Abstract
Purpose
This “consensus statement” aims to summarise the current evidence-based knowledge as to “timing” and planning the “extent“ of thyroid surgery in terms of an optimal balance between the prevention of thyroid malignancy (involving metastasis) and the risks associated with more extended surgery (permanent hypoparathyroidism, permanent paralysis of the recurrent laryngeal nerve). Surgery “in time” is influenced by genetic findings and age. Basal (and stimulated) calcitonin levels may individualise the timing and extent of surgery.
Materials and methods
The review of English-language studies addressing the management of REarranged during Transfection proto-oncogene mutation carriers including the time, extent of thyroid surgery and results. Evidence is mostly obtained from well-designed, non-experimental descriptive investigations, such as comparative, correlation and case–control studies (level III) with a grading of recommendation B, or from expert committee reports or opinions and/or the clinical experience of respected authorities (level IV) with a grading of recommendation C, respectively.
Results
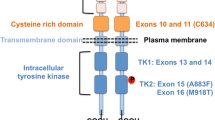
“Risk level D” includes multiple endocrine neoplasia 2B cases. Thyroidectomy is recommended within the first year of life, preferably as soon as possible, due to the very early transformation of C cell hyperplasia to more aggressive tumours. Calcitonin levels may be less helpful. In patients with codon 634 mutations (risk level C), thyroidectomy between ages 2 and 4 years has been proposed based upon evidence of age-dependent and codon-specific progression of early medullary thyroid cancer. In “risk level B” (codons 609, 611, 618, 620, 630 and 804), tandem mutation (804–778) patients should undergo thyroidectomy before the age of 6 years. “Risk level A” includes patients with mutations in codons 321, 515, 533, 600, 603, 606, 635, 649, 666, 768, 776, 790, 791, 804 (single mutation), 833, 844, 861, 891 or 912. Surgery may be postponed until the age of 10 years.
However, postponing surgery and avoiding central (level VI) neck dissection in patients with risk levels A to C are only justified in families with a less aggressive MTC history and in combination with the results of basal (and calcium- or pentagastrin-stimulated) serum calcitonin levels. The moment of transition from C cell hyperplasia to MTC seems to occur when calcitonin levels rise. In patients with normal basal and stimulated calcitonin levels, the chance of micro-MTC increases significantly.
Conclusions
Hereditary C cell disease acts as a model to apply the results of bedside genetic testing, age and calcitonin levels (genotype–age–calcitonin-concept) for the individual timing of thyroid surgery and its extent.
Similar content being viewed by others
References
Dralle H, Musholt TJ, Schabram J, Steinmuller T, Frilling A, Simon D, Goretzki PE, Niederle B, Scheuba C, Clerici T, Hermann M, Kussmann J, Lorenz K, Nies C, Schabram P, Trupka A, Zielke A, Karges W, Luster M, Schmid KW, Vordermark D, Schmoll HJ, Muhlenberg R, Schober O, Rimmele H, Machens A (2013) German Association of Endocrine Surgeons practice guideline for the surgical management of malignant thyroid tumors. Langenbecks Arch Surg 398(3):347–375. doi:10.1007/s00423-013-1057-6
Pacini F, Castagna MG, Brilli L, Pentheroudakis G (2010) Thyroid cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 21(Suppl 5):v214–219. doi:10.1093/annonc/mdq190
British Thyroid Association RCoP (2007) Guidelines for the management of thyroid cancer. http://www.british-thyroid-association.org/news/Docs/Thyroid_cancer_guidelines_2007.pdf
