Abstract
Purpose
SN 28049 is a new DNA-binding topoisomerase II poison identified by its curative activity against the murine colon 38 carcinoma. Previous studies showed activity to be associated with selective drug accumulation and retention in tumour tissue. Retention varied widely among different tumours and was related to antitumour activity. We determined whether differences in the uptake and retention of SN 28049 could be observed in vitro.
Methods
The Co38P and LLTC lines were derived from the murine colon 38 carcinoma and Lewis lung carcinoma (3LL), respectively. The NZM4, NZM10 and NZM52 human melanoma lines, as well as the CCRF/CEM, CEM/VLB100 and CEM/E1000 human leukaemia lines were also utilised. Cell-associated drug was measured by liquid chromatography–mass spectrometry, laser-scanning confocal microscopy and fluorescence microscopy. Data for SN 28049 were compared for four SN 28049 analogues, for the structurally related drug N-[2-(dimethylamino)-ethyl]acridine-4-carboxamide (DACA) and for doxorubicin.
Results
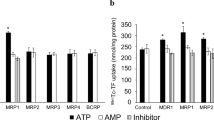
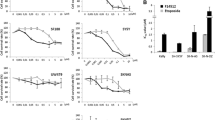
Cellular uptake of SN 28049 was rapid and associated with increased fluorescence in cytoplasmic vesicles or bodies. SN 28049 uptake after an incubation time of 1 h varied widely with different cell lines (2–98 pmol/106 cells) and did not correlate with growth inhibitory concentrations (IC50 values), which also varied widely (1.2–19 nM). Changes in the length of the N-linked side chain of SN 28049 had large effects on drug uptake by Co38P cells. SN 28049 uptake by CCRF/CEM cells was only slightly affected by the expression of P-glycoprotein (CEM/VLB100) or MRP1 protein (CEM/E1000). As measured by cytoplasmic fluorescence, SN 28049 was taken up rapidly and retained strongly by Co38P cells, DACA was taken up rapidly and retained poorly, and doxorubicin was taken up slowly and retained moderately.
Conclusions
The results suggest that SN 28049 is actively transported into cytoplasmic vesicles. While vesicle-associated drug is not important for intrinsic cytotoxicity, it may play a key role as a “slow release” form that modifies pharmacokinetics in multicellular structures such as tumours.









Similar content being viewed by others
References
Smalley KS, Flaherty KT (2009) Integrating BRAF/MEK inhibitors into combination therapy for melanoma. Br J Cancer 100:431–435
Brown JM, Wilson WR (2004) Exploiting tumor hypoxia in cancer treatment. Nat Rev Cancer 4:437–447
Deady LW, Rodemann T, Zhuang L, Baguley BC, Denny WA (2003) Synthesis and cytotoxic activity of carboxamide derivatives of benzo[b][1,6]naphthyridines. J Med Chem 46:1049–1054
Deady LW, Rogers ML, Zhuang L, Baguley BC, Denny WA (2005) Synthesis and cytotoxic activity of carboxamide derivatives of benzo[b][1,6]naphthyridin-(5H)ones. Bioorg Med Chem 13:1341–1355
Bridewell DJ, Porter AC, Finlay GJ, Baguley BC (2008) The role of topoisomerases and RNA transcription in the action of the antitumour benzonaphthyridine derivative SN 28049. Cancer Chemother Pharmacol 62:753–762
Drummond CJ, Finlay GJ, Broome L, Marshall ES, Richardson E, Baguley BC (2011) Action of SN 28049, a new DNA binding topoisomerase II-directed antitumour drug: comparison with doxorubicin and etoposide. Invest New Drugs 29:1102–1110
