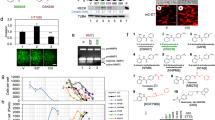
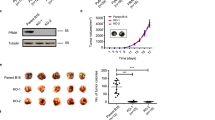
Abstract
Metastasis is the major cause of morbidity and mortality in cancer patients. An understanding of the genes that regulate metastasis and development of therapies to target these genes is needed urgently. Since members of the LIM kinase (LIMK) family are key regulators of the actin cytoskeleton and are involved in cell motility and invasion, LIMK is considered to be a good therapeutic target for metastatic disease. Here we investigated the consequences of LIMK inhibition on growth and metastasis of human and mouse mammary tumors. LIMK activity was reduced in tumor cells by expression of dominant-negative LIMK1, by RNA interference or with a selective LIMK inhibitor. The extent of phosphorylation of the LIMK substrate, cofilin, of proliferation and invasion in 2D and 3D culture and of tumor growth and metastasis in mice were assessed. Inhibition of LIMK activity efficiently reduced the pro-invasive properties of tumor cells in vitro. Tumors expressing dominant-negative LIMK1 grew more slowly and were less metastatic in mice. However, systemic administration of a LIMK inhibitor did not reduce either primary tumor growth or spontaneous metastasis. Surprisingly, metastasis to the liver was increased after administration of the inhibitor. These data raise a concern about the use of systemic LIMK inhibitors for the treatment of metastatic breast cancer.






Similar content being viewed by others
Abbreviations
- LIMK:
-
LIM kinase
- ADF:
-
Actin depolymerizing factor
- F-actin:
-
Filamentous action
- SSH:
-
Slingshot
- siRNA:
-
Small interfering RNA
References
Olson MF, Sahai E (2009) The actin cytoskeleton in cancer cell motility. Clin Exp Metastasis 26:273–287
Pollard TD, Borisy GG (2003) Cellular motility driven by assembly and disassembly of actin filaments. Cell 112:453–465
Wang W, Eddy R, Condeelis J (2007) The cofilin pathway in breast cancer invasion and metastasis. Nat Rev Cancer 7:429–440
Sahai E (2007) Illuminating the metastatic process. Nat Rev Cancer 7:737–749
Bamburg JR (1999) Proteins of the ADF/cofilin family: essential regulators of actin dynamics. Annu Rev Cell Dev Biol 15:185–230
Bamburg JR, Bernstein BW (2008) ADF/cofilin. Curr Biol 18:R273–R275
Yamaguchi H, Condeelis J (2007) Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochim Biophys Acta 1773:642–652
Arber S, Barbayannis FA, Hanser H et al (1998) Regulation of actin dynamics through phosphorylation of cofilin by LIM-kinase. Nature 393:805–809
Yang N, Higuchi O, Ohashi K et al (1998) Cofilin phosphorylation by LIM-kinase1 and its role in Rac-mediated actin reorganization. Nature 393:809–812
Sumi T, Matsumoto K, Takai Y et al (1999) Cofilin phosphorylation and actin cytoskeletal dynamics regulated by rho- and Cdc42-activated LIM-kinase 2. J Cell Biol 147:1519–1532
Bernard O (2007) LIM kinases, regulators of actin dynamics. Int J Biochem Cell Biol 39:1071–1076
Niwa R, Nagata-Ohashi K, Takeichi M et al (2002) Control of actin reorganization by Slingshot, a family of phosphatases that dephosphorylate ADF/cofilin. Cell 108:233–246
Gohla A, Birkenfeld J, Bokoch GM (2005) Chronophin, a novel HAD-type serine protein phosphatase, regulates cofilin-dependent actin dynamics. Nat Cell Biol 7:21–29
Maekawa M, Ishizaki T, Boku S et al (1999) Signaling from Rho to the actin cytoskeleton through protein kinases ROCK and LIM-kinase. Science 285:895–898
Edwards DC, Sanders LC, Bokoch GM et al (1999) Activation of LIM-kinase by Pak1 couples Rac/Cdc42 GTPase signalling to actin cytoskeletal dynamics. Nat Cell Biol 1:253–259
Dan C, Kelly A, Bernard O et al (2001) Cytoskeletal changes regulated by the PAK4 serine/threonine kinase are mediated by LIM kinase 1 and cofilin. J Biol Chem 276:32115–32121
Ohashi K, Nagata K, Maekawa M et al (2000) Rho-associated kinase ROCK activates LIM-kinase 1 by phosphorylation at threonine 508 within the activation loop. J Biol Chem 275:3577–3582
Sumi T, Matsumoto K, Nakamura T (2001) Specific activation of LIM kinase 2 via phosphorylation of threonine 505 by ROCK, a Rho-dependent protein kinase. J Biol Chem 276:670–676
Ikebe C, Ohashi K, Fujimori T et al (1997) Mouse LIM-kinase 2 gene: cDNA cloning, genomic organization, and tissue-specific expression of two alternatively initiated transcripts. Genomics 46:504–508
Nunoue K, Ohashi K, Okano I et al (1995) LIMK-1 and LIMK-2, two members of a LIM motif-containing protein kinase family. Oncogene 11:701–710
Acevedo K, Moussi N, Li R et al (2006) LIM kinase 2 is widely expressed in all tissues. J Histochem Cytochem 54:487–501
Davila M, Frost AR, Grizzle WE et al (2003) LIM kinase 1 is essential for the invasive growth of prostate epithelial cells: implications in prostate cancer. J Biol Chem 278:36868–36875
Saxena M, Singh S, Negi MP et al (2010) Expression profiling of G2/M phase regulatory proteins in normal, premalignant and malignant uterine cervix and their correlation with survival of patients. J Cancer Res Ther 6:167–171
