to test whether seedlings would be locally adapted to their parental soil type.
Results
We found three patterns: 1. Soils from beneath individual genotypes varied in pH, soil texture, nutrient content, microbial biomass and the physiological status of microorganisms. 2. Seedlings grown in local soils experienced 2.5-fold greater survival than seedlings planted in non-local soils. 3. Using a composite of height, number of leaves and leaf area to measure plant growth, seedlings grew ∼17.5% larger in their local soil than in non-local soil.
Conclusions
These data support the hypothesis that variation in soils across subtle gradients can act as an important selective agent, causing differential fitness and local adaptation in plants.


Similar content being viewed by others
References
Agresti A (2002) Categorical data analysis, 2nd edn. Wiley, New York
Agresti A (2007) An introduction to categorical data analysis, 2nd edn. Wiley, New York
Antonovics J (2006) Evolution in adjacent plant populations X: long-term persistence of prereproductive isolation at a mine boundary. Heredity 97:33–37
Ayres E, Steltzer H, Simmons BL, Simpson RT, Steinweg JM, Wallenstein MD et al (2009) Home-field advantage accelerates leaf litter decomposition in forests. Soil Biol Biochem 41:606–610
Bååth E, Anderson T-H (2003) Comparison of soil fungal/bacterial ratios in a pH gradient using physiological and PLFA-based techniques. Soil Biol Biochem 35:955–963
Bailey JK, Wooley SC, Lindroth RL, Whitham TG (2006) Importance of species interactions to community heritability: a genetic basis to trophic-level interactions. Ecol Lett 9:78–85
Bever JD, Westover KM, Antonovics J (1997) Incorporating the soil community into plant population dynamics: the utility of the feedback approach. J Ecol 85:561–573
Bezemer TM, Fountain M, Barea J, Christensen S, Dekker S, Duyts H, Van Hall R, Harvey JA, Hedlund K, Mikola J, Robin C, De Ruiter P, Setälä H, Scheu S, Šmilauer P, Van der Putten WH (2011) Divergent composition but similar function of soil food webs beneath individual plants: plant species and community effects. Ecology (In Press)
Bligh EG, Dwyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Codima JFCL, Jolliffe IT (1996) Size- and shape-related principal component analysis. Biometrics 52:710–716
Ellis AG, Weis AE (2006) Coexistence and differentiation of ‘flowering stones’: the role of local adaptation to soil microenvironment. J Ecol 94:322–335
Felix E, Tilley DR, Felton G, Flamino E (2008) Biomass production of hybrid poplar (Populus sp.) grown on deep-trenched municipal biosolids. Ecol Eng 33:8–14
Fleiss J, Levin B, Paik MC (2003) Statistical methods for rates and proportions, 3rd edn. Wiley, New York
Frostegård Å, Bååth E (1996) The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol Fert Soils 22:59–65
Frostegård Å, Bååth E, Tunlid A (1993) Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty acid analysis. Soil Biol Biochem 25:723–730
Gee GW, Bauder JM (1986) Particle-size analysis. Methods of soil analysis, Part 1, Physical and mineralogical methods. In: Agronomy monograph No. 9 2nd edn. American Society of Agronomy, Madison, pp 383–411
Hendershot WH, Lalande H, Duquette M (1993) Soil reaction and exchangeable acidity. In: Carter MR (ed) Soil sampling and methods of analysis. Lewis Publishers, CRC Press, Boca Raton, pp 141–146
Isebrands JG, Nelson ND (1982) Crown architecture of short rotation, intensively cultured Populus II. Branch morphology and distribution of leaves within the crown of Populus ‘Tristes’ as related to biomass production. Can J For Res 12:853–864
Johnson NC, Wilson GWT, Bowker MA, Wilson JA, Miller RM (2010) Resource limitation is a driver of local adaptation in mycorrhizal symbioses. P Natl Acad Sci USA 107:2093–2098
Keith AR, Bailey JK, Whitham TG (2010) A genetic basis to community repeatability and stability. Ecology 91:3398–3406
Macel M, Lawson CS, Mortimer SR, Šmilauerova M, Bischoff A et al (2007) Climate vs. soil factors in local adaptation in two common plant species. Ecology 88:424–433
Madritch MD, Donaldson JR, Lindroth RL (2006) Genetic identity of Populus tremuloides litter influences decomposition and nutrient release in a mixed forest stand. Ecosystems 9:528–537
Madritch MD, Greene SL, Lindroth RL (2009) Genetic mosaics of ecosystem functioning across aspen-dominated landscapes. Oecologia 160:119–127
O’Leary WM, Wilkinson SG (1988) Gram-positive bacteria. In: Ratledge C, Wilkinson SG (eds) Microbial lipids. Academic, London, pp 117–201
Olsson PA (1999) Signature fatty acids provide tools for determining of the distribution and interactions of mycorrhizal fungi in soils. FEMS Microbiol Ecol 29:303–310
Packer A, Clay K (2000) Soil pathogens and spatial patterns of seedling mortality in a temperate tree. Nature 404:278–281
Packer A, Clay K (2003) Soil pathogens and Prunus serotina seedling and sapling growth near conspecific trees. Ecology 84:108–119