Chen YY, Finlay GJ, Kirker JA, Marshall ES, Richardson E, Baguley BC (2011) In vivo and in vitro assessment of the action of SN 28049, a benzonaphthyridine derivative targeting topoisomerase II, on the murine Colon 38 carcinoma. Invest New Drugs 29:1504–1510
Muggia FM, Green MD (1991) New anthracycline antitumor antibiotics. Crit Rev Oncol Hematol 11:43–64
Nelson EM, Tewey K, Liu LF (1984) Mechanism of antitumor drug action: poisoning of mammalian DNA topoisomerase II on DNA by 4′-(9-acridinylamino)-methanesulfon-m-anisidide. Proc Natl Acad Sci USA 81:1361–1365
Tewey KM, Rowe TC, Yang L, Halligan BD, Liu LF (1984) Adriamycin-induced DNA damage mediated by mammalian DNA topoisomerase II. Science (New York, NY) 226:466-468
Atwell GJ, Rewcastle GW, Baguley BC, Denny WA (1987) Potential antitumor agents. 50. In vivo solid-tumor activity of derivatives of N-[2-(dimethylamino)ethyl]acridine-4-carboxamide. J Med Chem 30:664–669
Finlay GJ, Riou JF, Baguley BC (1996) From amsacrine to DACA (N-[2-(dimethylamino)ethyl]acridine-4-carboxamide): selectivity for topoisomerases I and II among acridine derivatives. Eur J Cancer 32A:708–714
Davey RA, Su GM, Hargrave RM, Harvie RM, Baguley BC, Davey MW (1997) The potential of N-[2-(dimethylamino)ethyl]acridine-4-carboxamide to circumvent three multidrug-resistance phenotypes in vitro. Cancer Chemother Pharmacol 39:424–430
McCrystal MR, Evans BD, Harvey VJ, Thompson PI, Porter DJ, Baguley BC (1999) Phase I study of the cytotoxic agent N-[2-(dimethylamino)ethyl]acridine-4-carboxamide. Cancer Chemother Pharmacol 44:39–44
Twelves C, Campone M, Coudert B, Van den BM, de Jonge M, Dittrich C, Rampling R, Sorio R, Lacombe D, de Balincourt C, Fumoleau P (2002) Phase II study of XR5000 (DACA) administered as a 120-h infusion in patients with recurrent glioblastoma multiforme. Ann Oncol 13:777–780
Baguley BC (2012) The development of new DNA intercalating anti-cancer drugs. In: Watanabe HS (ed) Horizons in cancer research, vol 48. Nova Publishers, pp 47–65. ISBN: 978-1-62100-524-7
Baguley BC, Zhuang L, Marshall E (1995) Experimental solid tumour activity of N-[2-(dimethylamino)ethyl]- acridine-4-carboxamide. Cancer Chemother Pharmacol 36:244–248
Lukka PB, Paxton JW, Kestell P, Baguley BC (2012) Comparison of a homologous series of benzonaphthyridine anti-cancer agents in mice: divergence between tumour and plasma pharmacokinetics. Cancer Chemother Pharmacol 70:151–160
Lukka PB, Chen YY, Finlay GJ, Joseph WR, Richardson E, Paxton JW, Baguley BC (2013) Tumour tissue selectivity in the uptake of SN 28049, a new topoisomerase II-directed anticancer agent. Cancer Chemother Pharmacol 72:1013–1022
Stones CJ, Kim JE, Joseph WR, Leung E, Marshall ES, Finlay GJ, Shelling AN, Baguley BC (2013) Comparison of responses of human melanoma cell lines to MEK and BRAF inhibitors. Front Genet 4:66–71
Griswold DP, Corbett TH (1975) A colon tumor model for anticancer agent evaluation. Cancer 36:2441–2444
Goldin A, Venditti JM, MacDonald JS, Muggia FM, Henney JE, DeVita VT Jr (1981) Current results of the screening program at the Division of Cancer Treatment, National Cancer Institute. Eur J Cancer 17:129–142
Wilkoff L, Dulmadge E, Chopra D (1980) Viability of cultured Lewis lung cell populations exposed to beta-retinoic acid. Exp Biol Med 163:233–236
