Abstract
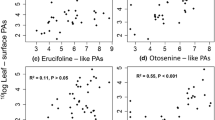
Lonicera maackii is an invasive shrub in North America for which allelopathic effects toward other plants or herbivores have been suspected. We characterized the major phenolic metabolites present in methanol extracts of L. maackii leaves. In addition, we examined the effects of methanol–water extracts of L. maackii leaves on seed germination of a target plant species and on feeding preference and growth rate of a generalist insect herbivore. A total of 13 individual major and minor compounds were detected in crude leaf extracts by high-performance liquid chromatography coupled to electronspray ionization-tandem mass spectrometry (ESI-MS/MS). Extracts were dominated by two major flavones, apigenin and luteolin, and their glucoside derivatives, apigenin-7-glucoside and luteolin-7-glucoside. Quantities of these compounds, along with chlorogenic acid, varied between two sampling points. Leaf extracts that contained these compounds were inhibitory to seed germination of Arabidopsis thaliana. In addition, treatment of artificial diet with leaf extracts deterred feeding of the generalist herbivore, Spodoptera exigua, in choice experiments but had no effect on growth rate in short-term no-choice bioassays. Purified apigenin tended to deter feeding by S. exigua and inhibited seed germination of A. thaliana. We conclude that leaves of L. maackii contain phenolic compounds, including apigenin and chlorogenic acid, capable of having biological effects on other plants and insects.



Similar content being viewed by others
References
Agrell, J., Oleszak, W., Stochmal, A., Olsen, M., and Andersen, P. 2003. Herbivore-induced responses in alfalfa (Medicago sativa). J. Chem. Ecol. 29:303–320.
Baerson, S. R., Sanchez-Moreiras, A., Pedrol-Bonjonch, N., Schulz, M., Kagan, I. A., Agarwal, A. K., eigosa, M. J., and Duke, S. O. 2005. Detoxification and transcriptome response in Arabidopsis seedlings exposed to the allelochemical benzoxazolin-2(3H)-one. J. Biol. Chem. 280:21867–21881.
Bais, H. P., Vepachedu, R., Gilroy, S., Callaway, R. M., and Vivanco, J. M. 2003. Allelopathy and exotic plant invasion: from molecules and genes to species interactions. Science. 301:1377–1380.
Bais, H. P., Walker, T. S., Stermitz, F. R., Hufbauer, R. A., and Vivanco, J. M. 2002. Enantiomeric-dependent phytotoxic and antimicrobial activity of (±)-catechin. A rhizosecreted racemic mixture from spotted knapweed. Plant Physiol. 128:1173–1179.
Blum, U. 1998. Effects of microbial utilization of phenolic acids and their phenolic acid breakdown products in allelopathic interactions. J. Chem. Ecol. 24:685–708.
Blum, U., and Gerig, T. M. 2006. Interrelationships between p-coumaric acid, evapotranspiration, soil water content, and leaf expansion. J. Chem. Ecol. 32:1573–1561.
Bonello, P., and Blodgett, J. T. 2003. Pinus nigra-Sphaeropsis sapinea as a model pathosystem to investigate local and systemic effects of fungal infection of pines. Physiol. Mol. Plant Pathol. 63:249–261.
Callaway, R. M., and Aschehoug, E. T. 2000. Invasive plants versus their new and old neighbors: a mechanism for exotic plant invasion. Science. 290:521–523.
Chaves, N., Sosa, T., and Escudero, J. C. 2001. Plant growth inhibiting flavonoids in exudate of Cistus ladanifer and in associated soils. J. Chem. Ecol. 27:623–631.
Collier, M. H., Vankat, J. L., and Hughes, M. R. 2002. Diminished plant richness and abundance below Lonicera maackii, an invasive shrub. Am. Mid. Nat. 147:60–71.
Cipollini, D., Stevenson, R., and Cipollini, K. 2008. Contrasting effects of allelochemicals from two invasive plants on the performance of a non-mycorrhizal plant. Intl. J. Plant. Sci., in press.
Clifford, M. N., Wu, W. G., and Kuhnert, N. 2006. The chlorogenic acids of Hemerocallis. Food Chem. 95:574–578.
Dorning, M., and Cipollini, D. 2006. Leaf and root extracts of the invasive shrub, Lonicera maackii, inhibit seed germination of three herbs with no autotoxic effects. Plant Ecol. 184:287–296.
Dowd, P. F., and Vega, F. E. 1996. Enzymatic oxidation products of allelochemicals as a basis for resistance against insects. Nat. Toxins. 4:85–91.
Eyles, A., Jones, W., iedl, K., Cipollini, D., Schwartz, S., Chan, K., Herms, D., and Bonello, P. 2007. Comparative phloem chemistry of Manchurian (Fraxinus mandshurica) and two North American ash species (F. americana and F. pennsylvanica). J. Chem. Ecol. 33:1430–1448.
Flamini, G., Braca, A., Cioni, P. J., and Morelli, I. 1997. Three new flavonoids and other constituents from Lonicera implexa. J. Nat. Prod. 60:449–452.
