Abstract
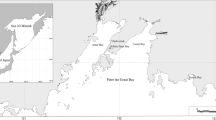
New phosphatase and DNase inhibition tests for assessing the total pollution of a natural marine ecosystem were applied. The seawater samples with different pollution degrees were collected in the Troitsa Bay of the Peter the Great Bay (the Sea of Japan). The sensitivity of the alkaline phosphatase test to integrated pollution was in accordance with the sensitivity of the standard sea urchin sperm cell toxicity test. The increased seawater pollution level was shown to result in an up to fourfold increase in specific activities of acid and alkaline phosphatases from the mussel Crenomytilus grayanus. It was demonstrated that a complex methodological approach can be used to assess marine water areas, as well as to assess the biological conditions of invertebrates adapting to different environmental and anthropogenic effects.

Similar content being viewed by others
References
Anderson, J. W., Neff, J. M., & Cox, B. A. (1974). Characteristics of dispersions and water-soluble extracts of crude and refined oils and their toxicity to estuarine crustaceans and fish. Marine Biology, 27(7), 679–690.
Belan, T. A. (2003). Benthos abundance pattern and species composition in conditions of pollution in Amurky Bay (the Peter the Great Bay, the Sea of Japan). Marine Pollution Bulletin, 46, 1111–1119.
Belcheva, N. N., Istomina, A. A., Kudryashova, Y. V., & Chelomin, V. P. (2013). Marine environment assessment based on oxidative stress indicators and heavy metal content in tissues of the mussel Crenomytilus grayanus (Dunker, 1853) (Bivalvia: Mytilidae). Russian Journal of Marine Biology, 39(4), 281–286.
Blackstock, J. (1984). Biochemical metabolic regulatory responses of marine invertebrates to natural environmental change and marine pollution. Oceanography and Marine Biology, An Annual Review edt. Harold Barne, 22, 263–316.
Bredford, M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Buznikov G.A., Podmariov, V.K. (1975). Sea urchins Strongylocentrotus drobachiensis, S. nudus, S. intermedius. Objects of biological development, ed. “Nauka”, Moscow, Russia, 188–214.
Carballeira, C., Ramos-Gomez, J., Martin-Diaz, L., & DellValls, T. A. (2012). Identification of specific malformations of sea urchin larvae for toxicity assessment: application to marine pisciculture effluents. Marine Environmental Research, 77, 12–22.
Chernyaev, A. P., Luk’yanova, O. N., & Cherkashin, S. A. (2006). Distribution of petrolium hydrocarbons and estimation of biota state in the Amursky Bay (Japan Sea). Ecological Chemistry, 15(1), 28–38.
Dinnel, P. A., Link, J. M., & Sober, Q. J. (1987). Improved methodology for a sea urchin sperm cell bioassay for marine waters. Archives of Environmental Contamination and Toxicology, 16, 23–32.
Dinnel, P.A., Pagano, G.C., Oshida, P.S. (1988). A sea urchin test system for marine environmental monitoring. Echinoderm biology. Eds. Burke et all. Netherlands, Rotterdam: Balkema, 611–619.
Dinnel, P. A., Link, J. M., Stober, Q. J., Letourneau, M. W., & Roberts, W. E. (1989). Comparative sensitivity of sea urchin sperm bioassays to metals and pesticides. Archives of Environmental Contamination and Toxicology, 18, 748–755.
Dinnel, P. A. (1995). Evolution and present status of the sea urchin sperm test. Russian Journal of Marine Biology, 21(6), 390–397.
Dixon, M., Webb, E.C. (1982). Enzymes, ed. “Mir”, Moskow, Russia, 2, 440–446.
Durkina, V. B., & Evtushenko, Z. S. (1991). Changes in activity of certain enzymes in sea urchin embryos and larvae after exposure of adult organisms to heavy metals. Marine Ecology Progress Series, 72, 111–115.
Elumalai, M., Antunes, C., & Guilhermino, L. (2005). Alteration of reproductive parameters in the crab Carcinus maenas after exposure to metals. Water Air and Soil Pollution, 160(1–4), 245–258.
Esimbekova, E. N., Kratasyuk, V. A., & Torgashina, I. G. (2007). Disk-shaped immobilized multicomponent reagent for bioluminescent analyses: correlation between activity and composition. Enzyme and Microbial Technology, 40, 343–346.
Evtugin, G. A., Budnikov, G. K., & Nikolskaya, E. B. (2002). Biochemical test based on stabilized cholinesterase preparations: new approaches. Journal Analytical Chemistry, 57, 1127–1132.