Chen H, Sippel RS, O’Dorisio MS, Vinik AI, Lloyd RV, Pacak K (2010) The North American Neuroendocrine Tumor Society consensus guideline for the diagnosis and management of neuroendocrine tumors: pheochromocytoma, paraganglioma, and medullary thyroid cancer. Pancreas 39(6):775–783. doi:10.1097/MPA.0b013e3181ebb4f0
Tuttle RM, Ball DW, Byrd D, Daniels GH, Dilawari RA, Doherty GM, Duh QY, Ehya H, Farrar WB, Haddad RI, Kandeel F, Kloos RT, Kopp P, Lamonica DM, Loree TR, Lydiatt WM, McCaffrey J, Olson JA Jr, Parks L, Ridge JA, Shah JP, Sherman SI, Sturgeon C, Waguespack SG, Wang TN, Wirth LJ (2010) Medullary carcinoma. J Natl Compr Canc Netw 8(5):512–530
Kloos RT, Eng C, Evans DB, Francis GL, Gagel RF, Gharib H, Moley JF, Pacini F, Ringel MD, Schlumberger M, Wells SA Jr (2009) Medullary thyroid cancer: management guidelines of the American Thyroid Association. Thyroid 19(6):565–612. doi:10.1089/thy.2008.0403
Machens A, Dralle H (2009) Prophylactic thyroidectomy in RET carriers at risk for hereditary medullary thyroid cancer. Thyroid 19(6):551–554. doi:10.1089/thy.2009.0144
Ball DW (2009) American Thyroid Association guidelines for management of medullary thyroid cancer: an adult endocrinology perspective. Thyroid 19(6):547–550. doi:10.1089/thy.2009.0147
Brauckhoff M, Gimm O (2009) Extrathyroidal manifestations of multiple endocrine neoplasia type 2. Thyroid 19(6):555–557. doi:10.1089/thy.2009.0151
Waguespack SG (2009) A perspective from pediatric endocrinology on the hereditary medullary thyroid carcinoma syndromes. Thyroid 19(6):543–546. doi:10.1089/thy.2009.0127
Daumerie C, Maiter D, Gruson D (2013) Serum calcitonin estimation in medullary thyroid cancer: basal or stimulated levels? Thyroid Res 6 Suppl 1:S4. doi:10.1186/1756-6614-6-S1-S4
Fugazzola L, De Leo S, Perrino M (2013) The optimal range of RET mutations to be tested: European comments to the guidelines of the American Thyroid Association. Thyroid Res 6 Suppl 1:S8. doi:10.1186/1756-6614-6-S1-S8
Jarzab B, Szpak-Ulczok S, Wloch J, Czarniecka A, Krajewska J (2013) Timing and criteria for prophylactic thyroidectomy in asymptomatic RET carriers—the role of Ct serum level. Thyroid Res 6 Suppl 1:S9. doi:10.1186/1756-6614-6-S1-S9
Elisei R et al (2012) 2012 European Thyroid Association, guidelines for genetic testing and its clinical consequences in medullary thyroid cancer. Eur Thyroid J 1:216–231
Maier RV (2006) What the surgeon of tomorrow needs to know about evidence-based surgery. Arch Surg 141(3):317–323. doi:10.1001/archsurg.141.3.317
Holzer S, Reiners C, Mann K, Bamberg M, Rothmund M, Dudeck J, Stewart AK, Hundahl SA (2000) Patterns of care for patients with primary differentiated carcinoma of the thyroid gland treated in Germany during 1996. U.S. and German Thyroid Cancer Group. Cancer 89(1):192–201. doi:10.1002/1097-0142(20000701)89:1<192::AID-CNCR26>3.0.CO;2-7
Hundahl SA, Cady B, Cunningham MP, Mazzaferri E, McKee RF, Rosai J, Shah JP, Fremgen AM, Stewart AK, Holzer S (2000) Initial results from a prospective cohort study of 5583 cases of thyroid carcinoma treated in the United States during 1996. U.S. and German Thyroid Cancer Study Group. An American College of Surgeons Commission on Cancer Patient Care Evaluation study. Cancer 89(1):202–217. doi:10.1002/1097-0142(20000701)89:1<202::AID-CNCR27>3.0.CO;2-A
Valle LA, Kloos RT (2011) The prevalence of occult medullary thyroid carcinoma at autopsy. J Clin Endocrinol Metab 96(1):E109–113. doi:10.1210/jc.2010-0959
