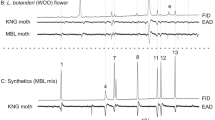
Abstract
Trees of the genus Glochidion (Phyllanthaceae) are pollinated by females of Epicephala moths (Gracillariidae) whose larvae consume the seeds of the flowers that they pollinate. Each Epicephala moth species is specific locally to a single host species, although two to four Glochidion hosts often cooccur. To investigate the role of olfactory signals in maintaining the plant−moth specificity, we analyzed floral scent composition of five Glochidion species by using gas chromatography–mass spectrometry (GC-MS) and conducted Y-tube olfactometer bioassays with Epicephala moths and their host flowers. The GC-MS analysis showed that the floral scents of the five Glochidion species are dominated by (R)-(−)- and (S)-(+)-linalool, and (E)- and (Z)-β-ocimene, and that each species produces 6–20 compounds. Transformation of scent profiles by using chord-normalized expected species shared distances and analysis of the data with nonmetric multidimensional scaling showed that floral volatiles of cooccurring Glochidion species can be distinguished by relative chemical composition, especially that of minor compounds. The bioassay with pollinators of Glochidion lanceolatum and Glochidion ruburm further indicated that Epicephala moths are capable of discriminating their hosts by using floral odor. The results suggest that the floral scent of Glochidion is one of the important key signals that mediate the encounters of the species-specific partners in the Glochidion–Epicephala mutualism.



Similar content being viewed by others
References
Andersson, S. and Dobson, H. E. M. 2003. Antennal responses to floral scents in the butterfly Heliconius melpomene. J. Chem. Ecol. 29:2319–2330.
Andersson, S., Nilsson, L. A., Groth, I., and Bergström, G. 2002. Floral scents in butterfly-pollinated plants: possible convergence in chemical composition. Bot. J. Linn. Soc. 140:129–153.
Bäckmann, A. C., Bengtsson, M., Borg-Karlsson, A. K., Liblikas, I., and Witzgall, P. 2001. Volatiles from apple (Malus domestica) eliciting antennal responses in female codling moth Cydia pomonella (L.) (Lepidoptera: Tortricidae): effect of plant injury and sampling technique. Z. Naturforsch. C 56:262–268.
Borg-Karlson, A. K., Valterova, I., and Nilsson, L. A. 1994. Volatile compounds from flowers of six species in the family Apiaceae: bouquets for different pollinators? Phytochemistry 35:111–119.
Bossart, J. L. 2003. Covariance of preference and performance on normal and novel hosts in a locally monophagous and locally polyphagous butterfly population. Oecologia 135:477–486.
Clarke, K. R. 1993. Nonparametric multivariate analyses of changes in community structure. Aust. J. Ecol. 18:117–143.
Cook, J. M. and Rasplus, J. Y. 2003. Mutualists with attitude: coevolving fig wasps and figs. Trends Ecol. Evol. 18:241–248.
Corner, E. J. H. 1965. Check-list of Ficus in Asia and Australasia with keys to identification. Gard. Bull. (Singapore) 21:1–185.
Davies, N. W. 1990. Gas chromatographic retention indices of monoterpenes and sesquiterpenes on methyl silicone and Carbowax 20 M phases. J. Chromatogr. 503:1–24.
Forister, M. L. 2004. Oviposition preference and larval performance within a diverging lineage of lycaenid butterflies. Ecol. Entomol. 29:264–272.
Gallagher, E. D. 1999. COMPAH documentation. http://www.es.umb.edu/edgwebp.htm
Gancel, A. L., Olle, D., Ollitrault, P., Luro, F., and Brillouet, J. M. 2002. Leaf and peel volatile compounds of an interspecific citrus somatic hybrid [Citrus aurantiflolia (Christm.) Swing. + Citrus paradisi MacFaydan]. Flavour Fragr. J. 17:416–424.
Gerlach, G. and Schill, R. 1991. Composition of orchid scents attracting euglossine bees. Bot. Acta 104:379–391.
Govaerts, R., Frodin, D. G., and Radcliffe-Smith, A. 2000. World checklist and bibliography of Euphorbiaceae. Royal Botanic Gardens, Kew, UK.
Grison-Pigé, L., Bessière, J. M., and Hossaert-Mckey, M. 2002a. Specific attraction of fig-pollinating wasps: Role of volatile compounds released by tropical figs. J. Chem. Ecol. 28:283–295.
