Abstract
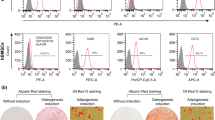
The aim of this study was to explore the effects of α-zearalanol (α-ZAL) on the proliferation of mouse bone-marrow-derived mesenchymal stem cells (BMSCs) and their differentiation into osteoblasts. Six- to eight-week-old BALB/C mice were used either as recipients or as bone marrow donors. BMSCs were isolated and collected using a differential adhesion method, with use of 10 % fetal bovine serum and Iscove’s modified Dulbecco’s medium. After the third generation, the BMSCs were randomly placed into the following subgroups: a control group, an osteogenic medium (OM) group, a 17β-estradiol group, an α-ZAL 10−7 mol/L group, an α-ZAL 10−6 mol/L group, and an α-ZAL 10−5 mol/L group. Flow cytometry was used to identify the BMSCs collected from the bone marrow. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide test was performed, and markers of the osteoblasts were measured in the different subgroups. In addition, expression of osteoprotegerin and expression of receptor activator of nuclear factor κB ligand were examined using Western blot. In contrast to the control and OM groups, BMSCs in the α-ZAL groups exhibited long fusiform shapes, and contact inhibition was observed when the cells were closely packed. After induction, the BMSCs grew well and exhibited triangular, star, polygonal, or irregular shapes. Clumps and multiple cells were evident. The trends of the proliferation and differentiation for the control, OM, 17β-estradiol, and α-ZAL groups were similar. Compared with the control and OM groups, in the α-ZAL groups the expression levels of alkaline phosphatase, procollagen type I N-terminal propeptide, bone morphogenetic protein 2, and osteocalcin were significantly increased (p < 0.05). In addition, α-ZAL inhibited osteoclastogenesis by increasing the expression of osteoprotegerin and decreasing the expression of nuclear factor κB ligand. In conclusion, α-ZAL can increase the proliferation of BMSCs and their differentiation into osteoblasts and can effectively suppress osteoclastogenesis.






Similar content being viewed by others
References
Jintaridth P, Tungtrongchitr R, Preutthipan S, Mutirangura A (2013) Hypomethylation of alu elements in post-menopausal women with osteoporosis. PLoS One 8:e70386
Bocheva G, Boyadjieva N (2011) Epigenetic regulation of fetal bone development and placental transfer of nutrients: progress for osteoporosis. Interdiscip Toxicol 4:167–172
Kung AW, Huang Q (2007) Genetic and environmental determinants of osteoporosis. J Musculoskelet Neuronal Interact 7:26
Kitazawa R, Kitazawa S (2007) Methylation status of a single CpG locus 3 bases upstream of TATA-box of receptor activator of nuclear factor-κB ligand (RANKL) gene promoter modulates cell-and tissue-specific RANKL expression and osteoclastogenesis. Mol Endocrinol 21:148–158
Villagra A, Gutierrez J, Paredes R, Sierra J, Puchi M, Imschenetzky M, Montecino M (2002) Reduced CpG methylation is associated with transcriptional activation of the bone-specific rat osteocalcin gene in osteoblasts. J Cell Biochem 85:112–122
Pacifici R (1998) Editorial: cytokines, estrogen, and postmenopausal osteoporosis—the second decade. Endocrinology 139:2659–2661
Baber RJ, Hara OJL, Boyle FM (2003) Hormone replacement therapy: to use or not to use? Med J Aust 178:630–633
Niu YB, Li YH, Kong XH, Zhang R, Sun Y, Li Q, Mei QB (2012) The beneficial effect of Radix Dipsaci total saponins on bone metabolism in vitro and in vivo and the possible mechanisms of action. Osteoporos Int 23:2649–2660
Dai S, Duan J, Lu Y, Cheng J, Ren J, Deng W, Wu Y (2004) α-Zearalanol, a phytoestrogen for cardiovascular therapy. Endocrine 25:117–119
Stob M, Baldwin RS, Tuite J, Andrews FN, Gillette KG (1962) Isolation of an anabolic, uterotrophic compound from corn infected with Gibberella zeae. Nature 196:1318
Liu T, Hou DD, Zhao Q, Liu W, Zhen PP, Xu JP, Wang W (2013) Phytoestrogen α-zearalanol attenuates homocysteine-induced apoptosis in human umbilical vein endothelial cells. Biomed Res Int 10:1155
Zong S, Wei B, Xiong C, Zhao Y, Zeng G (2012) The role of α-zearalanol in reversing bone loss induced by ovarian hormone deficiency in rats. J Bone Miner Metab 30:136–143
