Abstract
Purpose
Aim of this study was to compare the diagnostic performance of PI-RADS version 1 (v1) and version 2 (v2) in the detection of prostate cancer (PCa).
Methods
Multiparametric MRIs (mpMRI) of 50 consecutive patients with biopsy proven PCa, which had originally been evaluated according to PIRADS v1, were now retrospectively re-evaluated, comparing PI-RADS v1 and v2. MpMRI data were evaluated in comparison with histopathological whole-mount step-section slides. MRI examinations included T2-weighted, diffusion-weighted, and dynamic contrast-enhanced MRI.
Results
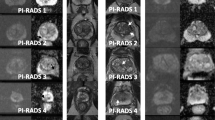
Overall PI-RADS v1 showed a significantly larger discriminative ability of tumor detection: PI-RADS v1 AUC 0.96 (95 % CI 0.94–0.98) and v2 AUC 0.90 (95 % CI 0.86–0.94). For peripheral zone lesions, PI-RADS v1 showed a significantly larger ability of PCa discrimination: v1 AUC 0.97 (95 % CI 0.95–0.99) and v2 AUC 0.92 (95 % CI 0.88–0.96). For transition zone lesions, PI-RADS v1 showed more discrimination: v1 AUC 0.96 (95 % CI 0.92–1.00) and v2 0.90 (95 % CI 0.83–0.97), but the difference was not significant. PI-RADS v2 resulted in significantly more false negative results (3 % in v1, 14 % in v2) and a comparable number of true positive results (82 % in v1, 80 % in v2).
Conclusion
PI-RADS v2 uses a simplified approach, but shows a lower diagnostic accuracy. This could lead to a higher rate of false negative results with the risk of missing tumors within low PI-RADS score levels. Therefore, its use cannot be recommended unconditionally, and further improvement should be considered.



Similar content being viewed by others
References
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, Mason M, Matveev V, Wiegel T, Zattoni F, Mottet N (2014) EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol 65(1):124–137. doi:10.1016/j.eururo.2013.09.046
Moore CM, Robertson NL, Arsanious N, Middleton T, Villers A, Klotz L, Taneja SS, Emberton M (2013) Image-guided prostate biopsy using magnetic resonance imaging-derived targets: a systematic review. Eur Urol 63(1):125–140. doi:10.1016/j.eururo.2012.06.004
Pummer K, Rieken M, Augustin H, Gutschi T, Shariat SF (2014) Innovations in diagnostic imaging of localized prostate cancer. World J Urol 32(4):881–890. doi:10.1007/s00345-013-1172-6
Cash H, Maxeiner A, Stephan C, Fischer T, Durmus T, Holzmann J, Asbach P, Haas M, Hinz S, Neymeyer J, Miller K, Gunzel K, Kempkensteffen C (2015) The detection of significant prostate cancer is correlated with the Prostate Imaging Reporting and Data System (PI-RADS) in MRI/transrectal ultrasound fusion biopsy. World J Urol. doi:10.1007/s00345-015-1671-8
Barentsz JO, Richenberg J, Clements R, Choyke P, Verma S, Villeirs G, Rouviere O, Logager V, Futterer JJ, European Society of Urogenital R (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22(4):746–757. doi:10.1007/s00330-011-2377-y
Radiology ACo MR Prostate Imaging Reporting and Data System version 2.0. Accessed April 2015, from http://www.acr.org/Quality-Safety/Resources/PIRADS/
Weinreb JC, Barentsz JO, Choyke PL, Cornud F, Haider MA, Macura KJ, Margolis D, Schnall MD, Shtern F, Tempany CM, Thoeny HC, Verma S (2015) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol. doi:10.1016/j.eururo.2015.08.052
Barentsz JO, Weinreb JC, Verma S, Thoeny HC, Tempany CM, Shtern F, Padhani AR, Margolis D, Macura KJ, Haider MA, Cornud F, Choyke PL (2015) Synopsis of the PI-RADS v2 guidelines for multiparametric prostate magnetic resonance imaging and recommendations for use. Eur Urol. doi:10.1016/j.eururo.2015.08.038
Rothke M, Blondin D, Schlemmer HP, Franiel T (2013) PI-RADS classification: structured reporting for MRI of the prostate. Rofo 185(3):253–261. doi:10.1055/s-0032-1330270
Portalez D, Mozer P, Cornud F, Renard-Penna R, Misrai V, Thoulouzan M, Malavaud B (2012) Validation of the European Society of Urogenital Radiology scoring system for prostate cancer diagnosis on multiparametric magnetic resonance imaging in a cohort of repeat biopsy patients. Eur Urol 62(6):986–996. doi:10.1016/j.eururo.2012.06.044
Roethke MC, Kuru TH, Schultze S, Tichy D, Kopp-Schneider A, Fenchel M, Schlemmer HP, Hadaschik BA (2014) Evaluation of the ESUR PI-RADS scoring system for multiparametric MRI of the prostate with targeted MR/TRUS fusion-guided biopsy at 3.0 Tesla. Eur Radiol 24(2):344–352. doi:10.1007/s00330-013-3017-5
