Abstract
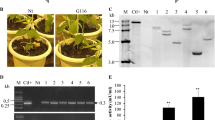
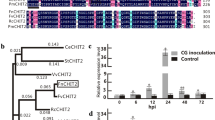
Plant fungal pathogens change their cell wall components during the infection process to avoid degradation by host lytic enzymes, and conversion of the cell wall chitin to chitosan is likely to be one infection strategy of pathogens. Thus, introduction of chitosan-degradation activity into plants is expected to improve fungal disease resistance. Chitosanase has been found in bacteria and fungi, but not in higher plants. Here, we demonstrate that chitosanase, Cho1, from Bacillus circulans MH-K1 has antifungal activity against the rice blast fungus Magnaporthe oryzae. Introduction of the cho1 gene conferred chitosanase activity to rice cells. Transgenic rice plants expressing Cho1 designed to be localized in the apoplast showed increased resistance to M. oryzae accompanied by increased generation of hydrogen peroxide in the infected epidermal cells. These results strongly suggest that chitosan exists in the enzyme-accessible surface of M. oryzae during the infection process and that the enhancement of disease resistance is attributable to the antifungal activity of the secreted Cho1 and to increased elicitation of the host defense response.







Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- GlcN:
-
d-Glucosamine
- GlcNAc:
-
N-Acetyl-d-glucosamine
- PCR:
-
Polymerase chain reaction
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- SDS-PAGE:
-
SDS-polyacrylamide gel electrophoresis
- CBB:
-
Coomassie brilliant blue
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- TLC:
-
Thin-layer chromatography
References
Ando A, Noguchi K, Yanagi M, Shinoyama H, Kagawa Y, Hirata H, Yabuki M, Fujii T (1992) Primary structure of chitosanase produced by Bacillus circulans MH-K1. J Gen Appl Microbiol 38:135–144
Boller T, He SY (2009) Innate immunity in plants: an arms race between pattern recognition receptors in plants and effectors in microbial pathogens. Science 324:742–744
Deising H, Siegrist J (1995) Chitin deacetylase activity of the rust Uromyces viciae-fabae is controlled by fungal morphogenesis. FEMS Microbiol Lett 127:207–211
dos Santos AL, El Gueddari NE, Trombotto S, Moerschbacher BM (2008) Partially acetylated chitosan oligo- and polymers induce an oxidative burst in suspension cultured cells of the gymnosperm Araucaria angustifolia. Biomacromolecules 9:3411–3415
El Gueddari NE, Rauchhaus U, Moerschbacher BM, Deising HB (2002) Developmentally regulated conversion of surface-exposed chitin to chitosan in cell walls of plant pathogenic fungi. New Phytol 156:103–112
El Quakfaoui S, Potvin S, Brzezinski R, Assekin A (1995) A Streptomyces chitosanase is active in transgenic tobacco. Plant Cell Rep 15:222–226
Fujikawa T, Kuga Y, Yano S, Yoshimi A, Tachiki T, Abe K, Nishimura M (2009) Dynamics of cell wall components of Magnaporthe grisea during infectious structure development. Mol Microbiol 73:553–570
Gao X, Ju W, Jung W, Park R (2008) Purification and characterization of chitosanase from Bacillus cereus D-11. Carbphydr Polym 72:513–520
Hendrix B, Stewart J (2003) Chitosanase may enhance anti-fungal defense responses in transgenic tobacco. Discovery 4:27–33
Iriti M, Faoro F (2009) Chitosan as a MAMP, searching for a PRR. Plant Signal Behav 4:66–68
Itoh Y, Takahashi K, Takizawa H, Nikaidou N, Tanaka H, Nishihashi H, Watanabe T, Nishizawa Y (2003) Family 19 chitinase of Streptomyces griseus HUT6037 increases plant resistance to the fungal disease. Biosci Biotechnol Biochem 67:847–855
Kamakura T, Yamaguchi S, Saitoh K, Teraoka T, Yamaguchi I (2002) A novel gene, CBP1, encoding a putative extracellular chitin-binding protein, may play an important role in the hydrophobic surface sensing of Magnaporthe grisea during appressorium differentiation. Mol Plant Microbe Interact 15:437–444
Kankanala P, Czymmek K, Valent B (2007) Roles for rice membrane dynamics and plasmodesmata during biotrophic invasion by the blast fungus. Plant Cell 19:706–724
Kishimoto K, Kouzai Y, Kaku H, Shibuya N, Minami E, Nishizawa Y (2010) Perception of the chitin oligosaccharides contributes to disease resistance to blast fungus Magnaporthe oryzae in rice. Plant J 64:343–354