Galgani, F., Cadiou, Y., & Gilbert, F. (1992). Simultaneous and iterative weighted regression analysis of toxicity tests using a microplate reader. Ecotoxicology and Environmental Safety, 23, 237–243.
Gonzalez-Fernandez, C., Albentosa, M., Campillo, J. A., Vinas, L., Fumega, J., Franco, A., Besada, V., Gonzalez-Quijano, A., & Bellas, J. (2015). Influence of mussel biological variability on pollution biomarkers. Environmental Research, 137, 14–31.
Khristoforova, N.K. (1989). Bioindication and monitoring of pollution of marine waters with heavy metals, ed. “Nayka”, Leningrad, Russia, 192.
Korneeva, G. A., Kharchenko, S. V., & Romankevich, E. A. (1990). Study of enzymatic hydrolysis of casein in sea water. Biology Bulletin of the Academy of Science of the USSR, 6, 821–826.
Korneeva, G. A. (1996). Application of test-system for monitoring of black sea waters. Biology Bulletin, 5, 492–499.
Korneeva, G. A. (2002). Biochemical (enzymatic) indication and testing of ecological state of the hydrosphere. Biology Bulletin, 1, 43–45.
Kozhenkova, E. N., Chernova, E. N., & Shulkin, V. M. (2006). Microelement composition of the green algae Ulva fenestrate from Peter the Great Bay, the Sea of Japan. Russian Journal of Marine Biology, 32(5), 346–352.
Kratasyuk, V. A., Esimbekova, E. N., Gladyshev, M. I., Khromihek, E. B., Kuznetsov, A. M., & Ivanova, E. A. (2001). The use of bioluminescent biotests for study of natural and laboratory aquatic ecosystems. Chemosphere, 42, 909–915.
Lan, W. G., Wong, K. W., Chen, N., & Sin, Y. M. (1995). Effect of combined copper, zinc, chromium and selenium by orthogonal array on alkaline phosphatase activity in liver of the red sea bream, Chrysophrys major. Aquaculture, 3–4, 219–230.
Lera, S., Macchia, S., & Pellegrini, D. (2006). Standardizing the methodology of sperm cell test with Paracentrotus lividus. Environmental Monitoring and Assessment, 122(1–3), 101–109.
McCarthy, S. D. S., Rafferty, S. P., & Frost, P. C. (2010). Responses of alkaline phosphatase activity to phosphorus stress in Daphnia magma. Journal Experimental Biology, 213(2), 256–261.
Menzorova, N. I., & Rasskazov, V. A. (1980). Effect of bivalent metal ions on enzymatic activity of Ca, Mg-dependent DNase from sea urchin Strongylocentrotus intermedius embryos. Russian Journal of Biochemistry, 3, 544–553.
Menzorova, N. I., & Rasskazov, V. A. (1999). Application of the sea urchin Strongylocentrotus intermedius embryo DNase test in the evaluation of marine water pollution by various toxicants. Russian Journal of Marine Biology, 25, 60–64.
Menzorova, N. I., Ivleva, A. D., Sibirtsev, Y. T., & Rasskazov, V. A. (2008). Phosphatases and phosphodiesterases isolated from the red king crab (Paralithodes camtschatica) hepatopancreas. Applied Biochemistry and Microbiology, 44, 93–97.
Menzorova, N. I., & Rasskazov, V. A. (2009). Evaluation of the ecologocal state of the Sea of Japan and the Sea of Okhotsk using the DNase test system. Oceanology, 49, 824–832.
Menzorova, N.I., Seytkalieva, A.V., Rasskazov, V.A. (2010). The method of integral evaluation of pollution of the marine and fresh water. Patent № 2396353 (RU).
Menzorova, N. I., Seytkalieva, A. V., & Rasskazov, V. A. (2014). Enzymatic methods for the determination of pollution in seawater using salt resistant alkaline phosphatase from eggs of the sea urchin Strongylocentrotus intermedius. Marine Pollution Bulletin, 79, 188–195.
Novelli, A. A., Losso, C., Ghetti, P. F., & Ghirardini, A. V. (2003). Toxicity of heavy metals using sperm cell and embryo toxicity bioassay with Paracentrotus lividus (echinodermata: Echindea): comparison with exposure concentrations in the Lagoon of Venice, Italy. Environmental Toxicology and Chemistry, 22(6), 1295–1301.
Olsen, R. L., Overbo, K., & Myrnes, B. (1991). Alkaline phosphatases from the hepatopancreas of shrimp (Pandalus borealis): a dimeric enzyme with catalytically active subunits. Comparative Biochemistry and Physiology, 99B(4), 755–761.
