Abstract
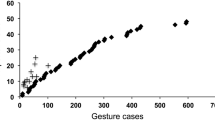
Great ape gestural communication is known to be intentional, elaborate and flexible; yet there is controversy over the best interpretation of the system and how gestures are acquired, perhaps because most studies have been made in restricted, captive settings. Here, we report the first systematic analysis of gesture in a population of wild chimpanzees. Over 266 days of observation, we recorded 4,397 cases of intentional gesture use in the Sonso community, Budongo, Uganda. We describe 66 distinct gesture types: this estimate appears close to asymptote, and the Sonso repertoire includes most gestures described informally at other sites. Differences in repertoire were noted between individuals and age classes, but in both cases, the measured repertoire size was predicted by the time subjects were observed gesturing. No idiosyncratic usages were found, i.e. no gesture type was used only by one individual. No support was found for the idea that gestures are acquired by ‘ontogenetic ritualization’ from originally effective actions; moreover, in detailed analyses of two gestures, action elements composing the gestures did not closely match those of the presumed original actions. Rather, chimpanzee gestures are species-typical; indeed, many are ‘family-typical’, because gesture types recorded in gorillas, orangutans and chimpanzee overlap extensively, with 24 gestures recorded in all three genera. Nevertheless, chimpanzee gestures are used flexibly across a range of contexts and show clear adjustment to audience (e.g. silent gestures for attentive targets, contact gestures for inattentive ones). Such highly intentional use of a species-typical repertoire raises intriguing questions for the evolution of advanced communication.






Similar content being viewed by others
References
Altmann J (1974) Observational study of behaviour: sampling methods. Behaviour 49:227–265
Armstrong DF, Wilcox S (2007) The gestural origin of language. Oxford University Press, Oxford
Arnold K, Zuberbuhler K (2006) Semantic combinations in primate calls. Nature 441:303
Arnold K, Zuberbuhler K (2008) Meaningful call combinations in a non-human primate. Curr Biol 18:R202–R203
Call J, Tomasello M (2007a) The gestural communication of apes and monkeys. Lawrence Erlbaum Associates, Hillsdale
Call J, Tomasello M (2007b) The gestural repertoire of chimpanzees (Pan troglodytes). In: Call J, Tomasello M (eds) The gestural communication of apes and monkeys. Lawrence Erlbaum Associates, Mahwah, pp 17–39
Cartmill EA, Byrne RW (2007) Orangutans modify their gestural signalling according to their audience’s comprehension. Curr Biol 17:1345–1348
Cartmill EA, Byrne RW (2010) Semantics of primate gestures: intentional meanings of orangutan gestures. Anim Cogn 13:793–804
Cheney DL, Seyfarth RM (1985) Vervet monkey alarm calls: manipulation through shared information? Behaviour 94:150–166
Cheney DL, Seyfarth RM (1990a) Attending to behaviour versus attending to knowledge: examining monkeys’ attribution of mental states. Anim Behav 40:742–753
Cheney DL, Seyfarth RM (1990b) How monkeys see the world: inside the mind of another species. University of Chicago Press, Chicago
Cheney DL, Seyfarth RM (1996) Function and intention in the calls of non-human primates. Proc Br Acad 88:59–76
Corballis MC (2010) The gestural origins of language. Wiley Interdiscip Rev Cogn Sci 1:2–7
Eggeling WJ (1947) Observations on the ecology of the Budongo Rain Forest, Uganda. J Ecol 34:20–87
Fouts RS, Fouts DH, Van Cantford TE (1989) The infant Loulis learns signs from cross-fostered chimpanzees. In: Gardner RA, Gardner BT, Van Cantford TE (eds) Teaching sign language to chimpanzees. State University of New York Press, New York, pp 280–292
Gardner RA, Gardner BT (1969) Teaching sign language to a chimpanzee. Science 165:664–672
Gardner RA, Gardner BT, Van Cantfort TE (1989) Teaching sign language to chimpanzees. SUNY Press, New York
Genty E, Byrne RW (2010) Why do gorillas make sequences of gestures? Anim Cogn 13:287–301
Genty E, Breuer T, Hobaiter C, Byrne RW (2009) Gestural communication of the gorilla (Gorilla gorilla): repertoire, intentionality and possible origins. Anim Cogn 12:527–546
Goodall J (1968) The behaviour of free-living chimpanzees of the Gombe Stream Reserve. Anim Behav Monogr 1:161–311
Goodall J (1986) The chimpanzees of Gombe: patterns of behavior. Harvard University Press, Cambridge
Hauser MD, Chomsky N, Fitch WT (2002) The faculty of language: what is it, who has it, and how did it evolve? Science 298:1569–1579
Hewes GW (1973) Primate communication and the gestural origins of language. Curr Anthropol 14:5–24
Hobaiter C, Byrne RW (2010) Able-bodied wild chimpanzees imitate a motor procedure used by a disabled individual to overcome handicap. PLoS One 5:e11959
Janik VM, Slater PJB (1997) Vocal learning in mammals. Adv Study Behav 26:59–99
Janik VM, Slater PJB (2003) Traditions in mammalian and avian vocal communication. In: Fragaszy DM, Perry S (eds) The biology of traditions: models and evidence. Cambridge University Press, Cambridge, pp 213–235
Laporte MNC, Zuberbuhler K (2010) Vocal greeting behaviour in wild chimpanzee females. Anim Behav 80:467–473
Leavens DA, Hopkins WD (1998) Intentional communication by chimpanzees: a cross-sectional study of the use of referential gestures. Dev Psychol 34:813–822
