Abstract
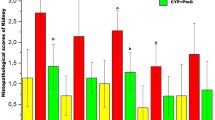
Purpose: Cisplatin (CP)-induced nephrotoxicity is associated with the increased generation of reactive oxygen metabolites and lipid peroxidation in kidney, caused by the decreased levels of antioxidants and antioxidant enzymes. The purpose of this study was to evaluate the role of Spirulina, blue–green alga with antioxidant properties, in the protection of cisplatin-induced nephrotoxicity in rat. Methods: Rats were treated with CP (6 mg/kg bw, single dose, intraperitoneally). Spirulina (1,000 mg/kg) was administered orally for 8 days and CP treatment was given on day 4. Nephrotoxicity was assessed, 6 days after the CP treatment, by measuring plasma urea, creatinine, urinary N-acetyl-(d-glucose-aminidase) (β-NAG) and histopathology of kidney. Results: Rats treated with CP showed marked nephrotoxicity as evidenced from the significant elevation in plasma urea, creatinine and urinary β-NAG. Histological assessment revealed marked proximal tubular necrosis and extensive epithelial vacuolization in the kidney of CP-treated rats. Superoxide dismutase, catalase and glutathione peroxidase were decreased and lipid peroxidation was increased in kidney tissue. Pretreatment with Spirulina protected the rats from CP-induced nephrotoxicity. The rise in plasma urea, creatinine, urinary β-NAG, plasma and kidney tissue MDA and histomorphological changes were significantly attenuated by Spirulina. In vitro studies using human ovarian cancer cells revealed that Spirulina did not interfere with the cytotoxic effects of CP on tumor cells. Conclusions: In summary, Spirulina significantly protected the CP-induced nephrotoxicity through its antioxidant properties.





Similar content being viewed by others
References
Loehrer PJ, Einhorn LH (1984) Drugs five years later Cisplatin. Ann Intern Med 100:704–713
Heidemann HT, Muller S, Mertins L, Stepan G, Hoffmann K, Ohnhaus EE (1989) Effect of aminophylline on cisplatin nephrotoxicity in the rat. Br J Pharmacol 97:313–318
Safirstein R, Winston J, Moel D, Dikman S, Guttenplan J (1987) Cisplatin nephrotoxicity: insights into mechanism. Int J Androl 10:325–346
Schaaf GJ, Maas RF, de Groene EM, Fink-Gremmels J (2002) Management of oxidative stress by heme oxygenase-1 in cisplatin-induced toxicity in renal tubular cells. Free Radic Res 36:835–843
Xiao T, Choudhary S, Zhang W, Ansari NH, Salahudeen A (2003) Possible involvement of oxidative stress in cisplatin-induced apoptosis in LLC-PK1 cells. J Toxicol Environ Health A 66:469–479
Cummings BS, Schnellmann RG (2002) Cisplatin-induced renal cell apoptosis: caspase 3-dependent and -independent pathways. J Pharmacol Exp Ther 302:8–17
Baliga R, Zhang Z, Baliga M, Ueda N, Shah SV (1998) In vitro and in vivo evidence suggesting a role for iron in cisplatin-induced nephrotoxicity. Kidney Int 53:394–401
Rybak LP, Ravi R, Somani SM (1995) Mechanism of protection by diethyldithiocarbamate against cisplatin ototoxicity: antioxidant system. Fundam Appl Toxicol 26:293–300
Antunes LM, Araujo MC, Darin JD, Bianchi ML (2000) Effects of the antioxidants curcumin and vitamin C on cisplatin-induced clastogenesis in Wistar rat bone marrow cells. Mutat Res 465:131–137
Davis CA, Nick HS, Agarwal A (2001) Manganese superoxide dismutase attenuates Cisplatin-induced renal injury: importance of superoxide. J Am Soc Nephrol 12:2683–2690
Appenroth D, Frob S, Kersten L, Splinter FK, Winnefeld K (1997) Protective effects of vitamin E and C on cisplatin nephrotoxicity in developing rats. Arch Toxicol 71:677–683
