Abstract
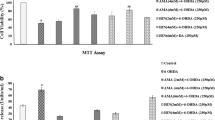
6-Hydroxydopamine (6-OHDA), a metabolite of dopamine is known to induce dopaminergic cell toxicity which makes that a suitable agent inducing an experimental model of Parkinson’s disease (PD). Agmatine has been shown to protect against some cellular and animal PD models. This study was aimed to assess whether agmatine prevents 6-OHDA-induced SH-SY5Y cell death and if yes, then how it affects Akt/glycogen synthesis kinase-3β (GSK-3β) and extracellular signal-regulated kinases (ERK) signals. The cells were treated with different drugs, and their viability was examined via MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide) assay and morphological observation. Western blot studies were done to assess cleaved caspase-3, Akt/GSK-3β, and ERK proteins. 6-OHDA-induced cell death and caspase-3 cleavage, while agmatine prevented those changes. 6-OHDA also decreased the amount of phosphorylated Akt (pAkt)/Akt while increased GSK-3β activity which was prevented by agmatine. Additionally, this toxin increased pERK/ERK ratio which was averted again by agmatine. The PI3/Akt inhibitor, LY294002, impeded the changes induced by agmatine, while ERK inhibitor (PD98059) did not disturb the effects of agmatine, and by itself, it preserved the cells against 6-OHDA toxicity. This study revealed that agmatine is protective in 6-OHDA model of PD and affects Akt/GSK-3β and ERK pathways.






Similar content being viewed by others
Change history
26 July 2018
The original version of this article unfortunately contained a mistake in the unit of agmatine doses. The agmatine doses were erroneously written in nanomolar in the published article. The correct effective doses of agmatine were 150 and 250 µM.
References
Andrew R, Watson DG, Best SA, Midgley JM, Wenlong H, Petty RK (1993) The determination of hydroxydopamines and other trace amines in the urine of parkinsonian patients and normal controls. Neurochem Res 18(11):1175–1177
Annunziato L, Amoroso S, Pannaccione A, Cataldi M, Pignataro G, D’Alessio A, Sirabella R, Secondo A, Sibaud L, Di Renzo GF (2003) Apoptosis induced in neuronal cells by oxidative stress: role played by caspases and intracellular calcium ions. Toxicol Lett 139(2–3):125–133
Arndt MA, Battaglia V, Parisi E, Lortie MJ, Isome M, Baskerville C, Pizzo DP, Ientile R, Colombatto S, Toninello A, Satriano J (2009) The arginine metabolite agmatine protects mitochondrial function and confers resistance to cellular apoptosis. Am J Physiol Cell Physiol 296(6):C1411–1419. doi:10.1152/ajpcell.00529.2008
Battaglia V, Rossi CA, Colombatto S, Grillo MA, Toninello A (2007) Different behavior of agmatine in liver mitochondria: inducer of oxidative stress or scavenger of reactive oxygen species? Biochim Biophys Acta 1768(5):1147–1153. doi:10.1016/j.bbamem.2007.01.011
Battaglia V, Grancara S, Satriano J, Saccoccio S, Agostinelli E, Toninello A (2010) Agmatine prevents the Ca(2+)-dependent induction of permeability transition in rat brain mitochondria. Amino Acids 38(2):431–437. doi:10.1007/s00726-009-0402-0
Blum D, Torch S, Nissou MF, Benabid AL, Verna JM (2000) Extracellular toxicity of 6-hydroxydopamine on PC12 cells. Neurosci Lett 283(3):193–196
Bogoyevitch MA, Court NW (2004) Counting on mitogen-activated protein kinases–ERKs 3, 4, 5, 6, 7 and 8. Cell Signal 16(12):1345–1354. doi:10.1016/j.cellsig.2004.05.004
Bove J, Prou D, Perier C, Przedborski S (2005) Toxin-induced models of Parkinson’s disease. NeuroRx 2(3):484–494. doi:10.1602/neurorx.2.3.484
Byczkowski JZ, Zychlinski L, Porter CW (1982) Inhibition of the bioenergetic functions of isolated rat liver mitochondria by polyamines. Biochem Pharmacol 31(24):4045–4053
