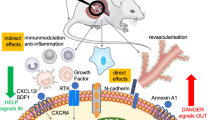
Abstract
Aims
Mesenchymal stem cells (MSCs) are multipotent cells with immunomodulatory properties. We tested the ability of MSCs to delay islet allograft rejection.
Methods
Mesenchymal stem cells were generated in vitro from C57BL/6 and BALB/c mice bone marrow, and their immunomodulatory properties were tested in vitro. We then tested the effect of a local or systemic administration of heterologous and autologous MSCs on graft survival in a fully allogeneic model of islet transplantation (BALB/c islets into C57BL/6 mice).
Results
In vitro, autologous, but not heterologous, MSCs abrogated immune cell proliferation in response to alloantigens and skewed the immune response toward a Th2 profile. A single dose of autologous MSCs co-transplanted under the kidney capsule with allogeneic islets delayed islet rejection, reduced graft infiltration, and induced long-term graft function in 30 % of recipients. Based on ex vivo analysis of recipient splenocytes, the use of autologous MSCs did not appear to have any systemic effect on the immune response toward graft alloantigens. The systemic injection of autologous MSCs or the local injection of heterologous MSCs failed to delay islet graft rejection.
Conclusion
Autologous, but not heterologous, MSCs showed multiple immunoregulatory properties in vitro and delayed allograft rejection in vivo when co-transplanted with islets; however, they failed to prevent rejection when injected systemically. Autologous MSCs thus appear to produce a local immunoprivileged site, which promotes graft survival.






Similar content being viewed by others
References
Nathan DM, Cleary PA, Backlund JY et al (2005) Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 353:2643–2653
Nathan DM, Lachin J, Cleary P et al (2003) Intensive diabetes therapy and carotid intima-media thickness in type 1 diabetes mellitus. N Engl J Med 348:2294–2303
Bloomgarden ZT (2004) Consequences of diabetes: cardiovascular disease. Diabetes Care 27:1825–1831
Bloomgarden ZT (2004) Diabetes complications. Diabetes Care 27:1506–1514
Trento M, Raballo M, Trevisan M et al (2012) A cross-sectional survey of depression, anxiety, and cognitive function in patients with type 2 diabetes. Acta Diabetol 49:199–203
Piarulli F, Sartore G, Lapolla A (2013) Glyco-oxidation and cardiovascular complications in type 2 diabetes: a clinical update. Acta Diabetol 50:101–110
Tarallo S, Beltramo E, Berrone E, Porta M (2012) Human pericyte-endothelial cell interactions in co-culture models mimicking the diabetic retinal microvascular environment. Acta Diabetol 49(Suppl 1):S141–S151
Fiorina P, Shapiro AM, Ricordi C, Secchi A (2008) The clinical impact of islet transplantation. Am J Transplant 8:1990–1997
Fiorina P, Vergani A, Petrelli A et al (2008) Metabolic and immunological features of the failing islet-transplanted patient. Diabetes Care 31:436–438
Fiorina P, Vezzulli P, Bassi R et al (2012) Near normalization of metabolic and functional features of the central nervous system in type 1 diabetic patients with end-stage renal disease after kidney-pancreas transplantation. Diabetes Care 35:367–374
Venturini M, Fiorina P, Maffi P et al (2006) Early increase of retinal arterial and venous blood flow velocities at color Doppler imaging in brittle type 1 diabetes after islet transplant alone. Transplantation 81:1274–1277
Del Carro U, Fiorina P, Amadio S et al (2007) Evaluation of polyneuropathy markers in type 1 diabetic kidney transplant patients and effects of islet transplantation: neurophysiological and skin biopsy longitudinal analysis. Diabetes Care 30:3063–3069
Fiorina P, Secchi A (2007) Pancreatic islet cell transplant for treatment of diabetes. Endocrinol Metab Clin North Am 36:999–1013; ix
Shapiro AM, Ricordi C, Hering BJ et al (2006) International trial of the Edmonton protocol for islet transplantation. N Engl J Med 355:1318–1330
Ansari MJ, Fiorina P, Dada S et al (2008) Role of ICOS pathway in autoimmune and alloimmune responses in NOD mice. Clin Immunol 126:140–147
Guleria I, Gubbels Bupp M, Dada S et al (2007) Mechanisms of PDL1-mediated regulation of autoimmune diabetes. Clin Immunol 125:16–25
Vergani A, D’Addio F, Jurewicz M et al (2010) A novel clinically relevant strategy to abrogate autoimmunity and regulate alloimmunity in NOD mice. Diabetes 59:2253–2264
