Abstract
Background
The underlying molecular mechanism that leads to development of chronic pancreatitis remains elusive. The aim of this study is to understand the downstream inflammatory signaling involved in progression of chronic pancreatitis, and to use withaferin A (WA), a small molecule inhibitor of nuclear factor κB (NFκB), to prevent progression of chronic pancreatitis.
Methods
Two different protocols were used to induce pancreatitis in mice: standard and stringent administration of cerulein. The severity of pancreatitis was assessed by means of pancreatic histology and serum amylase levels. Immunohistochemistry and flow-cytometric analysis was performed to visualize immune cell infiltration into the pancreas. Real-time PCR and Western blot were used to analyze the downstream signaling mechanism involved in the development of chronic pancreatitis.
Results
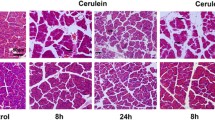
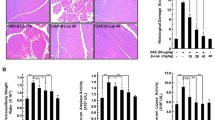
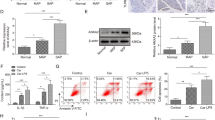
The severity of cerulein-induced pancreatitis was reduced significantly by WA, used as either preventive or curative treatment. Immune cell infiltration into the pancreas and acinar cell death were efficiently reduced by WA treatment. Expression of proinflammatory and proapoptotic genes regulated by NFκB activation was increased by cerulein treatment, and WA suppressed these genes significantly. Sustained endoplasmic reticulum stress activation by cerulein administration was reduced. NLRP3 inflammasome activation in cerulein-induced pancreatitis was identified, and this was also potently blocked by WA. The human pancreatitis tissue gene signature correlated with the mouse model.
Conclusions
Our data provide evidence for the role of NFκB in the pathogenesis of chronic pancreatitis, and strongly suggest that WA could be used as a potential therapeutic drug to alleviate some forms of chronic pancreatitis.







Similar content being viewed by others
References
Ammann RW, Muellhaupt B. The natural history of pain in alcoholic chronic pancreatitis. Gastroenterology. 1999;116(5):1132–40.
Mullady DK, Yadav D, Amann ST, et al. Type of pain, pain-associated complications, quality of life, disability and resource utilisation in chronic pancreatitis: a prospective cohort study. Gut. 2011;60(1):77–84.
Whitcomb DC. Genetic risk factors for pancreatic disorders. Gastroenterology. 2013;144(6):1292–302.
Huang H, Liu Y, Daniluk J, et al. Activation of nuclear factor-κB in acinar cells increases the severity of pancreatitis in mice. Gastroenterology. 2013;144(1):202–10.
Sah RP, Dudeja V, Dawra RK, et al. Cerulein-induced chronic pancreatitis does not require intra-acinar activation of trypsinogen in mice. Gastroenterology. 2013;144(5):1076–85.e2.
Aleksic T, Baumann B, Wagner M, et al. Cellular immune reaction in the pancreas is induced by constitutively active IκB kinase-2. Gut. 2007;56(2):227–36.
Baumann B, Wagner M, Aleksic T, et al. Constitutive IKK2 activation in acinar cells is sufficient to induce pancreatitis in vivo. J Clin Investig. 2007;117(6):1502–13.
Yan MX, Ren HB, Kou Y, et al. Involvement of nuclear factor kappa B in high-fat diet-related pancreatic fibrosis in rats. Gut Liver. 2012;6(3):381–7.
Altavilla D, Famulari C, Passaniti M, et al. Attenuated cerulein-induced pancreatitis in nuclear factor-kappaB-deficient mice. Lab Investig. 2003;83(12):1723–32.
Steinle AU, Weidenbach H, Wagner M, et al. NF-κB/Rel activation in cerulein pancreatitis. Gastroenterology. 1999;116(2):420–30.
Kaufman RJ. Stress signaling from the lumen of the endoplasmic reticulum: coordination of gene transcriptional and translational controls. Genes Dev. 1999;13(10):1211–33.