Grison-Pigé, L., Hossaert-Mckey, M., Greeff, J. M., and Bessière, J. M. 2002b. Fig volatile compounds—a first comparative study. Phytochemistry 61:61–71.
Hern, A. and Dorn, S. 1999. Sexual dimorphism in the olfactory orientation of adult Cydia pomonella in response to alpha-farnesene. Entomol. Exp. Appl. 92:63–72.
Herre, E. A. 1989. Coevolution of reproductive characteristics in 12 species of New World figs and their pollinator wasps. Experientia 45:637–647.
Herre, E. A. 1996. An overview of studies on a community of Panamanian figs. J. Biogeogr. 23:593–607.
Herre, E. A. and West, S, A. 1997. Conflict of interest in a mutualism: documenting the elusive fig wasp–seed trade-off. Proc. R. Soc. Lond. B. 264:1501–1507.
Hossaert-Mckey, M., Gibernau, M., and Frey, J. E. 1994. Chemosensory attraction of fig wasps to substances produced by receptive figs. Entomol. Exp. Appl. 70:185–191.
Janzen, D. H. 1979. How to be a fig. Annu. Rev. Ecol. Syst. 10:13–51.
Jürgens, A. and Dötterl, S. 2004. Chemical composition of anther volatiles in Ranunculaceae: genera-specific profiles in Anemone, Aquilegia, Caltha, Pulsatilla, Ranunculus, and Trollius species. Am. J. Bot. 91:1969–1980.
Kato, M., Takimura, A., and Kawakita, A. 2003. An obligate pollination mutualism and reciprocal diversification in the tree genus Glochidion (Euphorbiaceae). Proc. Natl. Acad. Sci. U S A 100:5264–5267.
Kawakita, A. and Kato, M. 2006. Assessment of the diversity and species specificity of the mutualistic association between Epicephala moths and Glochidion trees. Mol. Ecol. 15:3567–3581.
Kawakita, A., Takimura, A., Terachi, T., Sota, T., and Kato, M. 2004. Cospeciation analysis of an obligate pollination mutualism: Have Glochidion trees (Euphorbiaceae) and pollinating Epicephala moths (Gracillaridae) diversified in parallel? Evolution 58:2201–2214.
Knudsen, J. T. and Tollsten, L. 1993. Trends in floral scent chemistry in pollination syndromes: floral scent composition in moth-pollinated taxa. Bot. J. Linn. Soc. 113:263–284.
Knudsen, J. T., Tollsten, L, and Bergströn, L. G. 1993. Floral scents—a checklist of volatile compounds isolated by head-space techniques. Phytochemistry 33:253–280.
Lopez-Vaamonde, C., Dixon, D. J., Cook, J. M., and Rasplus, J. Y. 2002. Revision of the Australian species of Pleistodontes (Hymenoptera: Agaonidae) fig-pollinating wasps and their host–plant associations. Zool. J. Linn. Soc. 136:637–683.
Machado, C. A., Jousselin, E., Kjellberg, F., Compton, S. G, and Herre, E. A. 2001. Phylogenetic relationships, historical biogeography, and character evolution of fig-pollinating wasps. Proc. R. Soc. Lond. B. 268:685–694.
Makholela, T. and Manning, J. C. 2006. First report of moth pollination in Struthiola ciliata (Thymelaeaceae) in southern Africa. S. Afr. J. Bot. 72:597–603.
Minh Tu, N. T., Onishi, Y., Son, U. S., Ogawa, E., Ukeda, H., and Sawamura, M. 2003. Characteristic odour components of Citrus inflata Hort. ex Tanaka (Mochiyu) cold-pressed peel oil. Flavour Fragr. J. 18:454–459.
Miyake, T., Yamaoka, R., and Yahara T. 1998. Floral scents of hawkmoth-pollinated flowers in Japan. J. Plant. Res. 111:199–205.
Molbo, D., Machado, C. A., Sevenster, J. G., Keller, L., and Herre, E. A. 2003. Cryptic species of fig-pollinating wasps: implications for the evolution of the fig–wasps mutualism, sex allocation, and precision of adaptation. Proc. Natl. Acad. Sci. U S A 100:5867–5872.
Nefdt, R. J C. and Compton, S. G. 1996. Regulation of seed and pollinator production in the fig–fig wasp mutualism. J. Anim. Ecol. 65:170–182.
Pellmyr, O. 2003. Yuccas, yucca moths, and coevolution: a review. Ann. Mo. Bot. Gard. 90:33–55.