Parng, C., Seng, W. L., Semino, C., & McGrath, P. (2002). Zebrafish: a preclinical model for drug screening. Assay and Rug Development Technologies, 1(1), 41–48.
Pinsino, A., Mantranga, V., Trinchella, F., & Roccheri, M. C. (2010). Sea urchin embryos as an in vivo model for the assessment of manganese toxicity: developmental and stress response effects. Ecotoxicology, 19, 555–562.
Ragunathan, M. G., Deecaranan, M., & Jadhav, S. (1998). Effects of reserpine and chlorpromazine on the phosphatases activity of female fresh water crab, Paratelphusa hydrodromous (Herbst). Journal of Environmental Biology, 19(3), 231–236.
Shekhovtsova, T. N., Muginova, S. V., & Veselova, I. A. (2007). Enzymatic methods of analysis: novel approaches and applications. Russian Chemical Bulletin International Edition, 56, 606–620.
Shulkin, V.M. (2004). Metals in marine ecosystems of shallow. Ed. “Dal’nauka”, Vladivostok, Russia.
Suresh, P. G., Reju, M. K., & Mohandas, A. (1993). Hemolymph phosphatase activity levels in two fresh-water gastropods exposed to copper. Science of the Total Environment, 134(2), 1265–1277.
Tsvetkov, I. L., Zarubin, S. L., Urvantseva, G. A., Konichev, A. S., & Filippovich, Y. B. (1997). Acid phosphatase of hydrobionts as an enzymatic marker of biochemical adaptation to the action of toxicants. Biology Bulletin, 5, 539–545.
Tsvetkov, I. L., Popov, A. P., & Konichev, A. S. (2003). Acid phosphatase complex from the freshwater snail Viviparus viviparus L. under standard conditions and intoxication by cadmium ions. Biochemistry (Moscow), 68(12), 1327–1334.
Tsvetkov, I. L., Tsvetkova, M. A., Zarubin, S. L., Semernoi, V. P., & Konichev, A. S. (2006). Quality assessment of wastewater and natural waters by using a biochemical index—the activity of acid phosphatase in freshwater mollusks. Water Resources, 33(1), 56–63.
Van Dyk, S. J., & Pletschke, B. (2011). Review on the use of enzymes for the detection of organochlorine, organophosphate and carbamate pesticides in the environment. Chemosphere, 82, 291–307.
Vaschenko, M. A., & Naidenko, T. K. (1989). Effect of long-term exposure of sea urchins to hydrocarbons on development of their larvae. Russian Journal of Marine Biology, 1, 59–65.
Vaschenko, M. A. (2000). Pollution in Peter the Great Bay, Sea of Japan, and its biological consequences. Russian Journal of Marine Biology, 26, 155–166.
Vidal-Linan, L., & Bellas, J. (2013). Practical procedures for selected biomarkers in mussels, Mytilus galloprovincialis—implications for marine pollution monitoring. Science of the Total Environment, 461, 56–64.
Vijayavel, K., Anduselvam, C., Balasubramanian, M. P., Deepak, S. V., & Gopalakrishnan, S. (2006). Assessment of biochemical components and enzyme activities in the estuarine crab Scylla tranquebarica from naphthalene contaminated habitants. Ecotoxicology, 15, 469–476.
Yurovitzky, Y. G., & Sidorov, V. S. (1993). Ecologobiochemical monitoring and ecologobiochemical testing in regions of ecological trouble. Biology Bulletin, 1, 74–82.
Zhavoronkova, A. M., Muginova, S. V., Veselova, I. A., & Shekhovtsova, T. N. (2003). Determination of zinc (II) using native and immobilized alkaline phosphatases of different origin. Journal Analytical Chemistry, 58, 88–96.
Zhou, Q., Zhang, J., Fu, J., Shi, J., & Jiang, G. (2008). Biomonitoring: an appealing tool for assessment of metal pollution in the aquatic ecosystem. Analitica Chimica Acta, 606, 135–150.
Zhuravel, E. V., Markina, Z. V., Khristoforova, N. K., & Aizdaicer, N. A. (2006). Biotesting of water quality in Peter the Great Bay with the use of the microalga Dunaliella salina and embryos and larvae of the sea urchin Scaphechinus mirabilis. Russian Journal of Marine Biology, 32, 157–165.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seitkalieva, A.V., Menzorova, N.I. & Rasskazov, V.A. Application of different enzyme assays and biomarkers for pollution monitoring of the marine environment. Environ Monit Assess 188, 70 (2016). https://doi.org/10.1007/s10661-015-5066-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-015-5066-5