Abstract
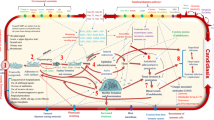
The human fungal pathogen Candida albicans encounters a wide range of pH stresses during its commensal and pathogenic lifestyles. It has been well studied that environmental pH regulates the yeast–filamentous growth transition in this fungus. White–opaque switching is another type of phenotypic transitions in C. albicans. White and opaque cells are two morphologically and functionally distinct cell types, which differ in many aspects including global gene expression profiles, virulence, mating competency, and susceptibility to antifungals. The switch between white and opaque cell types is heritable and epigenetically regulated. In a recently study, Sun et al. (Eukaryot Cell 14:1127–1134, 2015) reported that pH plays a critical role in the regulation of the white–opaque phenotypic switch and sexual mating in C. albicans via both the conserved Rim101-mediated pH sensing and cAMP signaling pathways. The effect of pH on the two biological processes may represent a balancing act between host environmental adaptation and sexual reproduction in this pathogenic fungus.

Similar content being viewed by others
References
Alby K, Bennett RJ (2009) Stress-induced phenotypic switching in Candida albicans. Mol Biol Cell 20:3178–3191. doi:10.1091/mbc.E09-01-0040
Anderson JM, Soll DR (1987) Unique phenotype of opaque cells in the white–opaque transition of Candida albicans. J Bacteriol 169:5579–5588
Barton NH, Charlesworth B (1998) Why sex and recombination? Science 281:1986–1990
Biswas S, Van Dijck P, Datta A (2007) Environmental sensing and signal transduction pathways regulating morphopathogenic determinants of Candida albicans. Microbiol Mol Biol Rev 71:348–376. doi:10.1128/MMBR.00009-06
Brown GD, Denning DW, Gow NA, Levitz SM, Netea MG, White TC (2012) Hidden killers: human fungal infections. Sci Transl Med 4:165rv113. doi:10.1126/scitranslmed.3004404
Davis D (2003) Adaptation to environmental pH in Candida albicans and its relation to pathogenesis. Curr Genet 44:1–7. doi:10.1007/s00294-003-0415-2
Davis DA (2009) How human pathogenic fungi sense and adapt to pH: the link to virulence. Curr Opin Microbiol 12:365–370. doi:10.1016/j.mib.2009.05.006
Davis D, Wilson RB, Mitchell AP (2000) RIM101-dependent and-independent pathways govern pH responses in Candida albicans. Mol Cell Biol 20:971–978
Du H, Guan G, Xie J, Cottier F, Sun Y, Jia W, Muhlschlegel FA, Huang G (2012) The transcription factor Flo8 mediates CO2 sensing in the human fungal pathogen Candida albicans. Mol Biol Cell 23:2692–2701. doi:10.1091/mbc.E12-02-0094
Fonzi WA (1999) PHR1 and PHR2 of Candida albicans encode putative glycosidases required for proper cross-linking of beta-1,3- and beta-1,6-glucans. J Bacteriol 181:7070–7079
Hnisz D, Schwarzmuller T, Kuchler K (2009) Transcriptional loops meet chromatin: a dual-layer network controls white–opaque switching in Candida albicans. Mol Microbiol 74:1–15. doi:10.1111/j.1365-2958.2009.06772.x
Huang G (2012) Regulation of phenotypic transitions in the fungal pathogen Candida albicans. Virulence 3:251–261
Huang G, Wang H, Chou S, Nie X, Chen J, Liu H (2006) Bistable expression of WOR1, a master regulator of white–opaque switching in Candida albicans. Proc Natl Acad Sci USA 103:12813–12818. doi:10.1073/pnas.0605270103
Huang G, Srikantha T, Sahni N, Yi S, Soll DR (2009) CO(2) regulates white-to-opaque switching in Candida albicans. Curr Biol 19:330–334. doi:10.1016/j.cub.2009.01.018
Huang G, Yi S, Sahni N, Daniels KJ, Srikantha T, Soll DR (2010) N-acetylglucosamine induces white to opaque switching, a mating prerequisite in Candida albicans. PLoS Pathog 6:e1000806. doi:10.1371/journal.ppat.1000806
Kararli TT (1995) Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm Drug Dispos 16:351–380
Kvaal C, Lachke SA, Srikantha T, Daniels K, McCoy J, Soll DR (1999) Misexpression of the opaque-phase-specific gene PEP1 (SAP1) in the white phase of Candida albicans confers increased virulence in a mouse model of cutaneous infection. Infect Immun 67:6652–6662
Li M, Martin SJ, Bruno VM, Mitchell AP, Davis DA (2004) Candida albicans Rim13p, a protease required for Rim101p processing at acidic and alkaline pHs. Eukaryot Cell 3:741–751. doi:10.1128/EC.3.3.741-751.2004
Lockhart SR, Pujol C, Daniels KJ, Miller MG, Johnson AD, Pfaller MA, Soll DR (2002) In Candida albicans, white–opaque switchers are homozygous for mating type. Genetics 162:737–745