Daniels GH (2011) Screening for medullary thyroid carcinoma with serum calcitonin measurements in patients with thyroid nodules in the United States and Canada. Thyroid 21(11):1199–1207. doi:10.1089/thy.2010.0297
Niccoli P, Wion-Barbot N, Caron P, Henry JF, de Micco C, Saint Andre JP, Bigorgne JC, Modigliani E, Conte-Devolx B (1997) Interest of routine measurement of serum calcitonin: study in a large series of thyroidectomized patients. The French Medullary Study Group. J Clin Endocrinol Metab 82(2):338–341
Ozgen AG, Hamulu F, Bayraktar F, Yilmaz C, Tuzun M, Yetkin E, Tuncyurek M, Kabalak T (1999) Evaluation of routine basal serum calcitonin measurement for early diagnosis of medullary thyroid carcinoma in seven hundred seventy-three patients with nodular goiter. Thyroid 9(6):579–582
Pacini F, Fontanelli M, Fugazzola L, Elisei R, Romei C, Di Coscio G, Miccoli P, Pinchera A (1994) Routine measurement of serum calcitonin in nodular thyroid diseases allows the preoperative diagnosis of unsuspected sporadic medullary thyroid carcinoma. J Clin Endocrinol Metab 78(4):826–829
Papi G, Corsello SM, Cioni K, Pizzini AM, Corrado S, Carapezzi C, Fadda G, Baldini A, Carani C, Pontecorvi A, Roti E (2006) Value of routine measurement of serum calcitonin concentrations in patients with nodular thyroid disease: a multicenter study. J Endocrinol Invest 29(5):427–437
Rieu M, Lame MC, Richard A, Lissak B, Sambort B, Vuong-Ngoc P, Berrod JL, Fombeur JP (1995) Prevalence of sporadic medullary thyroid carcinoma: the importance of routine measurement of serum calcitonin in the diagnostic evaluation of thyroid nodules. Clin Endocrinol (Oxf) 42(5):453–460
Biddinger P, Mukunda, R. (ed) (1993) Distribution of C cells in the normal and diseased thyroid gland. Pathology Annual 1:205-229; Virginia
Perry A, Molberg K, Albores-Saavedra J (1996) Physiologic versus neoplastic C-cell hyperplasia of the thyroid: separation of distinct histologic and biologic entities. Cancer 77(4):750–756. doi:10.1002/(SICI)1097-0142(19960215)77:4<750::AID-CNCR22>3.0.CO;2-Z
Rosai J (ed) (1992) Tumors of the thyroid gland. In: Atlas of tumor pathology. Armed Forces Institute of Pathology, Washington DC
Albores-Saavedra JA, Krueger JE (2001) C-cell hyperplasia and medullary thyroid microcarcinoma. Endocr Pathol 12(4):365–377
LiVolsi VA (1997) C cell hyperplasia/neoplasia. J Clin Endocrinol Metab 82(1):39–41
Kaserer K, Scheuba C, Neuhold N, Weinhausel A, Haas OA, Vierhapper H, Niederle B (2001) Sporadic versus familial medullary thyroid microcarcinoma: a histopathologic study of 50 consecutive patients. Am J Surg Pathol 25(10):1245–1251
Kaserer K, Scheuba C, Neuhold N, Weinhausel A, Vierhapper H, Haas OA, Niederle B (1998) C-cell hyperplasia and medullary thyroid carcinoma in patients routinely screened for serum calcitonin. Am J Surg Pathol 22(6):722–728
Kaserer K, Scheuba C, Neuhold N, Weinhausel A, Vierhapper H, Niederle B (2002) Recommendations for reporting C cell pathology of the thyroid. Wien Klin Wochenschr 114(7):274–278
Iwashita T, Kato M, Murakami H, Asai N, Ishiguro Y, Ito S, Iwata Y, Kawai K, Asai M, Kurokawa K, Kajita H, Takahashi M (1999) Biological and biochemical properties of Ret with kinase domain mutations identified in multiple endocrine neoplasia type 2B and familial medullary thyroid carcinoma. Oncogene 18(26):3919–3922. doi:10.1038/sj.onc.1202742
Elisei R, Cosci B, Romei C, Bottici V, Renzini G, Molinaro E, Agate L, Vivaldi A, Faviana P, Basolo F, Miccoli P, Berti P, Pacini F, Pinchera A (2008) Prognostic significance of somatic RET oncogene mutations in sporadic medullary thyroid cancer: a 10-year follow-up study. J Clin Endocrinol Metab 93(3):682–687. doi:10.1210/jc.2007-1714
