Abstract
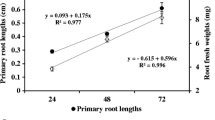
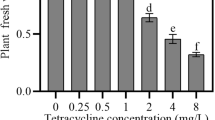
One commonly observed effect of phytotoxic compounds is the inhibition or delay of germination of sensitive seeds. Mustard (Sinapis alba L.) seeds were incubated with aqueous extracts of sunflower (Helianthus annuus L.) leaves. Although sunflower phytotoxins did not influence seed viability, extracts completely inhibited seed germination. Inhibition of germination was associated with alterations in reserve mobilization and generation of energy in the catabolic phase of germination. Degradation of lipids was suppressed by sunflower foliar extracts resulting in insufficient carbohydrate supply. The lack of respiratory substrates and decrease in energy (ATP) generation resulted in suppression of the anabolic phase of seed germination and ultimately growth inhibition.







Similar content being viewed by others
References
Abrahim, D., Braquini, W. L., Kelmer-Bracht, A. M., and Ishii-Iwamoto, E. L. 2000. Effects of four monoterpenes on germination, primary root growth, and mitochondrial respiration of maize. J. Chem. Ecol. 26:611–624.
Azania, A. A. P. M., Azania, C. A. M., Alives, P. L. C. A., Palaniraj, R., Kadian, H. S., Sati, S. C., Rawat, L. S., Dahiya, D. S., and Narwal, S. 2003. Allelopathic plants 7. Sunflower (Helianthus annuus L.). Allelopathy J. 11:1–20.
Baleroni, C. R. S., Ferrarese, M. L. L., Souza, N. E., and Ferrarese-Filho, O. 2000. Lipid accumulation during canola seed germination in response to cinnamic acid derivatives. Biol. Plant. 43:313–316.
Batish, D. R., Tung, P., Sing, H. P., and Kohli, R. K. 2002. Phytotoxicity of sunflower residues against some summer crops. J. Agron. Crop Sci. 188:19–24.
Bethke, P. C., Swanson, S. J., Hillmer, S., and Jones, R. L. 1998. From storage compartment to lytic organelle: The metamorphosis of the aleurone protein storage vacuole. Ann. Bot. 82:399–412.
Bewley, J. D. and Black, M. 1994. Seeds. Physiology of Development and Germination. Plenum Press, New York.
Bielawski, W., Dojczew, D., Kączkowski, J., and Kolbuszewska-Podres, W. 1994. Enzymes of protein breakdown in germinating Triticale grains resistant and susceptible to pre-harvest sprouting. Acta Physiol. Plant. 16:19–26.
Bogatek, R., ZARSKA-MACIEJEWSKA, B., Sińska, I., and LEWAK, ST. 1989. The embryonic axis controls lipid catabolism in cotyledons of apple seeds during germination. Physiol. Plant. 76:557–562.
Bogatek, R., Oracz, K., and Gniazdowska, A. 2005. Ethylene and ABA production in germinating seeds during allelopathy stress, pp. 292–296, in J.D.I. Harper, A. AN, H. WU, and J. H. Kent (eds.). Proceedings of the 4th Word Congress on Allelopathy. Charles Sturt University, Locked Bag 588, Wagga Wagga, NSW, Australia. International Allelopathy Society.
Bogatek, R., Gniazdowska, A., Zakrzewska, W., Oracz, K., and Gawroński, S. W. 2006. Allelopathic effect of sunflower extracts on mustard seed germination and seedling growth. Biol. Plant. 50:156–158.
Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal. Biochem. 72:248–254.
Cosgrove, D. J. 1997. Relaxation in a high-stress environment: The molecular bases of extensible cell walls and cell enlargement. Plant Cell 9:1031–1041.
Cruz-Ortega, R., Anaya, A. L., and Ramos, L. 1988. Effects of allelopathic compounds of corn pollen on respiration and cell division of watermelon. J. Chem. Ecol. 14:71–86.
Edelstein, M., Bradford, K. J., and Burger, D. W. 2001. Metabolic heat and CO2 production rates during germination of melon (Cucumis melo L.) seeds measured by microcalorimetry. Seed Sci. Res. 11:265–272.
Hosokawa, T., Okamoto, T., and Minamikawa, T. 1999. Characterisation of serine endopeptidases in cotyledons of germinated Vigna mungo seeds. J. Plant Res. 112:217–221.
Huang, A. H. C. 1992. Oil bodies and oleosins in seeds. Annu. Rev. Plant Phys. 43:177–200.
Inderjit and Callaway, R. M. 2003. Experimental designs for the study of allelopathy. Plant Soil 256:1–11.
Inderjit and Duke, S. O. 2003. Ecophysiological aspect of allelopathy. Planta 217:529–539.
Irons, S. and Burnside, O. 1982. Competitive and allelopathic effect of sunflower (Helianthus annuus). Weed Sci. 30:327–377.
Karnovsky, M. J. 1965. A formaldehyde–glutaraldehyde fixative of high osmolality for use in electron microscopy. J. Cell Biol. 25A:137–138.
Lacey, D. J. and Hills, M. J. 1996. Heterogeneity of the endoplasmic reticulum with respect to lipid synthesis in developing seeds of Brassica napus L. Planta 199:545–551.
Leather, G. R. 1987. Weed control using allelopathic sunflowers and herbicide. Plant Soil 98:17–231.