Kuchitsu K, Yazaki Y, Sakano K, Shibuya N (1997) Transient cytoplasmic pH change and ion fluxes through the plasma memberane in suspension-cultured rice cells triggered by N-acetylchitooligosaccharide elicitor. Plant Cell Physiol 38:1012
Latge JP (2007) The cell wall: a carbohydrate armour for the fungal cell. Mol Microbiol 66:279–290
Mitsutomi M, Kigoh H, Tomita H, Watanabe T (1996) Action patterns of microbial chitinases and chitosanases on partially N-acetylated chitosan. Chitin Enzymol 2:273–284
Mochizuki S, Saitoh K, Minami E, Nishizawa Y (2011) Localization of probe-accessible chitin and characterization of genes encoding chitin-binding domains during rice—Magnaporthe oryzae interactions. J Gen Plant Pathol 77:163–173
Nishizawa Y, Kawakami A, Hibi T, He DY, Shibuya N, Minami E (1999) Regulation of the chitinase gene expression in suspension-cultured rice cells by N-acetylchitooligosaccharides: differences in the signal transduction pathways leading to the activation of elicitor-responsive genes. Plant Mol Biol 39:907–914
Ride J, Barber M (1990) Purification and characterization of multiple forms of endochitinase from wheat leaves. Plant Sci 71:185–197
Saito J, Kita A, Higuchi Y, Nagata Y, Ando A, Miki K (1999) Crystal structure of chitosanase from Bacillus circulans MH-K1 at 1.6-Å resolution and its substrate recognition mechanism. J Biol Chem 274:30818–30825
Saito A, Ooya T, Miyatsuchi D, Fuchigami H, Terakado K, Nakayama SY, Watanabe T, Nagata Y, Ando A (2009) Molecular characterization and antifungal activity of a family 46 chitosanase from Amycolatopsis sp. CsO-2. FEMS Microbiol Lett 293:79–84
Shibuya N, Minami E (2001) Oligosaccharide signaling for defense responses in plant. Physiol Mol Plant Pathol 59:223–233
Tan SC, Tan TK, Wong SM, Khor E (1996) The chitosan yield of zygomycetes at their optimum harvesting time. Carbohydr Poly 30:239–242
Tanabe S, Okada M, Jikumaru Y, Yamane H, Kaku H, Shibuya N, Minami E (2006) Induction of resistance against rice blast fungus in rice plants treated with a potent elicitor, N-acetylchitooligosaccharide. Biosci Biotechnol Biochem 70:1599–1605
Tanabe S, Nishizawa Y, Minami E (2009) Effects of catalase on the accumulation of H2O2 in rice cells inoculated with rice blast fungus, Magnaporthe oryzae. Physiol Plant 137:148–154
Toki S, Hara N, Ono K, Onodera H, Tagiri A, Oka S, Tanaka H (2006) Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J 47:969–976
Torres MA, Jones JD, Dangl JL (2006) Reactive oxygen species signaling in response to pathogens. Plant Physiol 141:373–378
Yabuki M, Uchiyama A, Suzuki K, Ando A, Fujii T (1988) Purification and properties of chitosanase from Bacillus circulans MH-K1. J Gen Appl Microbiol 34:255–270
Yamada A, Shibuya N, Kodama O, Akatsuka T (1993) Induction of phytoalexin formation in suspension-cultured rice cells by N-acetylchitooligosaccharides. Biosci Biotechnol Biochem 57:405–409
Acknowledgments
We thank S. Omiya, A. Kikuchi, A. Kimura, M. Tomita, and R. Tokishige of Chiba University for providing information on chitosanase, recombinant Cho1 protein, and technical advice for detecting chitosanase activity. We also thank M. Nishimura and S. Tanabe of the National Institute of Agrobiological Sciences for fruitful discussions on the alteration of cell wall composition of M. oryzae and for providing technical advice regarding M. oryzae inoculation, respectively; and E. Nakajima, H. Kurano, and K. Iwasaki for producing rice transformants. This work was supported by the Program for Promotion of Basic and Applied Researches for Innovations in Bio-oriented Industry in Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by K. Toriyama.
Rights and permissions
About this article
Cite this article
Kouzai, Y., Mochizuki, S., Saito, A. et al. Expression of a bacterial chitosanase in rice plants improves disease resistance to the rice blast fungus Magnaporthe oryzae . Plant Cell Rep 31, 629–636 (2012). https://doi.org/10.1007/s00299-011-1179-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-011-1179-7