Chaffee RR, Arine RM, Rochelle RH (1979) The possible role of intracellular polyamines in mitochondrial metabolic regulation. Biochem Biophys Res Commun 86(2):293–299
Chen G, Bower KA, Ma C, Fang S, Thiele CJ, Luo J (2004) Glycogen synthase kinase 3beta (GSK3beta) mediates 6-hydroxydopamine-induced neuronal death. FASEB J 18(10):1162–1164. doi:10.1096/fj.04-1551fje
Cheung YT, Lau WK, Yu MS, Lai CS, Yeung SC, So KF, Chang RC (2009) Effects of all-trans-retinoic acid on human SH-SY5Y neuroblastoma as in vitro model in neurotoxicity research. Neurotoxicology 30(1):127–135. doi:10.1016/j.neuro.2008.11.001
Chu CT, Levinthal DJ, Kulich SM, Chalovich EM, DeFranco DB (2004) Oxidative neuronal injury. The dark side of ERK1/2. Eur J Biochem/FEBS 271(11):2060–2066. doi:10.1111/j.1432-1033.2004.04132.x
Condello S, Curro M, Ferlazzo N, Caccamo D, Satriano J, Ientile R (2011) Agmatine effects on mitochondrial membrane potential and NF-kappaB activation protect against rotenone-induced cell damage in human neuronal-like SH-SY5Y cells. J Neurochem 116(1):67–75. doi:10.1111/j.1471-4159.2010.07085.x
Condello S, Calabro E, Caccamo D, Curro M, Ferlazzo N, Satriano J, Magazu S, Ientile R (2012) Protective effects of agmatine in rotenone-induced damage of human SH-SY5Y neuroblastoma cells: fourier transform infrared spectroscopy analysis in a model of Parkinson’s disease. Amino Acids 42(2–3):775–781. doi:10.1007/s00726-011-0994-z
Embi N, Rylatt DB, Cohen P (1980) Glycogen synthase kinase-3 from rabbit skeletal muscle. Separation from cyclic-AMP-dependent protein kinase and phosphorylase kinase. Eur J Biochem/FEBS 107(2):519–527
Feng CQ, Ma WL, Song YB, Guo QY, Wu QH, Zheng WL (2002) Detection of cell apoptosis by MTT assay. Acad J First Med Coll PLA 22(3):262–263
Forno LS (1996) Neuropathology of Parkinson’s disease. J Neuropathol Exp Neurol 55(3):259–272
Freitas AE, Egea J, Buendia I, Navarro E, Rada P, Cuadrado A, Rodrigues AL, Lopez MG (2014) Agmatine induces Nrf2 and protects against corticosterone effects in hippocampal neuronal cell line. Mol Neurobiol. doi:10.1007/s12035-014-8827-1
Gilad GM, Gilad VH, Finberg JP, Rabey JM (2005) Neurochemical evidence for agmatine modulation of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) neurotoxicity. Neurochem Res 30(6–7):713–719. doi:10.1007/s11064-005-6865-9
Graham JD, Lewis MJ, Williams J (1974) Proceedings: the effect of delta-1-tetrahydrocannabinol on the noradrenaline and dopamine content of the brain and heart of the rat. Br J Pharmacol 52(3):446P
Greene LA, Levy O, Malagelada C (2011) Akt as a victim, villain and potential hero in Parkinson’s disease pathophysiology and treatment. Cell Mol Neurobiol 31(7):969–978. doi:10.1007/s10571-011-9671-8
Gu M, Cooper JM, Taanman JW, Schapira AH (1998) Mitochondrial DNA transmission of the mitochondrial defect in Parkinson’s disease. Ann Neurol 44(2):177–186. doi:10.1002/ana.410440207
Hanrott K, Gudmunsen L, O’Neill MJ, Wonnacott S (2006) 6-Hydroxydopamine-induced apoptosis is mediated via extracellular auto-oxidation and caspase 3-dependent activation of protein kinase Cdelta. J Biol Chem 281(9):5373–5382. doi:10.1074/jbc.M511560200
Hwang O (2013) Role of oxidative stress in Parkinson’s disease. Exp Neurobiol 22(1):11–17. doi:10.5607/en.2013.22.1.11
Isome M, Lortie MJ, Murakami Y, Parisi E, Matsufuji S, Satriano J (2007) The antiproliferative effects of agmatine correlate with the rate of cellular proliferation. Am J Physiol Cell Physiol 293(2):C705–711. doi:10.1152/ajpcell.00084.2007
Jung HJ, Jeon YH, Bokara KK, Koo BN, Lee WT, Park KA, Lee JE (2013) Agmatine promotes the migration of murine brain endothelial cells via multiple signaling pathways. Life Sci 92(1):42–50. doi:10.1016/j.lfs.2012.10.018