Bugalho MJ, Domingues R, Santos JR, Catarino AL, Sobrinho L (2007) Mutation analysis of the RET proto-oncogene and early thyroidectomy: results of a Portuguese cancer centre. Surgery 141(1):90–95. doi:10.1016/j.surg.2006.03.025
Elisei R, Romei C, Cosci B, Agate L, Bottici V, Molinaro E, Sculli M, Miccoli P, Basolo F, Grasso L, Pacini F, Pinchera A (2007) RET genetic screening in patients with medullary thyroid cancer and their relatives: experience with 807 individuals at one center. J Clin Endocrinol Metab 92(12):4725–4729. doi:10.1210/jc.2007-1005
Eng C, Mulligan LM, Smith DP, Healey CS, Frilling A, Raue F, Neumann HP, Ponder MA, Ponder BA (1995) Low frequency of germline mutations in the RET proto-oncogene in patients with apparently sporadic medullary thyroid carcinoma. Clin Endocrinol (Oxf) 43(1):123–127
Romei C, Cosci B, Renzini G, Bottici V, Molinaro E, Agate L, Passannanti P, Viola D, Biagini A, Basolo F, Ugolini C, Materazzi G, Pinchera A, Vitti P, Elisei R (2011) RET genetic screening of sporadic medullary thyroid cancer (MTC) allows the preclinical diagnosis of unsuspected gene carriers and the identification of a relevant percentage of hidden familial MTC (FMTC). Clin Endocrinol (Oxf) 74(2):241–247. doi:10.1111/j.1365-2265.2010.03900.x
Schuffenecker I, Ginet N, Goldgar D, Eng C, Chambe B, Boneu A, Houdent C, Pallo D, Schlumberger M, Thivolet C, Lenoir GM (1997) Prevalence and parental origin of de novo RET mutations in multiple endocrine neoplasia type 2A and familial medullary thyroid carcinoma. Le Groupe d’Etude des Tumeurs a Calcitonine. Am J Hum Genet 60(1):233–237
Wiench M, Wygoda Z, Gubala E, Wloch J, Lisowska K, Krassowski J, Scieglinska D, Fiszer-Kierzkowska A, Lange D, Kula D, Zeman M, Roskosz J, Kukulska A, Krawczyk Z, Jarzab B (2001) Estimation of risk of inherited medullary thyroid carcinoma in apparent sporadic patients. J Clin Oncol 19(5):1374–1380
Scheuba C, Kaserer K, Moritz A, Drosten R, Vierhapper H, Bieglmayer C, Haas OA, Niederle B (2009) Sporadic hypercalcitoninemia: clinical and therapeutic consequences. Endocr Relat Cancer 16(1):243–253. doi:10.1677/ERC-08-0059
Wolfe HJ, Melvin KE, Cervi-Skinner SJ, Saadi AA, Juliar JF, Jackson CE, Tashjian AH Jr (1973) C-cell hyperplasia preceding medullary thyroid carcinoma. N Engl J Med 289(9):437–441. doi:10.1056/NEJM197308302890901
McDermott MB, Swanson PE, Wick MR (1995) Immunostains for collagen type IV discriminate between C-cell hyperplasia and microscopic medullary carcinoma in multiple endocrine neoplasia, type 2a. Hum Pathol 26(12):1308–1312
Wells SA Jr, Pacini F, Robinson BG, Santoro M (2013) Multiple endocrine neoplasia type 2 and familial medullary thyroid carcinoma: an update. J Clin Endocrinol Metab 98(8):3149–3164. doi:10.1210/jc.2013-1204
Machens A, Lorenz K, Sekulla C, Hoppner W, Frank-Raue K, Raue F, Dralle H (2013) Molecular epidemiology of multiple endocrine neoplasia 2: implications for RET screening in the new millenium. Eur J Endocrinol 168(3):307–314. doi:10.1530/EJE-12-0919
Lips CJ, Landsvater RM, Hoppener JW, Geerdink RA, Blijham G, van Veen JM, van Gils AP, de Wit MJ, Zewald RA, Berends MJ et al (1994) Clinical screening as compared with DNA analysis in families with multiple endocrine neoplasia type 2A. N Engl J Med 331(13):828–835. doi:10.1056/NEJM199409293311302
Machens A, Dralle H (2007) Genotype-phenotype based surgical concept of hereditary medullary thyroid carcinoma. World J Surg 31(5):957–968. doi:10.1007/s00268-006-0769-y
