Abstract
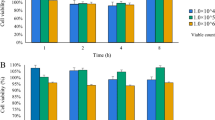
The potential efficacy of a probiotic-based preventative strategy against intestinal mucositis has yet to be investigated in detail. We evaluated supernatants (SN) from Escherichia coli Nissle 1917 (EcN) and Lactobacillus rhamnosus GG (LGG) for their capacity to prevent 5-fluorouracil (5-FU)-induced damage to intestinal epithelial cells. A 5-day study was performed. IEC-6 cells were treated daily from days 0 to 3, with 1 mL of PBS (untreated control), de Man Rogosa Sharpe (MRS) broth, tryptone soy roth (TSB), LGG SN, or EcN SN. With the exception of the untreated control cells, all groups were treated with 5-FU (5 μM) for 24 h at day 3. Transepithelial electrical resistance (TEER) was determined on days 3, 4, and 5, while activation of caspases 3 and 7 was determined on days 4 and 5 to assess apoptosis. Pretreatment with LGG SN increased TEER (p < 0.05) compared to controls at day 3. 5-FU administration reduced TEER compared to untreated cells on days 4 and 5. Pretreatment with MRS, LGG SN, TSB, and EcN SN partially prevented the decrease in TEER induced by 5-FU on day 4, while EcN SN also improved TEER compared to its TSB vehicle control. These differences were also observed at day 5, along with significant improvements in TEER in cells treated with LGG and EcN SN compared to healthy controls. 5-FU increased caspase activity on days 4 and 5 compared to controls. At day 4, cells pretreated with MRS, TSB, LGG SN, or EcN SN all displayed reduced caspase activity compared to 5-FU controls, while both SN groups had significantly lower caspase activity than their respective vehicle controls. Caspase activity in cells pretreated with MRS, LGG SN, and EcN SN was also reduced at day 5, compared to 5-FU controls. We conclude that pretreatment with selected probiotic SN could prevent or inhibit enterocyte apoptosis and loss of intestinal barrier function induced by 5-FU, potentially forming the basis of a preventative treatment modality for mucositis.



Similar content being viewed by others
References
Bowen JM, Gibson RJ, Cummins AG, Keefe DM (2006) Intestinal mucositis: the role of the Bcl-2 family, p53 and caspases in chemotherapy-induced damage. Support Care Canc 14:713–731
Soares PM, Mota JM, Gomes AS, Oliveira RB, Assreuy AM, Brito GA, Santos AA, Ribeiro RA, Souza MH (2008) Gastrointestinal dysmotility in 5-fluorouracil-induced intestinal mucositis outlasts inflammatory process resolution. Canc Chemother Pharmacol 63:91–98
Daniele B, Secondulfo M, De Vivo R, Pignata S, De Magistris L, Delrio P, Palaia R, Barletta E, Tambaro R, Carratu R (2001) Effect of chemotherapy with 5-fluorouracil on intestinal permeability and absorption in patients with advanced colorectal cancer. J Clin Gastroenterol 32:228–230
Duncan M, Grant G (2003) Oral and intestinal mucositis—causes and possible treatments. Aliment Pharmacol Ther 18:853–874
Sartor RB (2004) Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: antibiotics, probiotics, and prebiotics. Gastroenterology 126:1620–1633
Prisciandaro LD, Geier MS, Butler RN, Cummins AG, Howarth GS (2011) Probiotic factors partially improve parameters of 5-fluorouracil-induced intestinal mucositis in rats. Canc Biol Ther 11(7):671–677
Khailova L, Mount Patrick SK, Arganbright KM, Halpern MD, Kinouchi T, Dvorak B (2010) Bifidobacterium bifidum reduces apoptosis in the intestinal epithelium in necrotizing enterocolitis. Am J Physiol Gastrointest Liver Physiol 299:G1118–G1127
Philippe D, Favre L, Foata F, Adolfsson O, Perruisseau-Carrier G, Vidal K, Reuteler G, Dayer-Schneider J, Mueller C, Blum S (2011) Bifidobacterium lactis attenuates onset of inflammation in a murine model of colitis. World J Gastroenterol 17:459–469
Ueno N, Fujiya M, Segawa S, Nata T, Moriichi K, Tanabe H, Mizukami Y, Kobayashi N, Ito K, Kohgo Y (2011) Heat-killed body of Lactobacillus brevis SBC8803 ameliorates intestinal injury in a murine model of colitis by enhancing the intestinal barrier function. Inflamm Bowel Dis
Prisciandaro L, Geier M, Butler R, Cummins A, Howarth G (2009) Probiotics and their derivatives as treatments for inflammatory bowel disease. Inflamm Bowel Dis 15(12):1906–1914
Seth A, Yan F, Polk DB, Rao RK (2008) Probiotics ameliorate the hydrogen peroxide-induced epithelial barrier disruption by a PKC- and MAP kinase-dependent mechanism. Am J Physiol Gastrointest Liver Physiol 294:G1060–G1069
Sokol H, Pigneur B, Watterlot L et al (2008) Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci U S A 105:16731–16736
Donato KA, Gareau MG, Wang YJ, Sherman PM (2010) Lactobacillus rhamnosus GG attenuates interferon-γ and tumour necrosis factor-alpha-induced barrier dysfunction and pro-inflammatory signalling. Microbiology 156:3288–3297