Ron D, Walter P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol. 2007;8(7):519–29.
Schröder M, Kaufman RJ. The mammalian unfolded protein response. Annu Rev Biochem. 2005;74:739–89.
Sah RP, Garg SK, Dixit AK, et al. Endoplasmic reticulum stress is chronically activated in chronic pancreatitis. J Biol Chem. 2014;289(40):27551–61.
Schroder K, Tschopp J. The inflammasomes. Cell. 2010;140(6):821–32.
Heyninck K, Lahtela-Kakkonen M, Van der Veken P, et al. Withaferin A inhibits NF-kappaB activation by targeting cysteine 179 in IKKβ. Biochem Pharmacol. 2014;91(4):501–9.
Lara LF, Levy MJ. Idiopathic recurrent acute pancreatitis. MedGenMed. 2004;6(4):10.
Neuschwander-Tetri BA, Burton FR, Presti ME, et al. Repetitive self-limited acute pancreatitis induces pancreatic fibrogenesis in the mouse. Dig Dis Sci. 2000;45(4):665–74.
Mayer-Scholl A, Averhoff P, Zychlinsky A. How do neutrophils and pathogens interact? Curr Opin Microbiol. 2004;7(1):62–6.
Kim I, Xu W, Reed JC. Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities. Nat Rev Drug Discov. 2008;7(12):1013–30.
Walter P, Ron D. The unfolded protein response: from stress pathway to homeostatic regulation. Science. 2011;334(6059):1081–6.
Latz E, Xiao TS, Stutz A. Activation and regulation of the inflammasomes. Nat Rev Immunol. 2013;13(6):397–411.
Pandol SJ, Saluja AK, Imrie CW, et al. Acute pancreatitis: bench to the bedside. Gastroenterology. 2007;132(3):1127–51.
Grady T, Liang P, Ernst SA, et al. Chemokine gene expression in rat pancreatic acinar cells is an early event associated with acute pancreatitis. Gastroenterology. 1997;113(6):1966–75.
Gukovskaya AS, Gukovsky I, Zaninovic V, et al. Pancreatic acinar cells produce, release, and respond to tumor necrosis factor-alpha. Role in regulating cell death and pancreatitis. J Clin Investig. 1997;100(7):1853–62.
Muniraj T, Aslanian HR, Farrell J, et al. Chronic pancreatitis, a comprehensive review and update. Part I: epidemiology, etiology, risk factors, genetics, pathophysiology, and clinical features. Dis Mon. 2014;60(12):530–50.
Howes N, Neoptolemos JP. Risk of pancreatic ductal adenocarcinoma in chronic pancreatitis. Gut. 2002;51(6):765–6.
Malka D, Hammel P, Maire F, et al. Risk of pancreatic adenocarcinoma in chronic pancreatitis. Gut. 2002;51(6):849–52.
Saluja AK, Bhagat L, Lee HS, et al. Secretagogue-induced digestive enzyme activation and cell injury in rat pancreatic acini. Am J Physiol. 1999;276(4 Pt 1):G835–42.
Booth DM, Murphy JA, Mukherjee R, et al. Reactive oxygen species induced by bile acid induce apoptosis and protect against necrosis in pancreatic acinar cells. Gastroenterology. 2011;140(7):2116–25.
Chen X, Ji B, Han B, et al. NF-kappaB activation in pancreas induces pancreatic and systemic inflammatory response. Gastroenterology. 2002;122(2):448–57.
Mareninova OA, Hermann K, French SW, et al. Impaired autophagic flux mediates acinar cell vacuole formation and trypsinogen activation in rodent models of acute pancreatitis. J Clin Investig. 2009;119(11):3340–55.
Behrendorff N, Floetenmeyer M, Schwiening C, et al. Protons released during pancreatic acinar cell secretion acidify the lumen and contribute to pancreatitis in mice. Gastroenterology. 2010;139(5):1711–20, 20 e1–5.