Antunes LM, Francescato HD, Darin JD, de Lourdes PBM (2000) Effects of selenium pretreatment on cisplatin-induced chromosome aberrations in wistar rats. Teratog Carcinog Mutagen 20:341–348
Sueishi K, Mishima K, Makino K, Itoh Y, Tsuruya K, Hirakata H, Oishi R (2002) Protection by a radical scavenger edaravone against cisplatin-induced nephrotoxicity in rats. Eur J Pharmacol 451:203–208
Orditura M, De Vita F, Roscigno A, Infusino S, Auriemma A, Iodice P, Ciaramella F, Abbate G, Catalano G (1999) Amifostine: a selective cytoprotective agent of normal tissues from chemo-radiotherapy induced toxicity (Review). Oncol Rep 6:1357–1362
Ozen S, Akyol O, Iraz M, Sogut S, Ozugurlu F, Ozyurt H, Odaci E, Yildirim Z (2004) Role of caffeic acid phenethyl ester, an active component of propolis, against cisplatin-induced nephrotoxicity in rats. J Appl Toxicol 24:27–35
Khan Z, Bhadouria P, Bisen PS (2005) Nutritional and therapeutic potential of Spirulina. Curr Pharm Biotechnol 6:373–379
Ciferri O (1983) Spirulina, the edible microorganism. Microbiol Rev 47:551–578
Miranda MS, Cintra RG, Barros SB, Mancini Filho J (1998) Antioxidant activity of the microalga Spirulina maxima. Braz J Med Biol Res 31:1075–1079
Remirez D, Gonzalez R, Merino N, Rodriguez S, Ancheta O (2002) Inhibitory effects of Spirulina in zymosan-induced arthritis in mice. Mediators Inflamm 11:75–79
Chamorro G, Salazar M, Favila L, Bourges H (1996) Pharmacology and toxicology of Spirulina alga. Rev Invest Clin 48:389–399
Ayehunie S, Belay A, Baba TW, Ruprecht RM (1998) Inhibition of HIV-1 replication by an aqueous extract of Spirulina platensis (Arthrospira platensis). J Acquir Immune Defic Syndr Hum Retrovirol 18:7–12
Khan M, Shobha JC, Mohan IK, Naidu MU, Sundaram C, Singh S, Kuppusamy P, Kutala VK (2005) Protective effect of Spirulina against doxorubicin-induced cardiotoxicity. Phytother Res 19:1030–1037
Schwartz J, Shklar G, Reid S, Trickler D (1988) Prevention of experimental oral cancer by extracts of Spirulina–Dunaliella algae. Nutr Cancer 11:127–134
Weijl NI, Hopman GD, Wipkink-Bakker A, Lentjes EG, Berger HM, Cleton FJ, Osanto S (1998) Cisplatin combination chemotherapy induces a fall in plasma antioxidants of cancer patients. Ann Oncol 9:1331–1337
Horak E, Hopfer SM, Sunderman FW Jr (1981) Spectrophotometric assay for urinary N-acetyl-beta-d-glucosaminidase activity. Clin Chem 27:1180–1185
Bernheim FM, Bernheim BL, Wilbur KM (1948) The reaction between TBA and the oxidation products of certain lipids. J Biol Chem 174:257–264
McCord JM, Fridovich I (1969) Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244:6049–6055
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Baliga R, Ueda N, Walker PD, Shah SV (1999) Oxidant mechanisms in toxic acute renal failure. Drug Metab Rev 31:971–997
Somani SM, Ravi R, Rybak LP (1995) Diethyldithiocarbamate protection against cisplatin nephrotoxicity: antioxidant system. Drug Chem Toxicol 18:151–170
Yilmaz HR, Uz E, Yucel N, Altuntas I, Ozcelik N (2004) Protective effect of caffeic acid phenethyl ester (CAPE) on lipid peroxidation and antioxidant enzymes in diabetic rat liver. J Biochem Mol Toxicol 18:234–238
Nakano S, Gemba M (1989) Potentiation of cisplatin-induced lipid peroxidation in kidney cortical slices by glutathione depletion. Jpn J Pharmacol 50:87–92