Kulich SM, Chu CT (2001) Sustained extracellular signal-regulated kinase activation by 6-hydroxydopamine: implications for Parkinson’s disease. J Neurochem 77(4):1058–1066
Lee GT, Ha H, Lee HC, Cho YD (2003) Agmatine reduces hydrogen peroxide in mesangial cells under high glucose conditions. J Biochem Mol Biol 36(3):251–257
Lee ER, Kim JY, Kang YJ, Ahn JY, Kim JH, Kim BW, Choi HY, Jeong MY, Cho SG (2006) Interplay between PI3 K/Akt and MAPK signaling pathways in DNA-damaging drug-induced apoptosis. Biochim Biophys Acta 1763(9):958–968. doi:10.1016/j.bbamcr.2006.06.006
Lees AJ, Hardy J, Revesz T (2009) Parkinson’s disease. Lancet 373(9680):2055–2066. doi:10.1016/s0140-6736(09)60492-x
Ljungdahl A, Hokfelt T, Jonsson G, Sachs C (1971) Autoradiographic demonstration of uptake and accumulation of 3H-6-hydroxydopamine in adrenergic nerves. Experientia 27(3):297–299
Matheus FC, Aguiar AS Jr, Castro AA, Villarinho JG, Ferreira J, Figueiredo CP, Walz R, Santos AR, Tasca CI, Prediger RD (2012) Neuroprotective effects of agmatine in mice infused with a single intranasal administration of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Behav Brain Res 235(2):263–272. doi:10.1016/j.bbr.2012.08.017
Moosavi M, Khales GY, Abbasi L, Zarifkar A, Rastegar K (2012) Agmatine protects against scopolamine-induced water maze performance impairment and hippocampal ERK and Akt inactivation. Neuropharmacology 62(5–6):2018–2023. doi:10.1016/j.neuropharm.2011.12.031
Moosavi M, Zarifkar AH, Farbood Y, Dianat M, Sarkaki A, Ghasemi R (2014) Agmatine protects against intracerebroventricular streptozotocin-induced water maze memory deficit, hippocampal apoptosis and Akt/GSK3beta signaling disruption. Eur J Pharmacol 736:107–114. doi:10.1016/j.ejphar.2014.03.041
Nissim I, Horyn O, Nissim I, Daikhin Y, Wehrli SL, Yudkoff M (2008) 3-isobutylmethylxanthine inhibits hepatic urea synthesis: protection by agmatine. J Biol Chem 283(22):15063–15071. doi:10.1074/jbc.M800163200
Olmos G, DeGregorio-Rocasolano N, Paz Regalado M, Gasull T, Assumpcio Boronat M, Trullas R, Villarroel A, Lerma J, Garcia-Sevilla JA (1999) Protection by imidazol(ine) drugs and agmatine of glutamate-induced neurotoxicity in cultured cerebellar granule cells through blockade of NMDA receptor. Br J Pharmacol 127(6):1317–1326. doi:10.1038/sj.bjp.0702679
Porter AG, Janicke RU (1999) Emerging roles of caspase-3 in apoptosis. Cell Death Differ 6(2):99–104. doi:10.1038/sj.cdd.4400476
Ramachandiran S, Huang Q, Dong J, Lau SS, Monks TJ (2002) Mitogen-activated protein kinases contribute to reactive oxygen species-induced cell death in renal proximal tubule epithelial cells. Chem Res Toxicol 15(12):1635–1642
Reis DJ, Regunathan S (2000) Is agmatine a novel neurotransmitter in brain? Trends Pharmacol Sci 21(5):187–193
Ren Y, Jiang H, Yang F, Nakaso K, Feng J (2009) Parkin protects dopaminergic neurons against microtubule-depolymerizing toxins by attenuating microtubule-associated protein kinase activation. J Biol Chem 284(6):4009–4017. doi:10.1074/jbc.M806245200
Roskoski R Jr (2012) ERK1/2 MAP kinases: structure, function, and regulation. Pharmacol Res 66(2):105–143. doi:10.1016/j.phrs.2012.04.005
Santhanam AV, Viswanathan S, Dikshit M (2007) Activation of protein kinase B/Akt and endothelial nitric oxide synthase mediates agmatine-induced endothelium-dependent relaxation. Eur J Pharmacol 572(2–3):189–196. doi:10.1016/j.ejphar.2007.06.031
Satriano J, Isome M, Casero RA Jr, Thomson SC, Blantz RC (2001) Polyamine transport system mediates agmatine transport in mammalian cells. Am J Physiol Cell Physiol 281(1):C329–334
Schule B, Pera RA, Langston JW (2009) Can cellular models revolutionize drug discovery in Parkinson’s disease? Biochim Biophys Acta 1792(11):1043–1051. doi:10.1016/j.bbadis.2009.08.014
