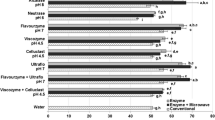
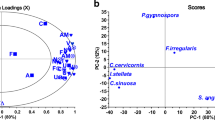
Abstract
The traditional method to obtain phycocolloids from seaweeds implies successive extraction steps with cold and hot water. The residual cake derived from phycocolloids obtaining process of red seaweed Porphyra columbina is a waste containing 27 % protein and 10.7-mg gallic acid equivalents (100 g)−1. Seaweeds contain functional proteins, and the enzymatic hydrolysis of these proteins has been shown to release bioactive peptides. The aims of this study were to extract bioactive peptides and polyphenols after enzymatic hydrolysis of the residual cake and to evaluate their ACE inhibitory and antioxidant capacities (TEAC, DPPH, and copper-chelating activity). Residual cake hydrolysate has low molecular weight peptides containing Asp, Glu, Ala, and Leu. Residual cake hydrolysate had higher protein solubility than residual cake. ACE inhibition (≈45 %) and radical scavenging activity (TEAC and DPPH inhibition) were attributed mainly to low molecular weight peptides (500 Da) and polyphenols compounds released during proteolysis. The 50 % inhibition protein concentration value (IC50) corresponded to residual cake hydrolysate was 1.01 ± 0.02 and 0.91 ± 0.01 g L−1, for ABTS and DPPH, respectively. Also, residual cake hydrolysate had high copper-chelating activity (≈97.5 %). Hydrolysis could be used as a means to obtain ACE inhibitory and antioxidant compounds (peptides and polyphenols) from algae protein waste and add value to the phycocolloids extraction process.





Similar content being viewed by others
References
Alaiz M, Navarro J, Giron J, Vioque E (1992) Amino acid analysis by high-performance liquid chromatography after derivatization with diethylethoxymethylenemalonate. J Chromatogr A 591:181–186
AOAC (1995) Official methods of analysis, 16th edn. AOAC International, Arlington
Athukorala Y, Jeon YJ (2005) Screening for angiotensin I-converting enzyme inhibitory activity of Ecklonia cava. J Food Sci Nutr 10:134–139
Balti R, Bougatef A, El Hadj Ali N, Ktari N, Jellouli K, Nedjar-Arroume N, Nasri M (2011) Comparative study on biochemical properties and antioxidative activity of cuttlefish (Sepia officinalis) protein hydrolysates produced by Alcalase and Bacillus licheniformis NH1 proteases. J Amino Acids 2011:107179
Chew YL, Lim YY, Omar M, Khoo KS (2008) Antioxidant activity of three edible seaweeds from two areas in South East Asia. LWT- Food Sci Technol 41:1067–1072
Cian R, Lugren P, Drago S (2011) Effect of extrusion process on antioxidant and ACE inhibition properties from bovine hemoglobin concentrate hydrolysates incorporated into expanded maize products. Int J Food Sci Nutr 62:774–780
Dávalos A, Miguel M, Bartolomé B, López-Fadiño R (2004) Antioxidant activity of peptides derived from egg white proteins by enzymatic hydrolysis. J Food Protect 67:1939–1944
Drago S, González R (2001). Foaming properties of enzymatically hydrolyzed wheat gluten. IFSET 1:269-273
Fleurence J (1999) Seaweed proteins: biochemical, nutritional aspects and potential uses. Trends Food Sci Technol 10:25–28
Galland-Irmouli A, Fleurence J, Lamghari R, Luçon M, Rouxel C, Barbaroux O, Bronowicki J, Villaume C, Guéant J (1999) Nutritional value of proteins from edible seaweed Palmaria palmata (Dulse). J Nutr Biochem 10:353–359
Gressler V, Yokoya N, Fujii M, Colepicolo P, Mancini Filho J, Torres R, Pinto E (2010) Lipid, fatty acid, protein, amino acid and ash contents in four Brazilian red algae species. Food Chem 120:585–590
Hayakari M, Kondo Y, Izumi H (1978) A rapid and simple spectrophotometric assay of angiotensin-converting enzyme. Anal Biochem 84:361–369
In M, Chae H, Oh N (2002) Process development for heme-enriched by enzymatic hydrolysis oh hemoglobin. Bioresource Technol 84:63–68
Kabirullah M, Wills R (1981) Functional properties of sunflower protein following partial hydrolysis with proteases. LWT- Food Sci Technol 14:232–236