Lohse MB, Johnson AD (2009) White–opaque switching in Candida albicans. Curr Opin Microbiol 12:650–654. doi:10.1016/j.mib.2009.09.010
Maeda T (2012) The signaling mechanism of ambient pH sensing and adaptation in yeast and fungi. FEBS J 279:1407–1413. doi:10.1111/j.1742-4658.2012.08548.x
Mathé L, Van Dijck P (2013) Recent insights into Candida albicans biofilm resistance mechanisms. Curr Genet 59:251–264
Miller MG, Johnson AD (2002) White–opaque switching in Candida albicans is controlled by mating-type locus homeodomain proteins and allows efficient mating. Cell 110:293–302
Ni M, Feretzaki M, Sun S, Wang X, Heitman J (2011) Sex in fungi. Annu Rev Genet 45:405–430. doi:10.1146/annurev-genet-110410-132536
Prasad R, Singh A (2013) Lipids of Candida albicans and their role in multidrug resistance. Curr Genet 59:243–250
Ramirez-Zavala B, Reuss O, Park YN, Ohlsen K, Morschhauser J (2008) Environmental induction of white–opaque switching in Candida albicans. PLoS Pathog 4:e1000089. doi:10.1371/journal.ppat.1000089
Selvig K, Alspaugh JA (2011) pH response pathways in fungi: adapting to host-derived and environmental signals. Mycobiology 39:249–256. doi:10.5941/MYCO.2011.39.4.249
Slutsky B, Staebell M, Anderson J, Risen L, Pfaller M, Soll DR (1987) “White–opaque transition”: a second high-frequency switching system in Candida albicans. J Bacteriol 169:189–197
Soellick TR, Uhrig JF (2001) Development of an optimized interaction-mating protocol for large-scale yeast two-hybrid analyses. Genome Biol 2:RESEARCH0052
Soll DR (2009) Why does Candida albicans switch? FEMS Yeast Res 9:973–989. doi:10.1111/j.1567-1364.2009.00562.x
Srikantha T, Borneman AR, Daniels KJ, Pujol C, Wu W, Seringhaus MR, Gerstein M, Yi S, Snyder M, Soll DR (2006) TOS9 regulates white–opaque switching in Candida albicans. Eukaryot Cell 5:1674–1687. doi:10.1128/EC.00252-06
Stewart E, Hawser S, Gow NA (1989) Changes in internal and external pH accompanying growth of Candida albicans: studies of non-dimorphic variants. Arch Microbiol 151:149–153
Sudbery PE (2011) Growth of Candida albicans hyphae. Nat Rev Microbiol 9:737–748. doi:10.1038/nrmicro2636
Sun Y, Cao C, Jia W, Tao L, Guan G, Huang G (2015) pH regulates white–opaque switching and sexual mating in Candida albicans. Eukaryot Cell 14:1127–1134
Vylkova S, Carman AJ, Danhof HA, Collette JR, Zhou H, Lorenz MC (2011) The fungal pathogen Candida albicans autoinduces hyphal morphogenesis by raising extracellular pH. MBio 2:e00055-00011. doi:10.1128/mBio.00055-11
Wang Y (2013) Fungal adenylyl cyclase acts as a signal sensor and integrator and plays a central role in interaction with bacteria. PLoS Pathog 9:e1003612. doi:10.1371/journal.ppat.1003612
Whiteway M, Bachewich C (2007) Morphogenesis in Candida albicans. Annu Rev Microbiol 61:529–553. doi:10.1146/annurev.micro.61.080706.093341
Xie J, Tao L, Nobile CJ, Tong Y, Guan G, Sun Y, Cao C, Hernday AD, Johnson AD, Zhang L, Bai FY, Huang G (2013) White–opaque switching in natural MTLa/alpha isolates of Candida albicans: evolutionary implications for roles in host adaptation, pathogenesis, and sex. PLoS Biol 11:e1001525. doi:10.1371/journal.pbio.1001525
Yanagishima N, Fujimura H (1981) Sex pheromones of the yeast Hansenula wingei and their relationship to sex pheromones in Saccharomyces cerevisiae and Saccharomyces kluyveri. Arch Microbiol 129:281–284
Zordan RE, Galgoczy DJ, Johnson AD (2006) Epigenetic properties of white–opaque switching in Candida albicans are based on a self-sustaining transcriptional feedback loop. Proc Natl Acad Sci USA 103:12807–12812. doi:10.1073/pnas.0605138103
Acknowledgments
The authors thank Drs. Mira Edgerton and Yue Wang for their insightful comments on the manuscript. The work in the Huang lab was supported by Grants from the Chinese National Natural Science Foundation (31370175, 31170086, and 81322026 to G.H.) and the “100 Talent Program” Grant from the Chinese Academy of Sciences (to G.H.).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Rights and permissions
About this article
Cite this article
Du, H., Huang, G. Environmental pH adaption and morphological transitions in Candida albicans . Curr Genet 62, 283–286 (2016). https://doi.org/10.1007/s00294-015-0540-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-015-0540-8