Abstract
Purpose
This study evaluated the baseline patient characteristics associated with the time to biochemical progression and overall survival in patients who participated in a phase II trial on zoledronic acid combined with the initial androgen-deprivation therapy for treatment-naïve bone-metastatic prostate cancer.
Methods
Patients received zoledronic acid 4 mg intravenously every 4 weeks for up to 24 months, concomitantly started with bicalutamide 80 mg orally every day and goserelin acetate 10.8 mg subcutaneously every 12 weeks.
Results
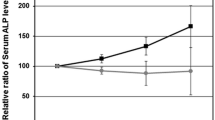
A total of 53 Japanese patients were enrolled between July 2008 and April 2010, and 52 patients were evaluable. Median follow-up period was 41.6 months. Updated median time to biochemical progression was 25.9 months (95 % confidence interval 14.5–49.9). Higher serum bone-specific alkaline phosphatase was an independent risk factor for time to biochemical progression based on multivariate analysis (hazard ratio 6.51; 95 % confidence interval 2.71–15.62; P < 0.001). Median time to biochemical progression for patients with serum bone-specific alkaline phosphatase level higher than 26 μg/L was 12.7 months. Multivariate analysis indicated that higher serum C-terminal telopeptide of type I collagen independently increased the risk of death (hazard ratio 9.62; 95 % confidence interval 2.11–43.89; P = 0.003). Median overall survival for patients with serum C-terminal telopeptide of type I collagen level higher than 8.0 ng/ml was 31.1 months.
Conclusions
Baseline bone markers can be useful as predictors for disease progression and survival time in patients with bone metastasis from treatment-naïve prostate cancer treated with upfront zoledronic acid concomitantly started with androgen-deprivation therapy.


Similar content being viewed by others
References
Coleman RE (2004) Bisphosphonates: clinical experience. Oncologist 9:14–27
Oefelein MG, Ricchiuti V, Conrad W, Resnick MI (2002) Skeletal fractures negatively correlate with overall survival in men with prostate cancer. J Urol 168:1005–1007
Nørgaard M, Jensen AØ, Jacobsen JB, Cetin K, Fryzek JP, Sørensen HT (2010) Skeletal related events, bone metastasis and survival of prostate cancer: a population based cohort study in Denmark (1999 to 2007). J Urol 184:162–167
Sathiakumar N, Delzell E, Morrisey MA et al (2011) Mortality following bone metastasis and skeletal-related events among men with prostate cancer: a population-based analysis of US medicare beneficiaries, 1999–2006. Prostate Cancer Prostatic Dis 14:177–183
Saad F, Gleason DM, Murray R et al (2002) A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst 94:1458–1468
Nozawa M, Inagaki T, Nagao K et al (2014) Phase II trial of zoledronic acid combined with androgen-deprivation therapy for treatment-naïve prostate cancer with bone metastasis. Int J Clin Oncol 19:693–701
Scher HI, Halabi S, Tannock I et al (2008) Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the prostate cancer clinical trials working group. J Clin Oncol 26:1148–1159
Maeda H, Koizumi M, Yoshimura K, Yamauchi T, Kawai T, Ogata E (1997) Correlation between bone metabolic markers and bone scan in prostatic cancer. J Urol 157:539–543
Aruga A, Koizumi M, Hotta R, Takahashi S, Ogata E (1997) Usefulness of bone metabolic markers in the diagnosis and follow-up of bone metastasis from lung cancer. Br J Cancer 76:760–764
Cook RJ, Coleman R, Brown J et al (2006) Markers of bone metabolism and survival in men with hormone-refractory metastatic prostate cancer. Clin Cancer Res 12:3361–3367
Smith MR, Cook RJ, Coleman R et al (2007) Predictors of skeletal complications in men with hormone-refractory metastatic prostate cancer. Urology 70:315–319
Lara PN Jr, Ely B, Quinn DI et al (2014) Serum biomarkers of bone metabolism in castration-resistant prostate cancer patients with skeletal metastases: results from SWOG 0421. J Natl Cancer Inst 106: dju013
Berruti A, Dogliotti L, Gorzegno G et al (1999) Differential patterns of bone turnover in relation to bone pain and disease extent in bone in cancer patients with skeletal metastases. Clin Chem 45:1240–1247
Hollier BG, Evans K, Mani SA (2009) The epithelial-to-mesenchymal transition and cancer stem cells: a coalition against cancer therapies. J Mammary Gland Biol Neoplasia 14:29–43
Lévesque JP, Helwani FM, Winkler IG (2010) The endosteal ‘osteoblastic’ niche and its role in hematopoietic stem cell homing and mobilization. Leukemia 24:1979–1992
Sun Y, Wang BE, Leong KG et al (2012) Androgen deprivation causes epithelial-mesenchymal transition in the prostate: implications for androgen-deprivation therapy. Cancer Res 72:527–536
Fizazi K, Carducci M, Smith M et al (2011) Denosumab versus zoledronic acid for treatment of bone metastases in men with castration-resistant prostate cancer: a randomised, double-blind study. Lancet 377:813–822
Parker C, Nilsson S, Heinrich D et al (2013) Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med 369:213–223
Smith DC, Smith MR, Sweeney C et al (2013) Cabozantinib in patients with advanced prostate cancer: results of a phase II randomized discontinuation trial. J Clin Oncol 31:412–419
Brown JE, Cook RJ, Major P et al (2005) Bone turnover markers as predictors of skeletal complications in prostate cancer, lung cancer, and other solid tumors. J Natl Cancer Inst 97:59–69
Sweeney CJ (2006) ECOG: CHAARTED–ChemoHormonal therapy versus androgen ablation randomized trial for extensive disease in prostate cancer. Clin Adv Hematol Oncol 4:588–590
Tombal B, Borre M, Rathenborg P et al (2014) Enzalutamide monotherapy in hormone-naive prostate cancer: primary analysis of an open-label, single-arm, phase 2 study. Lancet Oncol 15:592–600
Acknowledgments
The authors thank all patients and their families for participating in this study, all investigators and site staff for their contribution, and Marco De Velasco for reviewing the language.
Conflict of interest
The authors have no conflict of interest.
Ethical standard
The study was conducted according to the Declaration of Helsinki with approval from ethics committees at each institution. Written informed consent was obtained from all patients consistent with local requirements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nozawa, M., Hara, I., Matsuyama, H. et al. Significance of baseline bone markers on disease progression and survival in hormone-sensitive prostate cancer with bone metastasis. World J Urol 33, 1263–1268 (2015). https://doi.org/10.1007/s00345-014-1431-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-014-1431-1