Khantaphant S, Benjakul S, Kishimura H (2011) Antioxidative and ACE inhibitory activities of protein hydrolysates from the muscle of brownstripe red snapper prepared using pyloric caeca and commercial proteases. Process Biochem 46:318–327
Kong X, Guo M, Hua Y, Cao D, Zhang C (2008) Enzymatic preparation of immunomodulating hydrolysates from soy proteins. Bioresource Technol 99:8873–8879
Lourenço SO, Barbarino E, Lavín PL, Lanfer Marquez UM, Aidar E (2004) Distribution of intracellular nitrogen in marine microalgae. Calculation of new nitrogen-to-protein conversion factors. Eur J Phycol 39:17–32
Lowry O, Rosebrough N, Farr L, Randall R (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193–265
Mabeau S, Cavaloc E, Fleurence J, Lahaye M (1992) New seaweed based ingredients for the food industry. Int Food Ing 3:38–45
Megías C, Pedroche J, Yust M, Girón-Calle J, Alaiz M, Millán F, Vioque J (2008) Production of copper-chelating peptides after hydrolysis of sunflower proteins with pepsin and pancreatin. LWT- Food Sci Technol 41:1973–1977
Mohamed S, Hashim S, Rahman H (2012) Seaweeds: a sustainable functional food for complementary and alternative therapy. Trends Food Sci Technol 23:83–96
Moosmann B, Behl C (2002) Secretory peptide hormones are biochemical antioxidants: structure-activity relationship. Mol Pharmacol 61:260–268
Murakami M, Tonouchi H, Takahashi R, Kitazawa H, Kawai Y, Negishi H, Saito T (2004) Structural analysis of a new anti-hypertensive peptide (β-Lactosin B) isolated from a commercial whey product. J Dairy Sci 87:1967–1974
Ngo D-H, Wijesekara I, Vo T-S, Ta QV, Kim S-K (2011) Marine food-derived functional ingredients as potential antioxidants in the food industry: an overview. Food Res Int 44:523–529
Nielsen P, Petersen D, Dambmann C (2001) Improved method for determining food protein degree of hydrolysis. J Food Sci 66:642–646
Norziah MH, Ching CY (2000) Nutritional composition of edible seaweed Gracilaria changi. Food Chem 68:69–76
Pérez A, Farías S, Strobl A, Pérez L, López C, Piñeiro A, Roses O, Fajardo M (2007) Levels of essential and toxic elements in Porphyra columbina and Ulva sp. from San Jorge Gulf, Patagonia Argentina. Sci Total Environ 376:51–59
Qu W, Maa H, Pan Z, Luo L, Wanga Z, He R (2010) Preparation and antihypertensive activity of peptides from Porphyra yezoensis. Food Chem 123:14–20
Quian Z, Jung W, Kim S (2008) Free radical scavenging activity of a novel antioxidative peptide purified from hydrolysate of bullfrog skin, Rana catesbeiana Shaw. Bioresource Technol 99:1690–1698
Ren J, Zhao M, Shi J, Wang J, Jiang Y, Cui C, Kakuda Y, Xue S (2008) Purification and identification of antioxidant peptides from grass carp muscle hydrolysates by consecutive chromatography and electrospray ionization-mass spectrometry. Food Chem 108:727–736
Saiga A, Tanabe S, Nishimura T (2003) Antioxidant activity of peptides obtained from porcine myofibrillar proteins by protease treatment. J Agric Food Chem 51:3661–3667
Sánchez-Moreno C, Larrauri J, Saura-Calixto F (1998) A procedure to measure the antiradical efficiency of polyphenols. J Agric Food Chem 76:270–276
Sato M, Hoskawa T, Yamaguchi T, Nakano T, Muramoto K, Kahara T (2002) Angiotensin I-converting enzyme inhibitory peptides derived from wakame (Undaria pinnatifida) and their antihypertensive effect in spontaneously hypertensive rats. J Agric Food Chem 50:6245–6252
Schanderl S (1970) Tannins and related phenolics. In: Joslyn MA (ed) Methods in food: analysis physical, chemical and instrumental methods of analysis, 2nd edn. Academic, New York, pp 701–725
Sheih I, Fang T, Wu T (2009) Isolation and characterisation of a novel angiotensin I converting enzyme (ACE) inhibitory peptide from the algae protein waste. Food Chem 115:279–284
Song L, Li T, Yu R, Yan C, Ren S, Zhao Y (2008) Antioxidant activities of hydrolysates of Arca subcrenata prepared with three proteases. Mar Drugs 6:607–619