Wells SA Jr, Chi DD, Toshima K, Dehner LP, Coffin CM, Dowton SB, Ivanovich JL, DeBenedetti MK, Dilley WG, Moley JF et al (1994) Predictive DNA testing and prophylactic thyroidectomy in patients at risk for multiple endocrine neoplasia type 2A. Ann Surg 220(3):237–247, discussion 247–250
Eng C, Clayton D, Schuffenecker I, Lenoir G, Cote G, Gagel RF, van Amstel HK, Lips CJ, Nishisho I, Takai SI, Marsh DJ, Robinson BG, Frank-Raue K, Raue F, Xue F, Noll WW, Romei C, Pacini F, Fink M, Niederle B, Zedenius J, Nordenskjold M, Komminoth P, Hendy GN, Mulligan LM et al (1996) The relationship between specific RET proto-oncogene mutations and disease phenotype in multiple endocrine neoplasia type 2. International RET mutation consortium analysis. JAMA 276(19):1575–1579
Raue F, Frank-Raue K (2012) Genotype–phenotype correlation in multiple endocrine neoplasia type 2. Clinics (Sao Paulo) 67 Suppl 1:69–75
Waguespack SG, Rich TA, Perrier ND, Jimenez C, Cote GJ (2011) Management of medullary thyroid carcinoma and MEN2 syndromes in childhood. Nat Rev Endocrinol 7(10):596–607. doi:10.1038/nrendo.2011.139
Schuffenecker I, Virally-Monod M, Brohet R, Goldgar D, Conte-Devolx B, Leclerc L, Chabre O, Boneu A, Caron J, Houdent C, Modigliani E, Rohmer V, Schlumberger M, Eng C, Guillausseau PJ, Lenoir GM (1998) Risk and penetrance of primary hyperparathyroidism in multiple endocrine neoplasia type 2A families with mutations at codon 634 of the RET proto-oncogene. Groupe D’etude des Tumeurs a Calcitonine. J Clin Endocrinol Metab 83(2):487–491
Scholten A, Schreinemakers JM, Pieterman CR, Valk GD, Vriens MR, Borel Rinkes IH (2011) Evolution of surgical treatment of primary hyperparathyroidism in patients with multiple endocrine neoplasia type 2A. Endocr Pract 17(1):7–15. doi:10.4158/EP10050.OR
Brauckhoff M, Gimm O, Weiss CL, Ukkat J, Sekulla C, Brauckhoff K, Thanh PN, Dralle H (2004) Multiple endocrine neoplasia 2B syndrome due to codon 918 mutation: clinical manifestation and course in early and late onset disease. World J Surg 28(12):1305–1311. doi:10.1007/s00268-004-7637-4
Brauckhoff M, Machens A, Lorenz K, Bjoro T, Varhaug JE, Dralle H (2013) Surgical curability of medullary thyroid cancer in multiple endocrine neoplasia 2B: a changing perspective. Ann Surg. doi:10.1097/SLA.0b013e3182a6f43a
Brauckhoff M, Machens A, Hess S, Lorenz K, Gimm O, Brauckhoff K, Sekulla C, Dralle H (2008) Premonitory symptoms preceding metastatic medullary thyroid cancer in MEN 2B: an exploratory analysis. Surgery 144(6):1044–1050. doi:10.1016/j.surg.2008.08.028, discussion 1050–1043
Brandi ML, Gagel RF, Angeli A, Bilezikian JP, Beck-Peccoz P, Bordi C, Conte-Devolx B, Falchetti A, Gheri RG, Libroia A, Lips CJ, Lombardi G, Mannelli M, Pacini F, Ponder BA, Raue F, Skogseid B, Tamburrano G, Thakker RV, Thompson NW, Tomassetti P, Tonelli F, Wells SA Jr, Marx SJ (2001) Guidelines for diagnosis and therapy of MEN type 1 and type 2. J Clin Endocrinol Metab 86(12):5658–5671
Machens A, Niccoli-Sire P, Hoegel J, Frank-Raue K, van Vroonhoven TJ, Roeher HD, Wahl RA, Lamesch P, Raue F, Conte-Devolx B, Dralle H (2003) Early malignant progression of hereditary medullary thyroid cancer. N Engl J Med 349(16):1517–1525. doi:10.1056/NEJMoa012915
Machens A, Holzhausen HJ, Thanh PN, Dralle H (2003) Malignant progression from C-cell hyperplasia to medullary thyroid carcinoma in 167 carriers of RET germline mutations. Surgery 134(3):425–431
Schreinemakers JM, Vriens MR, Valk GD, de Groot JW, Plukker JT, Bax K, Hamming JF, van der Luijt RB, Aronson DC, Borel Rinkes IH (2010) Factors predicting outcome of total thyroidectomy in young patients with multiple endocrine neoplasia type 2: a nationwide long-term follow-up study. World J Surg 34(4):852–860. doi:10.1007/s00268-009-0370-2