Junker D, Schafer G, Edlinger M, Kremser C, Bektic J, Horninger W, Jaschke W, Aigner F (2013) Evaluation of the PI-RADS scoring system for classifying mpMRI findings in men with suspicion of prostate cancer. Biomed Res Int 2013:252939. doi:10.1155/2013/252939
Schimmoller L, Quentin M, Arsov C, Hiester A, Kropil P, Rabenalt R, Albers P, Antoch G, Blondin D (2014) Predictive power of the ESUR scoring system for prostate cancer diagnosis verified with targeted MR-guided in-bore biopsy. Eur J Radiol 83(12):2103–2108. doi:10.1016/j.ejrad.2014.08.006
Hamoen EH, de Rooij M, Witjes JA, Barentsz JO, Rovers MM (2015) Use of the Prostate Imaging Reporting and Data System (PI-RADS) for prostate cancer detection with multiparametric magnetic resonance imaging: a diagnostic meta-analysis. Eur Urol 67(6):1112–1121. doi:10.1016/j.eururo.2014.10.033
Junker D, Quentin M, Nagele U, Edlinger M, Richenberg J, Schaefer G, Ladurner M, Jaschke W, Horninger W, Aigner F (2015) Evaluation of the PI-RADS scoring system for mpMRI of the prostate: a whole-mount step-section analysis. World J Urol 33(7):1023–1030. doi:10.1007/s00345-014-1370-x
Rosenkrantz AB, Kim S, Campbell N, Gaing B, Deng FM, Taneja SS (2015) Transition zone prostate cancer: revisiting the role of multiparametric MRI at 3 T. AJR Am J Roentgenol 204(3):W266–W272. doi:10.2214/AJR.14.12955
Muller BG, Shih JH, Sankineni S, Marko J, Rais-Bahrami S, George A, de la Rosette JJ, Merino MJ, Wood BJ, Pinto P, Choyke PL, Turkbey B (2015) Prostate cancer: interobserver agreement and accuracy with the revised prostate imaging reporting and data system at multiparametric MR imaging. Radiology. doi:10.1148/radiol.2015142818
Baur AD, Maxeiner A, Franiel T, Kilic E, Huppertz A, Schwenke C, Hamm B, Durmus T (2014) Evaluation of the prostate imaging reporting and data system for the detection of prostate cancer by the results of targeted biopsy of the prostate. Invest Radiol 49(6):411–420. doi:10.1097/RLI.0000000000000030
Tan CH, Hobbs BP, Wei W, Kundra V (2015) Dynamic contrast-enhanced MRI for the detection of prostate cancer: meta-analysis. AJR Am J Roentgenol 204(4):W439–W448. doi:10.2214/AJR.14.13373
Vos EK, Litjens GJ, Kobus T, Hambrock T, Hulsbergen-van de Kaa CA, Barentsz JO, Huisman HJ, Scheenen TW (2013) Assessment of prostate cancer aggressiveness using dynamic contrast-enhanced magnetic resonance imaging at 3 T. Eur Urol 64(3):448–455. doi:10.1016/j.eururo.2013.05.045
Puech P, Sufana-Iancu A, Renard B, Lemaitre L (2013) Prostate MRI: can we do without DCE sequences in 2013? Diagn Interv Imaging 94(12):1299–1311. doi:10.1016/j.diii.2013.09.010
Rosenkrantz AB, Sabach A, Babb JS, Matza BW, Taneja SS, Deng FM (2013) Prostate cancer: comparison of dynamic contrast-enhanced MRI techniques for localization of peripheral zone tumor. AJR Am J Roentgenol 201(3):W471–W478. doi:10.2214/AJR.12.9737
Authors’ contribution
T Auer was involved in data management and wrote the manuscript. M Edlinger and U Nagele were involved in data analysis and edited the manuscript. J Bektic was involved in data collection. TRW Herrmann was edited the manuscript. G Schäfer was involved in data management. F Aigner was involved in project development. D Junker was involved in project development, data collection and management, data analysis, manuscript writing and editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None, nothing to disclose.
Ethical standards
The current manuscript presents a retrospective re-evaluation of patient data. All patient data had originally been collected in the course of a prospective study, which was approved by the local ethics committee in Innsbruck (Study number: AN4468 304/4.17) and has been published in WJUR (Junker et al. [15]). The original study as well as the current retrospective re-evaluation was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
All patients gave their informed consent prior to their inclusion.
Rights and permissions
About this article
Cite this article
Auer, T., Edlinger, M., Bektic, J. et al. Performance of PI-RADS version 1 versus version 2 regarding the relation with histopathological results. World J Urol 35, 687–693 (2017). https://doi.org/10.1007/s00345-016-1920-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-016-1920-5