Souza B, Cerqueira M, Bourbon A, Pinheiro A, Martins J, Texeira J, Coimbra M, Vicente A (2012) Chemical characterization and antioxidant activity of sulfated polysaccharide from the red seaweed Gracilaria birdiae. Food Hydrocoll 27:287–292
Suetsuna K, Chen JR (2001) Identification of antihypertensive peptides from peptic digests of two microalgae, Chlorella vulgaris and Spirulina platensis. Mar Biotechnol 3:305–309
Torres-Fuentes C, Alaiz M, Vioque J (2011) Affinity purification and characterisation of chelating peptides from chickpea protein hydrolysates. Food Chem 129:485–490
Turkmen N, Velioglu YS, Sari F, Polat G (2007) Effect of extraction conditions on measured total polyphenol contents and antioxidant and antibacterial activities of black tea. Molecules 12:484–496
Uma DB, Ho CW, Wan Aida WM (2010) Optimization of extraction parameters of total phenolic compounds from Henna (Lawsonia inermis) leaves. Sains Malaysiana 39:119–128
Urbano G, Goñi I (2002) Bioavailability of nutrients in rats fed on edible seaweeds, Nori (Porphyra tenera) and Wakame (Undaria pinnatifida), as a source of dietary fibre. Food Chem 76:281–286
Wang J, Zhao M, Zhao Q, Jiang Y (2007) Antioxidant properties of papain hydrolysates of wheat gluten in different oxidation systems. Food Chem 101:1658–1663
Wang Y, Wang Z, Cheng S, Han F (2008) Aqueous enzymatic extraction of oil and protein hydrolysates from peanut. Food Sci Technol Res 14:533–540
Wang T, Jónsdóttir R, Ólafsdóttir G (2009) Total phenolic compounds, radical scavenging and metal chelation of extracts form Icelandic seaweeds. Food Chem 116:240–248
Wang T, Jónsdóttir R, Kristinsson H, Hreggvidsson G, Jónsson J, Thorkelsson G, Ólafsdóttir G (2010) Enzyme-enhanced extraction of antioxidant ingredients from red algae Palmaria palmata. LWT- Food Sci Technol 43:1387–1393
Wijesekara I, Kim S (2010) Angiotensin-I-converting enzyme (ACE) inhibitors from marine resources: prospects in the pharmaceutical industry. Mar Drugs 8:1080–1093
Wilson J, Hayes M, Carney B (2011) Angiotensin-I-converting enzyme and prolyl endopeptidase inhibitory peptides from natural sources with a focus on marine processing by-products. Food Chem 129:235–244
Wu H, He H, Chen X, Sun C, Zhang Y, Zhou B (2008) Purification and identification of novel angiotensin-I-converting enzyme inhibitory peptides from shark meat hydrolysate. Process Biochem 43:457–461
Xie Z, Huang J, Xu X, Jin Z (2008) Antioxidant activity of peptides isolated from alfalfa leaf protein hydrolysate. Food Chem 111:370–376
You L, Zhao M, Cui C, Zhao H, Yang B (2009) Effect of degree of hydrolysis on the antioxidant activity of loach (Misgurnus anguillicaudatus) protein hydrolysates. Innov Food Sci Emerg Technol 10:235–240
You L, Regenstein JM, Liu RH (2010) Optimization of hydrolysis conditions for the production of antioxidant peptides from fish gelatin using response surface methodology. J Food Sci 75:582–587
Yust M, Pedroche J, Girón-Calle J, Vioque J, Millán F, Alaiz M (2004) Determination of tryptophan by high-performance liquid chromatography of alkaline hydrolysates with spectrophotometric detection. Food Chem 85:317–320
Zhang Z-S, Li D, Wang LJ, Ozkan N, Chen XD, Mao Z-H, Yang H-Z (2007) Optimisation of ethanol-water extraction of lignans from flax seed. J Sep Purif Technol 57:17–24
Acknowledgements
All authors read and approved the final manuscript. The authors are thankful to Lic. Maira Tittarelli for assistance and project CAI + D 2009-PI-54-258 of the Universidad Nacional del Litoral.
Conflicts of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cian, R.E., Alaiz, M., Vioque, J. et al. Enzyme proteolysis enhanced extraction of ACE inhibitory and antioxidant compounds (peptides and polyphenols) from Porphyra columbina residual cake. J Appl Phycol 25, 1197–1206 (2013). https://doi.org/10.1007/s10811-012-9913-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-012-9913-2