Vergani A, Fotino C, D’Addio F et al (2013) Effect of the purinergic inhibitor oxidized ATP in a model of islet allograft rejection. Diabetes 62:1665–1675
Maffi P, Bertuzzi F, De Taddeo F et al (2007) Kidney function after islet transplant alone in type 1 diabetes: impact of immunosuppressive therapy on progression of diabetic nephropathy. Diabetes Care 30:1150–1155
Friedenstein AJ, Chailakhjan RK, Lalykina KS (1970) The development of fibroblast colonies in monolayer cultures of guinea-pig bone marrow and spleen cells. Cell Tissue Kinet 3:393–403
Tyndall A, Walker UA, Cope A et al (2007) Immunomodulatory properties of mesenchymal stem cells: a review based on an interdisciplinary meeting held at the Kennedy Institute of Rheumatology Division, London, UK, 31 October 2005. Arthritis Res Ther 9:301
Cohen JA (2013) Mesenchymal stem cell transplantation in multiple sclerosis. J Neurol Sci 333:43–49
Fiorina P, Jurewicz M, Augello A et al (2009) Immunomodulatory function of bone marrow-derived mesenchymal stem cells in experimental autoimmune type 1 diabetes. J Immunol 183:993–1004
Fiorina P, Voltarelli J, Zavazava N (2011) Immunological applications of stem cells in type 1 diabetes. Endocr Rev 32:725–754
Oswald J, Boxberger S, Jorgensen B et al (2004) Mesenchymal stem cells can be differentiated into endothelial cells in vitro. Stem Cells 22:377–384
Le Blanc K, Samuelsson H, Lonnies L, Sundin M, Ringden O (2007) Generation of immunosuppressive mesenchymal stem cells in allogeneic human serum. Transplantation 84:1055–1059
De Miguel MP, Fuentes-Julian S, Blazquez-Martinez A et al (2012) Immunosuppressive properties of mesenchymal stem cells: advances and applications. Curr Mol Med 12:574–591
MacFarlane RJ, Graham SM, Davies PS et al (2013) Anti-inflammatory role and immunomodulation of mesenchymal stem cells in systemic joint diseases: potential for treatment. Expert Opin Ther Targets 17:243–254
Jurewicz M, Yang S, Augello A et al (2010) Congenic mesenchymal stem cell therapy reverses hyperglycemia in experimental type 1 diabetes. Diabetes 59:3139–3147
Papadopoulou A, Yiangou M, Athanasiou E et al (2012) Mesenchymal stem cells are conditionally therapeutic in preclinical models of rheumatoid arthritis. Ann Rheum Dis 71:1733–1740
Eggenhofer E, Renner P, Soeder Y et al (2011) Features of synergism between mesenchymal stem cells and immunosuppressive drugs in a murine heart transplantation model. Transpl Immunol 25:141–147
Ding Y, Xu D, Feng G, Bushell A, Muschel RJ, Wood KJ (2009) Mesenchymal stem cells prevent the rejection of fully allogenic islet grafts by the immunosuppressive activity of matrix metalloproteinase-2 and -9. Diabetes 58:1797–1806
Carvello M, Petrelli A, Vergani A et al (2012) Inotuzumab ozogamicin murine analog-mediated B-cell depletion reduces anti-islet allo- and autoimmune responses. Diabetes 61:155–165
Serreze DV, Chapman HD, Varnum DS et al (1996) B lymphocytes are essential for the initiation of T cell-mediated autoimmune diabetes: analysis of a new “speed congenic” stock of NOD.Ig mu null mice. J Exp Med 184:2049–2053
Vergani A, Clissi B, Sanvito F, Doglioni C, Fiorina P, Pardi R (2009) Laser capture microdissection as a new tool to assess graft-infiltrating lymphocytes gene profile in islet transplantation. Cell Transplant 18:827–832
Vergani A, Tezza S, D’Addio F et al (2013) Long-term heart transplant survival by targeting the ionotropic purinergic receptor P2X7. Circulation 127:463–475
Astorri E, Fiorina P, Gavaruzzi G, Astorri A, Magnati G (1997) Left ventricular function in insulin-dependent and in non-insulin-dependent diabetic patients: radionuclide assessment. Cardiology 88:152–155
Paroni R, Fermo I, Fiorina P, Cighetti G (2005) Determination of asymmetric and symmetric dimethylarginines in plasma of hyperhomocysteinemic subjects. Amino Acids 28:389–394
Fiorina P, Vergani A, Dada S et al (2008) Targeting CD22 reprograms B-cells and reverses autoimmune diabetes. Diabetes 57:3013–3024
Francese R, Fiorina P (2010) Immunological and regenerative properties of cord blood stem cells. Clin Immunol 136:309–322
D’Addio F, Maffi P, Vezzulli P et al (2014) Islet transplantation stabilizes hemostatic abnormalities and cerebral metabolism in individuals with type 1 diabetes. Diabetes Care 37:267–276