Lin PW, Nasr TR, Berardinelli AJ, Kumar A, Neish AS (2008) The probiotic Lactobacillus GG may augment intestinal host defense by regulating apoptosis and promoting cytoprotective responses in the developing murine gut. Pediatr Res 64:511–516
Ukena SN, Singh A, Dringenberg U, Engelhardt R, Seidler U, Hansen W, Bleich A, Bruder D, Franzke A, Rogler G, Suerbaum S, Buer J, Gunzer F, Westendorf AM (2007) Probiotic Escherichia coli Nissle 1917 inhibits leaky gut by enhancing mucosal integrity. PLoS One 2:e1308
Logan RM, Stringer AM, Bowen JM, Yeoh AS, Gibson RJ, Sonis ST, Keefe DM (2007) The role of pro-inflammatory cytokines in cancer treatment-induced alimentary tract mucositis: pathobiology, animal models and cytotoxic drugs. Cancer Treat Rev 33:448–460
Zyrek AA, Cichon C, Helms S, Enders C, Sonnenborn U, Schmidt MA (2007) Molecular mechanisms underlying the probiotic effects of Escherichia coli Nissle 1917 involve ZO-2 and PKCzeta redistribution resulting in tight junction and epithelial barrier repair. Cell Microbiol 9:804–816
Basuroy S, Sheth P, Kuppuswamy D, Balasubramanian S, Ray RM, Rao RK (2003) Expression of kinase-inactive c-Src delays oxidative stress-induced disassembly and accelerates calcium-mediated reassembly of tight junctions in the Caco-2 cell monolayer. J Biol Chem 278:11916–11924
Otte JM, Mahjurian-Namari R, Brand S, Werner I, Schmidt WE, Schmitz F (2009) Probiotics regulate the expression of COX-2 in intestinal epithelial cells. Nutr Cancer 61:103–113
Johnson-Henry KC, Donato KA, Shen-Tu G, Gordanpour M, Sherman PM (2008) Lactobacillus rhamnosus strain GG prevents enterohemorrhagic Escherichia coli O157:H7-induced changes in epithelial barrier function. Infect Immun 76:1340–1348
Myllyluoma E, Ahonen AM, Korpela R, Vapaatalo H, Kankuri E (2008) Effects of multispecies probiotic combination on Helicobacter pylori infection in vitro. Clin Vaccine Immunol 15:1472–1482
Sanchez B, Schmitter JM, Urdaci MC (2009) Identification of novel proteins secreted by Lactobacillus rhamnosus GG grown in de Mann-Rogosa-Sharpe broth. Lett Appl Microbiol 48:618–622
Lin YP, Thibodeaux CH, Pena JA, Ferry GD, Versalovic J (2008) Probiotic Lactobacillus reuteri suppress proinflammatory cytokines via c-Jun. Inflamm Bowel Dis 14:1068–1083
Yan F, Cao H, Cover TL, Whitehead R, Washington MK, Polk DB (2007) Soluble proteins produced by probiotic bacteria regulate intestinal epithelial cell survival and growth. Gastroenterology 132:562–575
Oxoid (2010) Dehydrated culture media: modified tryptone soya broth. [Online]. Available at http://www.oxoid.com/UK/blue/prod_detail/prod_detail.asp?pr=CM0989&sec=2&org=72&c=UK&lang=EN (accessed on February 1st 2011)
Tuhacek LM, Mackey AD, Li N, DeMarco VG, Stevens G, Neu J (2004) Substitutes for glutamine in proliferation of rat intestinal epithelial cells. Nutrition 20:292–297
Li N, Neu J (2009) Glutamine deprivation alters intestinal tight junctions via a PI3-K/Akt mediated pathway in Caco-2 cells. J Nutr 139:710–714
Potsic B, Holliday N, Lewis P, Samuelson D, DeMarco V, Neu J (2002) Glutamine supplementation and deprivation: effect on artificially reared rat small intestinal morphology. Pediatr Res 52:430–436
Sukhotnik I, Mogilner JG, Karry R, Shamian B, Lurie M, Kokhanovsky N, Ure BM, Coran AG (2009) Effect of oral glutamine on enterocyte turnover during methotrexate-induced mucositis in rats. Digestion 79:5–13
Windmueller HG, Spaeth AE (1974) Uptake and metabolism of plasma glutamine by the small intestine. J Biol Chem 249:5070–5079
Oxoid (2010) Dehydrated culture media: MRS broth (De Man, Rogosa, Sharpe). [Online]. Available at http://www.oxoid.com/UK/blue/prod_detail/prod_detail.asp?pr=CM0359&c=UK&lang=EN (accessed on February 1st 2011)
Yu LC, Flynn AN, Turner JR, Buret AG (2005) SGLT-1-mediated glucose uptake protects intestinal epithelial cells against LPS-induced apoptosis and barrier defects: a novel cellular rescue mechanism? FASEB J 19:1822–1835
Conflict of interest
I, Luca Prisciandaro, state that there is no conflict of interest associated with this manuscript. I declare that the current work was not financially sponsored by any organization. The authorship of this manuscript is as described in the attached Authorship/Disclosure form, with myself as the first author. I have a full control of all primary data related to this manuscript and would be happy to allow the journal to review this data if necessary.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prisciandaro, L.D., Geier, M.S., Chua, A.E. et al. Probiotic factors partially prevent changes to caspases 3 and 7 activation and transepithelial electrical resistance in a model of 5-fluorouracil-induced epithelial cell damage. Support Care Cancer 20, 3205–3210 (2012). https://doi.org/10.1007/s00520-012-1446-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-012-1446-3