Yoshioka K, Foletta V, Bernard O et al (2003) A role for LIM kinase in cancer invasion. Proc Natl Acad Sci USA 100:7247–7252
Bagheri-Yarmand R, Mazumdar A, Sahin AA et al (2006) LIM kinase 1 increases tumor metastasis of human breast cancer cells via regulation of the urokinase-type plasminogen activator system. Int J Cancer 118:2703–2710
Scott RW, Hooper S, Crighton D et al (2010) LIM kinases are required for invasive path generation by tumor and tumor-associated stromal cells. J Cell Biol 191:169–185
Horita Y, Ohashi K, Mukai M et al (2008) Suppression of the invasive capacity of rat ascites hepatoma cells by knockdown of Slingshot or LIM kinase. J Biol Chem 283:6013–6021
Zebda N, Bernard O, Bailly M et al (2000) Phosphorylation of ADF/cofilin abolishes EGF-induced actin nucleation at the leading edge and subsequent lamellipod extension. J Cell Biol 151:1119–1128
Wang W, Mouneimne G, Sidani M et al (2006) The activity status of cofilin is directly related to invasion, intravasation, and metastasis of mammary tumors. J Cell Biol 173:395–404
Ross-Macdonald P, de Silva H, Guo Q et al (2008) Identification of a nonkinase target mediating cytotoxicity of novel kinase inhibitors. Mol Cancer Ther 7:3490–3498
Foletta VC, Moussi N, Sarmiere PD et al (2004) LIM kinase 1, a key regulator of actin dynamics, is widely expressed in embryonic and adult tissues. Exp Cell Res 294:392–405
Lelekakis M, Moseley JM, Martin TJ et al (1999) A novel orthotopic model of breast cancer metastasis to bone. Clin Exp Metastasis 17:163–170
Barkan D, Kleinman H, Simmons JL et al (2008) Inhibition of metastatic outgrowth from single dormant tumor cells by targeting the cytoskeleton. Cancer Res 68:6241–6250
Lyons AB (2000) Analysing cell division in vivo and in vitro using flow cytometric measurement of CFSE dye dilution. J Immunol Methods 243:147–154
Eckhardt BL, Parker BS, van Laar RK et al (2005) Genomic analysis of a spontaneous model of breast cancer metastasis to bone reveals a role for the extracellular matrix. Mol Cancer Res 3:1–13
Shaw KR, Wrobel CN, Brugge JS (2004) Use of three-dimensional basement membrane cultures to model oncogene-induced changes in mammary epithelial morphogenesis. J Mammary Gland Biol Neoplasia 9:297–310
Korpal M, Ell BJ, Buffa FM et al (2011) Direct targeting of Sec23a by miR-200s influences cancer cell secretome and promotes metastatic colonization. Nat Med 17:1101–1108
Harrison BA, Whitlock NA, Voronkov MV et al (2009) Novel class of LIM-kinase 2 inhibitors for the treatment of ocular hypertension and associated glaucoma. J Med Chem 52:6515–6518
Amano T, Kaji N, Ohashi K et al (2002) Mitosis-specific activation of LIM motif-containing protein kinase and roles of cofilin phosphorylation and dephosphorylation in mitosis. J Biol Chem 277:22093–22102
Kaji N, Muramoto A, Mizuno K (2008) LIM kinase-mediated cofilin phosphorylation during mitosis is required for precise spindle positioning. J Biol Chem 283:4983–4992
Davila M, Jhala D, Ghosh D et al (2007) Expression of LIM kinase 1 is associated with reversible G1/S phase arrest, chromosomal instability and prostate cancer. Mol Cancer 6:40
Croft DR, Olson MF (2006) The Rho GTPase effector ROCK regulates cyclin A, cyclin D1, and p27Kip1 levels by distinct mechanisms. Mol Cell Biol 26:4612–4627
Oser M, Condeelis J (2009) The cofilin activity cycle in lamellipodia and invadopodia. J Cell Biochem 108:1252–1262
Prudent R, Vassal-Stermann E, Nguyen CH et al (2012) Pharmacological inhibition of LIM kinase stabilizes microtubules and inhibits neoplastic growth. Cancer Res 72(17):4429–4439
Acknowledgments
We thank Christina Restall for technical assistance and advice, Dr Bala Murthy for advice on FACS analysis, Dr. Roger Tsien for provision of the cherry fluorescent protein vector, Alison Gregg and Julia Morizzi for analysis of BMS3 levels in mice, Dr. Siddhartha Deb for pathology advice and Dr. Duncan Campbell for assistance with the statistical analysis. This work was supported by grants from the NIH (R21CA098229) and from the NHMRC of Australia, Fellowship support from NHMRC (OB) and from NBCF (Australia) (RLA). The authors acknowledge financial support from the Cancer Therapeutics CRC, established and supported under the Australian Government’s Cooperative Research Centre Program.
Conflict of interest
No potential conflicts of interest were disclosed.
Author information
Authors and Affiliations
Corresponding authors
Additional information
R. Li and J. Doherty contributed equally to this study. R.L. Anderson and O. Bernard are equal senior authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, R., Doherty, J., Antonipillai, J. et al. LIM kinase inhibition reduces breast cancer growth and invasiveness but systemic inhibition does not reduce metastasis in mice. Clin Exp Metastasis 30, 483–495 (2013). https://doi.org/10.1007/s10585-012-9553-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-012-9553-6