Gu F, Molano I, Ruiz P, Sun L, Gilkeson GS (2012) Differential effect of allogeneic versus syngeneic mesenchymal stem cell transplantation in MRL/lpr and (NZB/NZW)F1 mice. Clin Immunol 145:142–152
Eliopoulos N, Stagg J, Lejeune L, Pommey S, Galipeau J (2005) Allogeneic marrow stromal cells are immune rejected by MHC class I- and class II-mismatched recipient mice. Blood 106:4057–4065
Ito T, Itakura S, Todorov I et al (2010) Mesenchymal stem cell and islet co-transplantation promotes graft revascularization and function. Transplantation 89:1438–1445
Borg DJ, Weigelt M, Wilhelm C et al (2014) Mesenchymal stromal cells improve transplanted islet survival and islet function in a syngeneic mouse model. Diabetologia 57:522–531
Rossignol J, Boyer C, Leveque X et al (2011) Mesenchymal stem cell transplantation and DMEM administration in a 3NP rat model of Huntington’s disease: morphological and behavioral outcomes. Behav Brain Res 217:369–378
Park KS, Kim YS, Kim JH et al (2010) Trophic molecules derived from human mesenchymal stem cells enhance survival, function, and angiogenesis of isolated islets after transplantation. Transplantation 89:509–517
Favaro E, Carpanetto A, Lamorte S et al (2014) Human mesenchymal stem cell-derived microvesicles modulate T cell response to islet antigen glutamic acid decarboxylase in patients with type 1 diabetes. Diabetologia 57:1664–1673
Cantaluppi V, Biancone L, Figliolini F et al (2012) Microvesicles derived from endothelial progenitor cells enhance neoangiogenesis of human pancreatic islets. Cell Transplant 21:1305–1320
Yagi H, Soto-Gutierrez A, Parekkadan B et al (2010) Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cell Transplant 19:667–679
Glennie S, Soeiro I, Dyson PJ, Lam EW, Dazzi F (2005) Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood 105:2821–2827
Jiang XX, Zhang Y, Liu B et al (2005) Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 105:4120–4126
Spaggiari GM, Capobianco A, Becchetti S, Mingari MC, Moretta L (2006) Mesenchymal stem cell-natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation. Blood 107:1484–1490
Corcione A, Benvenuto F, Ferretti E et al (2006) Human mesenchymal stem cells modulate B-cell functions. Blood 107:367–372
Duprez IR, Johansson U, Nilsson B, Korsgren O, Magnusson PU (2011) Preparatory studies of composite mesenchymal stem cell islets for application in intraportal islet transplantation. Ups J Med Sci 116:8–17
Acknowledgments
Paolo Fiorina is the recipient of a JDRF Career Development Award, an ASN Career Development Award, and an ADA Mentor-based Fellowship Grant. P.F. is also supported by a Translational Research Program (TRP) Grant from Boston Children’s Hospital; Harvard Stem Cell Institute Grant (“Diabetes Program” DP-0123-12-00); Italian Ministry of Health Grant RF- 2010-2303119. P. F. and Andrea Vergani are supported of an Italian Ministry of Health Grant: (“Staminali” RF-FSR-2008-1213704). A.V. is supported by the “AMD-SID Pasquale di Coste Scolarship”. Francesca D’Addio is a recipient of Italian Scientists and Scholars of North America Foundation (ISSNAF)-Fondazione Marche Fellowship. Roberto Bassi is supported by an ADA Mentor-based Fellowship Grant to P.F and by an AST Genentech Clinical Science Fellowship Grant. A.V. conducted this study as partial fulfillment of his PhD in Molecular Medicine, San Raffaele University, Milan, Italy.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
Principles of laboratory animal care” (NIH publication No. 86-23, revised 1985) were followed, as well as specific national laws (e.g., the current version of the German Law on the Protection of Animals) were applicable.
Informed consent
This study does not involve human subjects. No informed consent needs to be obtained.
Author information
Authors and Affiliations
Corresponding author
Additional information
Managed by Massimo Federici.
Moufida Ben Nasr and Andrea Vergani have contributed equally to the paper.
Rights and permissions
About this article
Cite this article
Ben Nasr, M., Vergani, A., Avruch, J. et al. Co-transplantation of autologous MSCs delays islet allograft rejection and generates a local immunoprivileged site. Acta Diabetol 52, 917–927 (2015). https://doi.org/10.1007/s00592-015-0735-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-015-0735-y