Marshall ES, Finlay GJ, Matthews JH, Shaw JH, Nixon J, Baguley BC (1992) Microculture-based chemosensitivity testing: a feasibility study comparing freshly explanted human melanoma cells with human melanoma cell lines. J Natl Cancer Inst 84:340–345
Marshall ES, Holdaway KM, Shaw JH, Finlay GJ, Matthews JH, Baguley BC (1993) Anticancer drug sensitivity profiles of new and established melanoma cell lines. Oncol Res 5:301–309
Kim JE, Stones C, Joseph WR, Leung E, Finlay GJ, Shelling AN, Phillips WA, Shepherd PR, Baguley BC (2012) Comparison of growth factor signalling pathway utilisation in cultured normal melanocytes and melanoma cell lines. BMC Cancer 12:141
Beck WT, Mueller TJ, Tanzer LR (1979) Altered surface membrane glycoproteins in Vinca alkaloid-resistant human leukemic lymphoblasts. Cancer Res 39:2070–2076
Davey RA, Longhurst TJ, Davey MW, Belov L, Harvie RM, Hancox D, Wheeler H (1995) Drug resistance mechanisms and MRP expression in response to epirubicin treatment in a human leukaemia cell line. Leuk Res 19:275–282
Lukka PB, Paxton JW, Atwell GJ, Kestell P, Baguley BC (2012) A rapid LC-MS/MS method for the quantitation of a series of benzonaphthyridine derivatives: application to in vivo pharmacokinetic and lipophilicity studies in drug development. J Pharm Biomed Anal 63:9–16
Haldane A, Finlay GJ, Hay MP, Denny WA, Baguley BC (1999) Cellular uptake of N-[2-(dimethylamino)ethyl]acridine-4-carboxamide (DACA). Anticancer Drug Des 14:275–280
Kazmi F, Hensley T, Pope C, Funk RS, Loewen GJ, Buckley DB, Parkinson A (2013) Lysosomal sequestration (trapping) of lipophilic amine (cationic amphiphilic) drugs in immortalized human hepatocytes (Fa2 N-4 cells). Drug Metab Dispos 41:897–905
Lemieux B, Percival MD, Falgueyret JP (2004) Quantitation of the lysosomotropic character of cationic amphiphilic drugs using the fluorescent basic amine Red DND-99. Anal Biochem 327:247–251
Larsen AK, Escargueil AE, Skladanowski A (2000) Resistance mechanisms associated with altered intracellular distribution of anticancer agents. Pharmacol Ther 85:217–229
Wright PK (2008) Targeting vesicle trafficking: an important approach to cancer chemotherapy. Recent Pat Anticancer Drug Discov 3:137–147
Yamagishi T, Sahni S, Sharp DM, Arvind A, Jansson PJ, Richardson DR (2013) P-glycoprotein mediates drug resistance via a novel mechanism involving lysosomal sequestration. J Biol Chem 288:31761–31771
Lukka PB, Paxton JW, Kestell P, Baguley BC (2010) Pharmacokinetics and distribution of SN 28049, a novel DNA binding anticancer agent, in mice. Cancer Chemother Pharmacol 65:1145–1152
Harris JR, Timberlake N, Henson P, Schimke P, Belli JA (1979) Adriamycin uptake in V79 and Adriamycin resistant Chinese hamster cells. Int J Radiat Oncol Biol Phys 5:1235–1239
Baguley BC (2010) Multidrug resistance in cancer. Methods Mol Biol 596:1–14
Acknowledgments
We are grateful to Cancer Society Auckland, the Lang Foundation and Auckland UniServices Ltd for financial support.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Y.Y., Lukka, P.B., Joseph, W.R. et al. Selective cellular uptake and retention of SN 28049, a new DNA-binding topoisomerase II-directed antitumor agent. Cancer Chemother Pharmacol 74, 25–35 (2014). https://doi.org/10.1007/s00280-014-2469-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-014-2469-x