Sakamoto F, Hashimoto Y, Kishimoto N, Honda Y, Matsumoto N (2013) The utility of human dedifferentiated fat cells in bone tissue engineering in vitro. Cytotechnology 10:1007
Reddi AH (1994) Bone and cartilage differentiation. Curr Opin Genet Dev 4:737–744
Ishihara A, Bertone AL (2012) Cell-mediated and direct gene therapy for bone regeneration. Expert Opin Biol Ther 12:411–423
Wei L, Yi PS, Guo YZ (2008) Effects of estrogen on rat osteoblasts in vitro. J Clin Rehabil Tissue Eng Res 12:11
Zaidi M, Moonga BS, Sun L, Epstein S, Huang CLH, Abe E (2003) Understanding osteoclast formation and function: implications for future therapies for osteoporosis. Curr Opin Orthop 14:341–350
Pantschenko AG, Zhang W, Nahounou M, Mccarthy MB, Stover ML, Lichtler AC, Gronowicz GA (2005) Effect of osteoblast-targeted expression of Bcl-2 in bone: differential response in male and female mice. J Bone Miner Res 20:1414–1429
Parikka V, Peng Z, Hentunen T, Risteli J, Elo T, Väänänen HK, Härkönen P (2005) Estrogen responsiveness of bone formation in vitro and altered bone phenotype in aged estrogen receptor-α-deficient male and female mice. Eur J Endocrinol 152:301–314
Kevorkova O, Martineau C, Martin-Falstrault L, Sanchez-Dardon J, Brissette L, Moreau R (2013) Low-bone-mass phenotype of deficient mice for the cluster of differentiation 36 (CD36). PLoS One 8:e77701
Eriksen EF (2010) Cellular mechanisms of bone remodeling. Rev Endocr Metab Disord 11:219–227
Jönsson S, Hjorth-Hansen H, Olsson B et al (2012) Imatinib inhibits proliferation of human mesenchymal stem cells and promotes early but not late osteoblast differentiation in vitro. J Bone Miner Metab 30:119–123
Peng H, Wright V, Usas A, Gearhart B, Shen HC, Cummins J, Huard J (2002) Synergistic enhancement of bone formation and healing by stem cell-expressed VEGF and bone morphogenetic protein-4. J Clin Investig 110:751–759
Kiyozuka Y, Miyazaki H, Yoshizawa K, Senzaki H, Yamamoto D, Inoue K, Tsubura A (1999) Case report: an autopsy case of malignant mesothelioma with osseous and cartilaginous differentiation (bone morphogenetic protein-2 in mesothelial cells and its tumor). Dig Dis Sci 44:1626–1631
Prall WC, Haasters F, Heggebö J, Polzer H, Schwarz C, Gassner C, Schieker M (2013) Mesenchymal stem cells from osteoporotic patients feature impaired signal transduction but sustained osteoinduction in response to BMP-2 stimulation. Biochem Biophys Res Commun 440:617–622
Hashemibeni B, Jafary F, Esmaeil N, Goharian V, Feizi G, Heidari F, Zarkesh SH (2013) Comparison of phenotypic characterization between differentiated osteoblasts from stem cells and calvaria osteoblasts in vitro. Int J Prev Med 4:180
Schäck LM, Noack S, Winkler R, Wißmann G, Behrens P, Wellmann M, Hoffmann A (2013) The phosphate source influences gene expression and quality of mineralization during in vitro osteogenic differentiation of human mesenchymal stem cells. PLoS One 8:e65943
Foresta C, Strapazzon G, De TL, Gianesello L, Calcagno A, Pilon C, Vettor R (2010) Evidence for osteocalcin production by adipose tissue and its role in human metabolism. J Clin Endocrinol Metab 95:3502–3506
Pérez SM, Somoza MJM, Barros AF, Rey JMG, García GA (2010) RANK/RANKL/OPG role in distraction osteogenesis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 109:679–686
Niu YB, Li YH, Kong XH, Zhang R, Sun Y, Li Q, Mei QB (2012) The beneficial effect of Radix Dipsaci total saponins on bone metabolism in vitro and in vivo and the possible mechanisms of action. Osteoporos Int 23:2649–2660
Dong Y, Yang N, Liu Y, Li Q, Zuo P (2011) The neuroprotective effects of phytoestrogen α-zearalanol on β-amyloid-induced toxicity in differentiated PC-12 cells. Eur J Pharmacol 670:392–398
Acknowledgment
We thank the Natural Science Foundation of Guangxi (2014GXNSFAA118400) for its support.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
G. Zeng, Y. Fang, and J. Peng contributed equally to this work and should be considered as co-first authors.
About this article
Cite this article
Zong, S., Zeng, G., Fang, Y. et al. The effects of α-zearalanol on the proliferation of bone-marrow-derived mesenchymal stem cells and their differentiation into osteoblasts. J Bone Miner Metab 34, 151–160 (2016). https://doi.org/10.1007/s00774-015-0659-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-015-0659-1