Sah RP, Dawra RK, Saluja AK. New insights into the pathogenesis of pancreatitis. Curr Opin Gastroenterol. 2013;29(5):523–30.
Lerch MM, Gorelick FS. Models of acute and chronic pancreatitis. Gastroenterology. 2013;144(6):1180–93.
Algul H, Treiber M, Lesina M, et al. Pancreas-specific RelA/p65 truncation increases susceptibility of acini to inflammation-associated cell death following cerulein pancreatitis. J Clin Investig. 2007;117(6):1490–501.
Neuhofer P, Liang S, Einwachter H, et al. Deletion of IκBα activates RelA to reduce acute pancreatitis in mice through up-regulation of Spi2A. Gastroenterology. 2013;144(1):192–201.
Gukovsky I, Gukovskaya A. Nuclear factor-κB in pancreatitis: jack-of-all-trades, but which one is more important? Gastroenterology. 2013;144(1):26–9.
He S, Wang L, Miao L, et al. Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-α. Cell. 2009;137(6):1100–11.
Ozaki E, Campbell M, Doyle SL. Targeting the NLRP3 inflammasome in chronic inflammatory diseases: current perspectives. J Inflamm Res. 2015;8:15–27.
Hoque R, Sohail M, Malik A, et al. TLR9 and the NLRP3 inflammasome link acinar cell death with inflammation in acute pancreatitis. Gastroenterology. 2011;141(1):358–69.
Lin Y, Lin L-J, Jin Y, et al. Correlation between serum levels of high mobility group box-1 protein and pancreatitis: a meta-analysis. BioMed Res Int. 2015;2015:10.
Kaileh M, Vanden Berghe W, Heyerick A, et al. Withaferin a strongly elicits IκB kinase β hyperphosphorylation concomitant with potent inhibition of its kinase activity. J Biol Chem. 2007;282(7):4253–64.
Thaiparambil JT, Bender L, Ganesh T, et al. Withaferin A inhibits breast cancer invasion and metastasis at sub-cytotoxic doses by inducing vimentin disassembly and serine 56 phosphorylation. Int J Cancer. 2011;129(11):2744–55.
Mohan R, Hammers HJ, Bargagna-Mohan P, et al. Withaferin A is a potent inhibitor of angiogenesis. Angiogenesis. 2004;7(2):115–22.
Vanden Berghe W, Sabbe L, Kaileh M, et al. Molecular insight in the multifunctional activities of withaferin A. Biochem Pharmacol. 2012;84(10):1282–91.
Hoesel B, Schmid JA. The complexity of NF-κB signaling in inflammation and cancer. Mol Cancer. 2013;12:86.
Sutherland DE, Radosevich DM, Bellin MD, et al. Total pancreatectomy and islet autotransplantation for chronic pancreatitis. J Am Coll Surg. 2012;214(4):409–24; discussion 424-6.
Bellin MD, Gelrud A, Arreaza-Rubin G, et al. Total pancreatectomy with islet autotransplantation: summary of an NIDDK workshop. Ann Surg. 2015;261(1):21–9.
SoRelle JA, Itoh T, Peng H, et al. Withaferin A inhibits pro-inflammatory cytokine-induced damage to islets in culture and following transplantation. Diabetologia. 2013;56(4):814–24.
Acknowledgments
The authors gratefully acknowledge Morihito Takita, Prathab Balaji Saravanan, and Faisal Kunnathodi. Technical assistance by Nofit Borenstein-Auerbach, Omar Khan, Ana Rahman, and Yoshiko Tamura is also acknowledged. The manuscript editing service provided by Cynthia Orticio is highly appreciated.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kanak, M.A., Shahbazov, R., Yoshimatsu, G. et al. A small molecule inhibitor of NFκB blocks ER stress and the NLRP3 inflammasome and prevents progression of pancreatitis. J Gastroenterol 52, 352–365 (2017). https://doi.org/10.1007/s00535-016-1238-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-016-1238-5