Kruidering M, Van de Water B, de Heer E, Mulder GJ, Nagelkerke JF (1997) Cisplatin-induced nephrotoxicity in porcine proximal tubular cells: mitochondrial dysfunction by inhibition of complexes I to IV of the respiratory chain. J Pharmacol Exp Ther 280:638–649
Richter C, Gogvadze V, Laffranchi R, Schlapbach R, Schweizer M, Suter M, Walter P, Yaffee M (1995) Oxidants in mitochondria: from physiology to Diseases. Biochim Biophys Acta 1271:67–74
Sener G, Satiroglu H, Kabasakal L, Arbak S, Oner S, Ercan F, Keyer-Uysa M (2000) The protective effect of melatonin on cisplatin nephrotoxicity. Fundam Clin Pharmacol 14:553–560
Shiraishi F, Curtis LM, Truong L, Poss K, Visner GA, Madsen K, Nick HS, Agarwal A (2000) Heme oxygenase-1 gene ablation or expression modulates cisplatin-induced renal tubular apoptosis. Am J Physiol Renal Physiol 278:F726–F736
Agarwal A, Nick HS (2000) Renal response to tissue injury: lessons from heme oxygenase-1 GeneAblation and expression. J Am Soc Nephrol 11:965–973
Bhat VB, Madyastha KM (2000) C-phycocyanin: a potent peroxyl radical scavenger in vivo and in vitro. Biochem Biophys Res Commun 275:20–25
Romay C, Armesto J, Remirez D, Gonzalez R, Ledon N, Garcia I (1998) Antioxidant and anti-inflammatory properties of C-phycocyanin from blue–green Algae. Inflamm Res 47:36–41
Khan M, Varadharaj S, Ganesan LP, Shobha JC, Naidu MU, Parinandi NL, Tridandapani S, Kutala VK, Kuppusamy P (2006) C-phycocyanin protects against ischemia–reperfusion injury of heart through involvement of p38 MAPK and ERK signaling. Am J Physiol Heart Circ Physiol (in press)
Mathew B, Sankaranarayanan R, Nair PP, Varghese C, Somanathan T, Amma BP, Amma NS, Nair MK (1995) Evaluation of chemoprevention of oral cancer with Spirulina fusiformis. Nutr Cancer 24:197–202
Premkumar K, Pachiappan A, Abraham SK, Santhiya ST, Gopinath PM, Ramesh A (2001) Effect of Spirulina fusiformis on cyclophosphamide and mitomycin-C induced genotoxicity and oxidative stress in mice. Fitoterapia 72:906–911
Upasani CD, Khera A, Balaraman R (2001) Effect of lead with vitamin E, C, or Spirulina on malondialdehyde, conjugated dienes and hydroperoxides in rats. Indian J Exp Biol 39:70–74
Premkumar K, Abraham SK, Santhiya ST, Ramesh A (2004) Protective effect of Spirulina fusiformis on chemical-induced genotoxicity in mice. Fitoterapia 75:24–31
Wang Y, Chang CF, Chou J, Chen HL, Deng X, Harvey BK, Cadet JL, Bickford PC (2005) Dietary supplementation of with blueberries, spinach, or spirulina reduces ischemic brain damage. Exp Neurol 193:75–84
Farooq SM, Asokan D, Sakthivel R, Kalaiselvi P, Varalakshmi P (2004) Salubrious effect of C-phycocyanin against oxalate-mediated renal cell injury. Clin Chim Acta 348:199–205
Lau AH (1999) Apoptosis induced by cisplatin nephrotoxic injury. Kidney Int 56:1295–1298
Wu YJ, Muldoon LL, Neuwelt EA (2005) The chemoprotective agent N-acetylcysteine blocks cisplatin-induced apoptosis through caspase signaling pathway. J Pharmacol Exp Ther 312:424–431
Acknowledgment
We thank M/s Parry Neutraceuticals, Chennai, India for providing the pure powder of Spirulina for our study and Jing Fang for her help in the study with ovarian cancer cells. This work was partially supported by the National Institutes of Health Grant CA-102264.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohan, I.K., Khan, M., Shobha, J.C. et al. Protection against cisplatin-induced nephrotoxicity by Spirulina in rats. Cancer Chemother Pharmacol 58, 802–808 (2006). https://doi.org/10.1007/s00280-006-0231-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-006-0231-8