Song JX, Shaw PC, Wong NS, Sze CW, Yao XS, Tang CW, Tong Y, Zhang YB (2012) Chrysotoxine, a novel bibenzyl compound selectively antagonizes MPP(+), but not rotenone, neurotoxicity in dopaminergic SH-SY5Y cells. Neurosci Lett 521(1):76–81. doi:10.1016/j.neulet.2012.05.063
Stambolic V, Woodgett JR (1994) Mitogen inactivation of glycogen synthase kinase-3 beta in intact cells via serine 9 phosphorylation. Biochem J 303(Pt 3):701–704
Subramaniam S, Unsicker K (2010) ERK and cell death: ERK1/2 in neuronal death. FEBS J 277(1):22–29
Tian LL, Zhou Z, Zhang Q, Sun YN, Li CR, Cheng CH, Zhong ZY, Wang SQ (2007) Protective effect of (±) isoborneol against 6-OHDA-induced apoptosis in SH-SY5Y cells. Cell Phys Biochem 20(6):1019–1032. doi:10.1159/0000110682
Tieu K, Zuo DM, Yu PH (1999) Differential effects of staurosporine and retinoic acid on the vulnerability of the SH-SY5Y neuroblastoma cells: involvement of bcl-2 and p53 proteins. J Neurosci Res 58(3):426–435
Troussard AA, Tan C, Yoganathan TN, Dedhar S (1999) Cell-extracellular matrix interactions stimulate the AP-1 transcription factor in an integrin-linked kinase- and glycogen synthase kinase 3-dependent manner. Mol Cell Biol 19(11):7420–7427
Turenne GA, Price BD (2001) Glycogen synthase kinase3 beta phosphorylates serine 33 of p53 and activates p53′s transcriptional activity. BMC cell Biol 2:12
Wang X, Martindale JL, Holbrook NJ (2000) Requirement for ERK activation in cisplatin-induced apoptosis. J Biol Chem 275(50):39435–39443. doi:10.1074/jbc.M004583200
Wang WP, Iyo AH, Miguel-Hidalgo J, Regunathan S, Zhu MY (2006) Agmatine protects against cell damage induced by NMDA and glutamate in cultured hippocampal neurons. Brain Res 1:210–216. doi:10.1016/j.brainres.2006.02.024
Wu N, Su RB, Li J (2008) Agmatine and imidazoline receptors: their role in opioid analgesia, tolerance and dependence. Cell Mol Neurobiol 28(5):629–641. doi:10.1007/s10571-007-9164-y
Xing C, Peng Y, Chang R, Yin Y, Xie Z (2005) Effects of insulin-like growth factor-1 on okadaic acid-induced apoptosis in SH-SY5Y cells. Cell Biol Int 29(9):803–808. doi:10.1016/j.cellbi.2005.04.012
Yamanaka H, Hayashi Y, Iwasaki M, Kamibayashi T, Yamatodani A, Mashimo T (2010) Activation of phosphatidylinositol 3-kinase/Akt signaling pathway and endogenous nitric oxide are needed for the antiarrhythmic effect of centrally administered rilmenidine. Eur J Pharmacol 647(1–3):155–160. doi:10.1016/j.ejphar.2010.08.035
Zhu JH, Kulich SM, Oury TD, Chu CT (2002) Cytoplasmic aggregates of phosphorylated extracellular signal-regulated protein kinases in Lewy body diseases. Am J Pathol 161(6):2087–2098. doi:10.1016/s0002-9440(10)64487-2
Zhuang S, Schnellmann RG (2006) A death-promoting role for extracellular signal-regulated kinase. J Pharmacol Exp Ther 319(3):991–997. doi:10.1124/jpet.106.107367
Zhuang S, Yan Y, Daubert RA, Han J, Schnellmann RG (2007) ERK promotes hydrogen peroxide-induced apoptosis through caspase-3 activation and inhibition of Akt in renal epithelial cells. Am J Physiol Renal Physiol 292(1):F440–447. doi:10.1152/ajprenal.00170.2006
Acknowledgments
This work was derived from the thesis of Esmat Amiri and supported by a Grant (No. 92-6735) from Shiraz University of Medical Sciences, Shiraz, Iran. The authors would like to appreciate Ms. Mehrnaz Gholami for her help in editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Amiri, E., Ghasemi, R. & Moosavi, M. Agmatine Protects Against 6-OHDA-Induced Apoptosis, and ERK and Akt/GSK Disruption in SH-SY5Y Cells. Cell Mol Neurobiol 36, 829–838 (2016). https://doi.org/10.1007/s10571-015-0266-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-015-0266-7