Lewitt, J., Lovett, J. V., and Garlick, P. R. 1984. Datura stramonium allelochemicals: Longevity in soil, and ultrastructural effects on root tip cells of Helianthus annuus L. New Phytol. 97:213–218.
Lin, Y. H. and Huang, A. H. C. 1983. Lipase and lipid bodies of rape and mustard seeds. Arch. Biochem. Biophys. 225:360–369.
Macias, F. A., Varela, R. M., Torres, A., and Molinillo, J. M. G. 2000. Potential allelopathic activity of natural plant heliannanes: A proposal of absolute configuration and nomenclature. J. Chem. Ecol. 26:2173–2186.
Macias, F. A., Molinillo, J. M. G., Galindo, J. C. G., Varela, R. M., Simonet, A. M., and Castellano, D. 2001. The use of allelopathic studies in the search for natural herbicides. J. Crop Prod. 4:237–255.
Macias, F. A., Varela, R. M., Torres, A., Galindo, J. L. G., and Molinillo, J. M. G. 2002. Allelochemicals from sunflowers: Chemistry, bioactivity and applications, pp. 73–87, in Inderjit and A.U. Mallik (eds.). Chemical Ecology of Plants: Allelopathy in Aquatic and Terrestrial Ecosystems. Birkhauser Verlag, Basel.
Maffei, M., Bertea, C. M., Garneri, F., and Scanneri, S. 1999. Effect of benzoic acid hydroxy- and methoxy-ring substituents during cucumber (Cucumis sativus L.) germination. I. Isocitrate lyase and catalase activity. Plant Sci. 141:139–147.
Murphy, D. J., Cummins, I., and Ryan, J. 1989. Immunocytochemical and biochemical study of the biosynthesis and mobilisation of the major seed storage proteins of Brassica napus. Plant Physiol. Biochem. 27:647–657.
Olempska-Beer, Z. and Bautz-Freeze, E. 1984. Optimal extraction conditions for high-performance liquid chromatography determination of nucleotides in yeast. Anal. Biochem. 140:236–224.
Peňuelas, J., Ribas-Carbo, M., and Giles, L. 1996. Effect of allelochemicals on plant respiration and oxygen isotope fractionation by alternative oxidase. J. Chem. Ecol. 22:801–805.
Reigosa, M. J., Sanchez-Moreiras, A., and Gonzales, L. 1999. Ecophysiological approach in allelopathy. Crit. Rev. Plant Sci. 18:577–608.
Reynolds, E. S. 1963. The use of lead citrate at high pH as an electron opaque stain for electron microscopy. J. Cell Biol. 17:208–213.
Rice, E. L. 1984. Allelopathy. Academic Press, Orlando. 422 pp.
Rosnitschek, I. and Theimer, T. T. 1980. Properties of membrane-bound triglyceride lipase of rapeseed (Brassica napus L.) cotyledons. Planta 148:193–198.
Saglio, P. H. M., Daniels, M. J., and Pradet, A. 1979. ATP and energy charge as criteria of growth and metabolic activity of mullicutes: Application to Spiroplasma citri. J. Gen. Microbiol. 110:13–20.
Schlereth, A., Standhardt, D., Mock, H-P., and Müntz, K. 2001. Stored cysteine proteinases start globulin mobilization in protein bodies of embryonic axes and cotyledons during vetch (Vicia sativa L.) seed germination. Planta 212:718–727.
Schon, M. K. and Einhellig, F. 1982. Allelopathic effects of cultivated sunflower on grain sorghum. Bot. Gaz. 143:505–510.
Smith, N. B., Criddle, R. S., and Hansen, L. D. 2000. Plant growth, respiration and environmental stress. J. Plant Biol. 27:89–97.
Smith, B. N., Harris, L. C., McCarlie, V. M., Stradling, D. L., Thygerson, T., Walker, J., Criddle, R. S., and Hansen, L. D. 2001. Unit 1. Time, plant growth, respiration, and temperature, pp 1–11, in M Pessarakli (ed.) Handbook of Plant and Crop Physiology. Marcel Dekker, New York.
Spoelstra, P., Joosen, R. V. L., Van der Plas, L. H. W., and Hilhorst, H. W. M. 2002. The distribution of ATP within tomato (Lycopersicon esculentum Mill.) embryos correlates with germination whereas total ATP concentration does not. Seed Sci. Res. 12:231–238.
Wanner, G. and Theimer, R. R. 1978. Membranous appendices of spherosomes. Possible role in fat utilization in germinating oil seeds. Planta 140:163–169.
Werker, E. and Vaughan, J. G. 1974. Anatomical and ultrastructural changes in aleurone and myrosin cells of Sinapis alba during germination. Planta 116:243–255.
Weston, L. A. and Duke, S. O. 2003. Weed and crop allelopathy. Crit. Rev. Plant Sci. 22:367–389.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ewa Kupidłowska and Agnieszka Gniazdowska contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kupidłowska, E., Gniazdowska, A., Stępień, J. et al. Impact of Sunflower (Helianthus annuus L.) Extracts Upon Reserve Mobilization and Energy Metabolism in Germinating Mustard (Sinapis alba L.) Seeds. J Chem Ecol 32, 2569–2583 (2006). https://doi.org/10.1007/s10886-006-9183-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-006-9183-z