Graf, B. A., Milbury, P. E., and Blumberg, J. B. 2005. Flavonols, flavones, flavanones, and human health: epidemiological evidence. J. Med. Food. 8:281–290.
Haribal, M., and enwick, J. A. A. 1998. Isovitexin 6″″-O-β-D-glucopyranoside: a feeding deterrent to Pieris napi oleracea from Alliaria petiolata. Phytochemistry. 47:1237–1240.
Harrington, R. A., Brown, B. J., and eich, P. B. 1989. Ecophysiology of exotic and native shrubs in Southern Wisconsin. Oecologia. 80:356–367.
Hierro, J. L., and Callaway, R. W. 2003. Allelopathy and exotic plant invasion. Plant Soil. 256:29–39.
Hutchinson, T. F., and Vankat, J. L. 1997. Invasibility and effects of Amur honeysuckle in southwestern Ohio forests. Cons. Biol. 11:1117–1124.
Hvattum, E. 2002. Determination of phenolic compounds in rose hip (Rosa canina) using liquid chromatography coupled to electrospray ionisation tandem mass spectrometry and diode-array detection. Rapid Comm. Mass Spec. 16:655–662.
Iwashina, T. 2003. Flavonoid function and activity to plants and other organisms. Biol. Sci. Space. 17:24–44.
Maatta, K. R., Kamal-Eldin, A., and Torronen, A. R. 2003. High-performance liquid chromatography (HPLC) analysis of phenolic compounds in berries with diode array and electrospray ionization mass spectrometric (MS) detection: Ribes species. J. Agric. Food. Chem. 51:6736–6744.
Markham, K. R. 1982. Techniques of flavonoid identification. Academic, London.
Oberdorster, E., Clay, M. A., Cottam, D. M., Wilmot, F. A., McLachlan, J. A., and Milner, M. J. 2001. Common phytochemicals are ecdysteroid agonists and antagonists: a possible evolutionary link between vertebrate and invertebrate steroid hormones. J. Steroid Biochem. Mol. Biol. 77:229–238.
Pennacchio, M., Jefferson, L., and Havens, K. 2005. Arabidopsis thaliana: A new test species for phytotoxic bioassays. J. Chem. Ecol. 31:1877–1885.
Ridenour, W. M., and Callaway, R. M. 2001. The relative importance of allelopathy in interference: the effects of an invasive weed on a native bunchgrass. Oecologia. 126:444–450.
Rodriguez, B., odriguez, B., de la Torre, M. C., Simmonds, M. S. J., and Blaney, W. M. 1999. From a phagostimulant natural product to semisynthetic antifeedants against Spodoptera littoralis larvae: chemical transformations of the neoclerodane diterpenoid scutelgalin B. J. Nat. Prod. 62:594–600.
Skulman, B. W., Mattice, J. D., Cain, M. D., and Gbur, E. E. 2004. Evidence for allelopathic interference of Japanese honeysuckle (Lonicera japonica) to loblolly and shortleaf pine regeneration. Weed Sci. 52:433–439.
Sripad, G., Prakash, V., and Marasing ao, M. S. 1982. Extractability of polyphenols of sunflower seed in various solvents. J. Biosci. 4:145–152.
Tegelberg, R., and Julkunen-Titto, R. 2001. Quantitative changes in secondary metabolites in dark-leaved willow (Salix myrsinifolia) exposed to enhanced ultraviolet-b radiation. Physiol. Plant. 113:541–547.
Trisel, D. E. 1997. The invasive shrub, Lonicera maackii (Rupr.) Herder (Caprifoliaceae): Factors contributing to its success and its effect on native species. PhD Dissertation, Miami University, Oxford, USA.
USDA, NRCS. 2007. The PLANTS Database (http://plants.usda.gov, 4 December 2007). National Plant Data Center, Baton Rouge, LA., USA.
Verbeek, R., Plomp, A. C., Van Tol, E. A. F., and Van Noort, J. M. 2004. The flavones luteolin and apigenin inhibit in vitro antigen-specific proliferation and interferon-gamma production by murine and human autoimmune T cells. Biochem. Pharmacol. 68:621–629.
Wu, W., Yan, C. Y., Li, L., Liu, Z. Q., and Liu, S. Y. 2004. Studies on the flavones using liquid chromatography-electrospray ionization tandem mass spectrometry. J. Chrom. A. 1047:213–220.
Zhu, X. F., Zhang, H. X., and Lo, R. 2004. Phenolic compounds from the leaf extract of artichoke (Cynara scolymus L.) and their antimicrobial activities. J. Agric. Food. Chem. 52:7272–7278.
Acknowledgments
We thank the Ohio Plant Biotechnology Consortium and Wright State University for funding. Comments by two anonymous reviewers substantially improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cipollini, D., Stevenson, R., Enright, S. et al. Phenolic Metabolites in Leaves of the Invasive Shrub, Lonicera maackii, and Their Potential Phytotoxic and Anti-Herbivore Effects. J Chem Ecol 34, 144–152 (2008). https://doi.org/10.1007/s10886-008-9426-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-008-9426-2