Leavens DA, Russell JL, Hopkins WD (2005) Intentionality as measured in the persistence and elaboration of communication by chimpanzees (Pan troglodytes). Child Dev 76:291–306
Liebal K, Call J, Tomasello M (2004a) The use of gesture sequences in chimpanzees. Am J Primatol 64:377–396
Liebal K, Pika S, Call J, Tomasello M (2004b) To move or not to move; how apes adjust to the attentional state of others. Interact Stud 5:199–219
Liebal K, Pika S, Tomasello M (2006) Gestural communication of orangutans (Pongo pygmaeus). Gesture 6:1–38
Miles HL (1986) Cognitive development in a signing orangutan. Primate Rep 14:179–180
Nishida T, Kano T, Goodall J, McGrew WC, Nakamura M (1999) Ethogram and ethnography of Mahale chimpanzees. Anthropol Sci 107:141–188
Patterson F, Linden E (1981) The education of Koko. Holt, Rinehart, and Linden, New York
Pika S, Liebal K, Tomasello M (2003) Gestural communication in young gorillas (Gorilla gorilla): gestural repertoire, learning, and use. Am J Primatol 60:95–111
Plooij F (1979) How wild chimpanzee babies trigger the onset of mother–infant play and what the mother makes of it. In: Bullowa M (ed) Before speech: the beginnings of interpersonal communication. Cambridge University Press, Cambridge, pp 223–243
Plooij FX (1984) The behavioral development of free-living chimpanzee babies and infants. Ablex Publishing Corporation, Norwood
Pollick AS, de Waal FBM (2007) Ape gestures and language evolution. Proc Natl Acad Sci 104:8184–8189
Rendall D, Owren MJ, Ryan MJ (2009) What do animal signals mean? Anim Behav 78:233–240
Reynolds V (2005) The chimpanzees of the Budongo Forest. Oxford University Press, Oxford
Ristau C (1991) Aspects of the cognitive ethology of an injury-feigning bird, the piping plover. In: Ristau C (ed) Cognitive ethology: the minds of other animals. Lawrence Erlbaum Associates, Hillsdale, pp 91–126
Rizzolatti G, Arbib MA (1998) Language within our grasp. Trends Neurosci 21:188–194
Seyfarth RM, Cheney DL (1986) Vocal development in vervet monkeys. Anim Behav 34:1640–1658
Seyfarth RM, Cheney DL, Marler P (1980) Vervet monkey alarm calls: semantic communication in a free-ranging primate. Anim Behav 28:1070–1094
Seyfarth RM, Cheney DL, Bergman TJ (2005) Primate social cognition and the origins of language. Trends Cogn Sci 9:264–266
Snowdon CT (1990) Language capacities of nonhuman animals. Yearb Phys Anthropol 33:215–243
Taglialatela JP, Savage-Rumbaugh S, Baker LA (2003) Vocal production by a language-competent Pan paniscus. Int J Primatol 24:1–17
Tanner JE, Byrne RW (1996) Representation of action through iconic gesture in a captive lowland gorilla. Curr Anthropol 37:162–173
Tanner JE, Byrne RW (1999) The development of spontaneous gestural communication in a group of zoo-living lowland gorillas. In: Parker ST, Mitchell RW, Miles HL (eds) The mentalities of gorillas and orangutans. Comparative perspectives. Cambridge University Press, Cambridge, pp 211–239
Tomasello M (1990) Cultural transmission in the tool use and communicatory signaling of chimpanzees? In: Parker ST, Gibson KR (eds) “Language” and intelligence in monkeys and apes. Cambridge University Press, Cambridge, pp 274–311
Tomasello M, Call J (2007) Intentional communication in nonhuman primates. In: Call J, Tomasello M (eds) The gestural communication of apes and monkeys. Lawrence Erlbaum Associates, Mahwah, pp 1–15
Tomasello M, George B, Kruger A, Farrar J, Evans E (1985) The development of gestural communication in young chimpanzees. J Hum Evol 14:175–186
Tomasello M, Gust D, Frost TA (1989) A longitudinal investigation of gestural communication in young chimpanzees. Primates 30:35–50
Tomasello M, Call J, Nagell C, Olguin R, Carpenter M (1994) The learning and use of gestural signals by young chimpanzees: a trans-generational study. Primates 35:137–154
Tutin CEG, McGrew WC (1973) Chimpanzee copulatory behavior. Folia Primatol 19:237–256
Vauclair J (2004) Lateralization of communicative signals in nonhuman primates and the hypothesis of the gestural origin of language. Interact Stud 5:365–386
Zuberbuhler K (2002) A syntactic rule in forest monkey communication. Anim Behav 63:293–299
Zuberbuhler K, Cheney D, Seyfarth R (1999) Conceptual semantics in a nonhuman primate. J Comp Psychol 113:33–42
Acknowledgments
We thank all the staff of the Budongo Conservation Field Station, especially Amati Stephen, and thank the BCFS project’s founder Vernon Reynolds and its current scientific director Klaus Zuberbühler for allowing us to work at the site and to use data from the project records. For permission to work in Uganda, we thank the Uganda National Council for Science and Technology, the Presidents Office, the Uganda Wildlife Authority and the Uganda Forest Authority. Fieldwork of CH was generously supported by grants from the Wenner-Gren Foundation (http://wennergren.org) and the Russell Trust. The thoughtful comments of three anonymous referees were useful in improving the clarity of our theory and writing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hobaiter, C., Byrne, R.W. The gestural repertoire of the wild chimpanzee. Anim Cogn 14, 745–767 (2011). https://doi.org/10.1007/s10071-011-0409-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-011-0409-2