Pellmyr, O., Tang, W., Groth, I., Bergstrom, G., and Thien, L. B. 1991. Cycad cone and angiosperm floral volatiles inferences for the evolution of insect pollination. Biochem. Syst. Ecol. 19:623–628.
Pettersson, S., Ervik, F., and Knudsen, J. K. 2004. Floral scent of bat-pollinated species: West Africa vs. the New World. Biol. J. Linn. Soc. 82:161–168.
Proctor, M., Yeo, P., and Lack, A. 1996. The natural history of pollination. Timber Press, Portland.
Raguso, R. A. and Pellmyr, O. 1998. Dynamic headspace analysis of floral volatiles: a comparison of methods. Oikos 81:238–254.
Raguso, R. A., Light, D. M., and Pichersky, E. 1996. Electroantennogram responses of Hyles lineata (Sphingidae: Lepidoptera) to volatile compounds from Clarkia breweri (Onagraceae) and other moth-pollinated flowers. J. Chem. Ecol. 22:1735–1765.
Raguso, R. A., Levin, R. A., Foose, S. E., Holmberg, M. W., and Mcdade, L. A. 2003. Fragrance chemistry, nocturnal rhythms and pollination “syndromes” in Nicotiana. Phytochemistry 63:265–284.
Sagrero-Nieves, L., Bartley, J. P., Espinosa, B. G., Domingues, X. A., and Verde S. J. 1997. Essential oil composition of Aristolochia brevipes Benth. Flavour Fragr. J. 12:401–403.
Singer, M. C., Ng, D., and Thomas, C. D. 1988. Heritability of oviposition preference and its relationship to offspring performance within a single insect population. Evolution 42:977–985.
Song, Q., Yang, D., Zhang, G., and Yang, C. 2001. Volatiles from Ficus hispida and their attractiveness to fig wasps. J. Chem. Ecol. 27:1929–1942.
Svensson, G. P., Hickman, M. O., Bartram, S., Boland, W., Pellmyr, O., and Raguso, R. A. 2005. Chemistry and geographic variation of floral scent in Yucca filamentosa (Agavaceae). Am. J. Bot. 92:1624–1631.
Traxler, M. A. and Joern, A. 1999. Performance tradeoffs for two hosts within and between populations of the oligophagous grasshopper Hesperotettix viridis (Acrididae). Oikos 87:239–250.
Trueblood, D. D., Gallagher, E. D., and Gould, D. M. 1994. Three stages of seasonal succession on the Savin Hill Cove mudflat, Boston Harbor. Limnol. Oceanogr. 39:1440–1454.
Via, S. 1986. Genetic covariance between oviposition preference and larval performance in an insect herbivore. Evolution 40:778–785.
Ware, A. B. and Compton, S. G. 1994. Responses of fig wasps to host plant volatile cues. J. Chem. Ecol. 20:785–802.
Weiblen, G. D. 2002. How to be a fig wasp. Annu. Rev. Ecol. Syst. 47:299–330.
Weiblen, G. D. 2004. Correlated evolution in fig pollination. Syst. Biol. 53:128–139.
Weiblen, G. D. and Bush, G. L. 2002. Speciation in fig pollinators and parasites. Mol. Ecol. 11:1573–1578.
Weiblen, G. D., Yu, D. W., and Stuart, S. A. 2001. Pollination and parasitism in functionally dioecious figs. Proc. R. Soc. Lond. B. 268:651–659.
Yokoyama, J. 2003. Cospeciation of figs and fig-wasps: a case study of endemic species pairs in the Ogasawara Islands. Popul. Ecol. 45:249–256.
Acknowledgements
We appreciate Y. Inui for providing the opportunity to conduct this study; T. Y. Chiang, T. W. Hsu, and S. C. Liu for assistance in the field in Taiwan; Y. Yamaoka and I. Shimizu for allowing us to use the GC and GC-MS; Y. Kumano and N. Fujiwara-Tsujii for technical advice in collecting and analyzing the floral scents; and the subject editor and anonymous reviewers for valuable comments on the manuscript. This work was supported by Grant-in-Aid for Scientific Research 15370012 from Japan Ministry of Education, Culture, Sports, Science and Technology (to M. K.) and by Grant for Basic Science Research Project from Sumitomo Foundation (to T. O.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Okamoto, T., Kawakita, A. & Kato, M. Interspecific Variation of Floral Scent Composition in Glochidion and its Association with Host-specific Pollinating Seed Parasite (Epicephala). J Chem Ecol 33, 1065–1081 (2007). https://doi.org/10.1007/s10886-007-9287-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-007-9287-0