Shepet K, Alhefdhi A, Lai N, Mazeh H, Sippel R, Chen H (2013) Hereditary medullary thyroid cancer: age-appropriate thyroidectomy improves disease-free survival. Ann Surg Oncol 20(5):1451–1455. doi:10.1245/s10434-012-2757-9
Toledo SP, Lourenco DM Jr, Santos MA, Tavares MR, Toledo RA, Correia-Deur JE (2009) Hypercalcitoninemia is not pathognomonic of medullary thyroid carcinoma. Clinics (Sao Paulo) 64(7):699–706. doi:10.1590/S1807-59322009000700015
Kratzsch J, Petzold A, Raue F, Reinhardt W, Brocker-Preuss M, Gorges R, Mann K, Karges W, Morgenthaler N, Luster M, Reiners C, Thiery J, Dralle H, Fuhrer D (2011) Basal and stimulated calcitonin and procalcitonin by various assays in patients with and without medullary thyroid cancer. Clin Chem 57(3):467–474. doi:10.1373/clinchem.2010.151688
Machens A, Haedecke J, Holzhausen HJ, Thomusch O, Schneyer U, Dralle H (2000) Differential diagnosis of calcitonin-secreting neuroendocrine carcinoma of the foregut by pentagastrin stimulation. Langenbecks Arch Surg 385(6):398–401
Bieglmayer C, Scheuba C, Niederle B, Flores J, Vierhapper H (2002) Screening for medullary thyroid carcinoma: experience with different immunoassays for human calcitonin. Wien Klin Wochenschr 114(7):267–273
Bieglmayer C, Vierhapper H, Dudczak R, Niederle B (2007) Measurement of calcitonin by immunoassay analyzers. Clin Chem Lab Med 45(5):662–666. doi:10.1515/CCLM.2007.124
Basuyau JP, Mallet E, Leroy M, Brunelle P (2004) Reference intervals for serum calcitonin in men, women, and children. Clin Chem 50(10):1828–1830. doi:10.1373/clinchem.2003.026963
Verga U, Morpurgo PS, Vaghi I, Radetti G, Beck-Peccoz P (2006) Normal range of calcitonin in children measured by a chemiluminescent two-site immunometric assay. Horm Res 66(1):17–20. doi:10.1159/000092848
Pina G, Dubois S, Murat A, Berger N, Niccoli P, Peix JL, Cohen R, Guillausseau C, Charrie A, Chabre O, Cornu C, Borson-Chazot F, Rohmer V (2013) Is basal ultrasensitive measurement of calcitonin capable of substituting for the pentagastrin-stimulation test? Clin Endocrinol (Oxf) 78(3):358–364. doi:10.1111/cen.12001
Colombo C, Verga U, Mian C, Ferrero S, Perrino M, Vicentini L, Dazzi D, Opocher G, Pelizzo MR, Beck-Peccoz P, Fugazzola L (2012) Comparison of calcium and pentagastrin tests for the diagnosis and follow-up of medullary thyroid cancer. J Clin Endocrinol Metab 97(3):905–913. doi:10.1210/jc.2011-2033
Machens A, Dralle H (2010) Biomarker-based risk stratification for previously untreated medullary thyroid cancer. J Clin Endocrinol Metab 95(6):2655–2663. doi:10.1210/jc.2009-2368
Scheuba C, Kaserer K, Weinhausl A, Pandev R, Kaider A, Passler C, Prager G, Vierhapper H, Haas OA, Niederle B (1999) Is medullary thyroid cancer predictable? A prospective study of 86 patients with abnormal pentagastrin tests. Surgery 126(6):1089–1095, discussion 1096
Machens A, Lorenz K, Dralle H (2009) Individualization of lymph node dissection in RET (rearranged during transfection) carriers at risk for medullary thyroid cancer: value of pretherapeutic calcitonin levels. Ann Surg 250(2):305–310. doi:10.1097/SLA.0b013e3181ae333f
Elisei R, Romei C, Renzini G, Bottici V, Cosci B, Molinaro E, Agate L, Cappagli V, Miccoli P, Berti P, Faviana P, Ugolini C, Basolo F, Vitti P, Pinchera A (2012) The timing of total thyroidectomy in RET gene mutation carriers could be personalized and safely planned on the basis of serum calcitonin: 18 years experience at one single center. J Clin Endocrinol Metab 97(2):426–435. doi:10.1210/jc.2011-2046