Pregitzer C, Bailey JK, Hart SC, Schweitzer JA (2010) Soils as agents of selection: feedbacks between plants and soils alter seedling survival and performance. Evol Ecol 24:1045–1059
Reynolds HL, Packer A, Bever JD, Clay K (2003) Grassroots ecology: plant-microbe-soil interactions as drivers of plant community structure and dynamics. Ecology 84:2281–2291
Ronsheim ML (1996) Evidence against a frequency-dependent advantage for sexual reproduction in Allium vineale. Am Nat 147:718–734
Sambatti JBM, Rice KJ (2006) Local adaptation, patterns of selection, and gene flow in the Californian serpentine sunflower (Helianthus exilis). Evolution 60:696–710
Schmitt J, Gamble SE (1990) The effect of distance from the parental site on offspring performance and inbreeding depression in Impatiens capensis: a test of the local adaptation hypothesis. Evolution 44:2022–2030
Schweitzer JA, Bailey JK, Fisher DG, LeRoy CJ, Lonsdorf EV, Whitham TG et al (2008) Plant-soil-microorganism interactions: heritable relationship between plant genotype and associated soil microorganisms. Ecology 89:773–781
Schweitzer JA, Bailey JK, Fisher DG, LeRoy CJ, Whitham TG, Hart SC (2011) Functional and heritable consequences of genotypic variation on community assembly and ecosystem processes. In: Ohgushi T, Schmidt O, Holt R (eds) Ecology and evolution of trait-mediated indirect interactions: linking evolution, community, and ecosystem. Cambridge University Press, Cambridge
Smith DS, Bailey JK, Shuster SM, Whitham TG (2011) A geographic mosaic of trophic interactions and selection: trees, aphids and birds. J Evol Biol 42:422–429
Strickland MS, Lauber C, Fierer N, Bradford MA (2009) Testing the functional significance of microbial community composition. Ecology 90:441–451
Waser NM, Price MV (1985) Reciprocal transplant experiments with Delphinium nelsonii (Ranunculaceae): evidence for local adaptation. Am J Bot 72:1726–1732
White DC, Ringleberg DB (1998) Signature lipid biomarker analysis. In: Burlage RS, Atlas R, Stahl D, Gessey G, Sayler G (eds) Techniques in microbial ecology. Oxford University Press, New York, pp 255–272
White DC, Davis WM, Nichols JS, King JD, Bobbie RJ (1979) Determination of the sedimentary microbial biomass by extractable lipid phosphate. Oecologia 40:51–62
Whitham TG, Bailey JK, Schweitzer JA, Shuster SM, Bangert RK, LeRoy CJ et al (2006) A framework for community and ecosystem genetics: from genes to ecosystems. Nat Rev Genet 7:510–523
Whitham TG, DiFazio SP, Schweitzer JA, Shuster SM, Allan GJ, Bailey JK, Woolbright SA (2008) Extending genomics to natural communities and ecosystems. Science 320:492–495
Wilkinson SG (1988) Gram-negative bacteria. In: Ratledge C, Wilkinson SG (eds) Microbial lipids. Academic, London, pp 299–488
Wright JW, Stanton ML, Scherson R (2006) Local adaptation to serpentine and non-serpentine soils in Collinsia sparsiflora. Evol Ecol Res 8:1–21
Zelles L (1999) Fatty acid patterns of phospholipids and lipopolysaccarides in the characterization of microbial communities in soil: a review. Biol Fert Soils 3:111–129
Acknowledgements
The authors thank Clara Pregitzer, Todd Wojtowicz, Nashelly Meneses, Rocio Meneses, Brad Blake and Phil Patterson for their help in the field or greenhouse. Special thanks to Steve Overby and Dana Erickson (U.S. Forest Service, RMRS) for lab space and support for the PLFA/NFLA analyses. We would also like to thank Dylan Fischer and two anonymous reviewers for their comments, which greatly improved the manuscript. This research was supported by a National Science Foundation IGERT traineeship awarded to D.S.S. and by National Science Foundation FIBR grant DEB-0425908.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Harry Olde Venterink.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
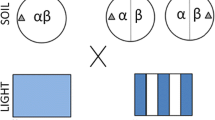
Design matrix for examining differential survival and performance between local and non-local soils. This graphic depicts how the five source soils were classified as local or non-local. Soils were considered local when seeds, which were collected from the same tree as the soil, were planted in that soil. Conversely, soils were considered non-local when seedlings collected from a different tree were planted in the soil. (PDF 34 kb)
Online Resource 2
Matrix of survival of all seedling families in all five soils. The black bars represent survival in local soils and gray bars represent survival in non-local soils. The numbers on the y-axis are the percent survival in each soil. (PDF 46 kb)
Online Resource 3
Table of statistics showing all of the analyses, what they tested, their respective test statistic and their corresponding p-value or confidence interval. (DOC 24 kb)
Rights and permissions
About this article
Cite this article
Smith, D.S., Schweitzer, J.A., Turk, P. et al. Soil-mediated local adaptation alters seedling survival and performance. Plant Soil 352, 243–251 (2012). https://doi.org/10.1007/s11104-011-0992-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-011-0992-7