Scheuba C, Kaserer K, Bieglmayer C, Asari R, Riss P, Drosten R, Niederle B (2007) Medullary thyroid microcarcinoma recommendations for treatment—a single-center experience. Surgery 142 (6):1003–1010; discussion 1010 e1001-1003. doi:10.1016/j.surg.2007.09.016
Lau GS, Lang BH, Lo CY, Tso A, Garcia-Barcelo MM, Tam PK, Lam KS (2009) Prophylactic thyroidectomy in ethnic Chinese patients with multiple endocrine neoplasia type 2A syndrome after the introduction of genetic testing. Hong Kong Med J 15(5):326–331
Rohmer V, Vidal-Trecan G, Bourdelot A, Niccoli P, Murat A, Wemeau JL, Borson-Chazot F, Schvartz C, Tabarin A, Chabre O, Chabrier G, Caron P, Rodien P, Schlumberger M, Baudin E (2011) Prognostic factors of disease-free survival after thyroidectomy in 170 young patients with a RET germline mutation: a multicenter study of the Groupe Francais d’Etude des Tumeurs Endocrines. J Clin Endocrinol Metab 96(3):E509–518. doi:10.1210/jc.2010-1234
Raval MV, Sturgeon C, Bentrem DJ, Elaraj DM, Stewart AK, Winchester DJ, Ko CY, Reynolds M (2010) Influence of lymph node metastases on survival in pediatric medullary thyroid cancer. J Pediatr Surg 45(10):1947–1954. doi:10.1016/j.jpedsurg.2010.06.013
de Groot JW, Plukker JT, Wolffenbuttel BH, Wiggers T, Sluiter WJ, Links TP (2006) Determinants of life expectancy in medullary thyroid cancer: age does not matter. Clin Endocrinol (Oxf) 65(6):729–736. doi:10.1111/j.1365-2265.2006.02659.x
Morris LF, Waguespack SG, Edeiken-Monroe BS, Lee JE, Rich TA, Ying AK, Warneke CL, Evans DB, Perrier ND, Grubbs EG (2013) Ultrasonography should not guide the timing of thyroidectomy in pediatric patients diagnosed with multiple endocrine neoplasia syndrome 2A through genetic screening. Ann Surg Oncol 20(1):53–59. doi:10.1245/s10434-012-2589-7
Alevizaki M (2013) Management of hyperparathyroidism (PHP) in MEN2 syndromes in Europe. Thyroid Res 6 Suppl 1:S10. doi:10.1186/1756-6614-6-S1-S10
Dralle H. MA, Bracukhoff M. (2012) Syndromic medullary thyroid carcinoma: MEN 2A and MEN 2B. Surgery of the thyroid and parathyroid glands 2nd edition. Philadelphia, PA, Elsevier, Saunders
Frank-Raue K, Buhr H, Dralle H, Klar E, Senninger N, Weber T, Rondot S, Hoppner W, Raue F (2006) Long-term outcome in 46 gene carriers of hereditary medullary thyroid carcinoma after prophylactic thyroidectomy: impact of individual RET genotype. Eur J Endocrinol 155(2):229–236. doi:10.1530/eje.1.02216
Dralle H, Gimm O, Simon D, Frank-Raue K, Gortz G, Niederle B, Wahl RA, Koch B, Walgenbach S, Hampel R, Ritter MM, Spelsberg F, Heiss A, Hinze R, Hoppner W (1998) Prophylactic thyroidectomy in 75 children and adolescents with hereditary medullary thyroid carcinoma: German and Austrian experience. World J Surg 22(7):744–750, discussion 750–741
Gonzalez-Sanchez C, Franch-Arcas G, Gomez-Alonso A (2013) Morbidity following thyroid surgery: does surgeon volume matter? Langenbecks Arch Surg 398(3):419–422. doi:10.1007/s00423-012-1027-4
Stavrakis AI, Ituarte PH, Ko CY, Yeh MW (2007) Surgeon volume as a predictor of outcomes in inpatient and outpatient endocrine surgery. Surgery 142(6):887–899. doi:10.1016/j.surg.2007.09.003, discussion 887–899
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niederle, B., Sebag, F. & Brauckhoff, M. Timing and extent of thyroid surgery for gene carriers of hereditary C cell disease—a consensus statement of the European Society of Endocrine Surgeons (ESES). Langenbecks Arch Surg 399, 185–197 (2014). https://doi.org/10.1007/s00423-013-1139-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-013-1139-5