Abstract
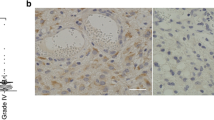
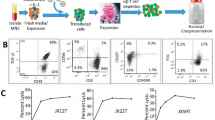
The standard treatment of high-grade glioma presents a combination of radiotherapy, chemotherapy and surgery. Immunotherapy is proposed as a potential adjunct to standard cytotoxic regimens to target remaining microscopic disease following resection. We have shown ex vivo expanded/activated γδ T cells to be a promising innate lymphocyte therapy based on their recognition of stress antigens expressed on gliomas. However, successful integration of γδ T cell therapy protocols requires understanding the efficacy and safety of adoptively transferred immune cells in the post-treatment environment. The unique features of γδ T cell product and the environment (hypoxia, inflammation) can affect levels of expression of key cell receptors and secreted factors and either promote or hinder the feasibility of γδ T cell therapy. We investigated the potential for the γδ T cells to injure normal brain tissue that may have been stressed by treatment. We evaluated γδ T cell toxicity by assessing actual and correlative toxicity indicators in several available models including: (1) expression of stress markers on normal primary human astrocytes (as surrogate for brain parenchyma) after irradiation and temozolomide treatment, (2) cytotoxicity of γδ T cells on normal and irradiated primary astrocytes, (3) microglial activation and expression of stress-induced ligands in mouse brain after whole-brain irradiation and (4) expression of stress-induced markers on human brain tumors and on normal brain tissue. The lack of expression of stress-induced ligands in all tested models suggests that γδ T cell therapy is safe for brain tumor patients who undergo standard cytotoxic therapies.






Similar content being viewed by others
Abbreviations
- BrdU:
-
Bromodeoxyuridine
- CAR:
-
Chimeric antigen receptor
- CTL:
-
Cytotoxic T lymphocytes
- DAPI:
-
4′,6-diamidino-2-phenylindole, nuclear counterstaining dye
- DDR:
-
DNA damage response
- E:T:
-
Effector-to-target ratio
- FFPE:
-
Formalin-fixed paraffin-embedded specimens
- GBM:
-
Glioblastoma multiforme
- γδ T:
-
Gamma delta T cells
- IHC:
-
Immunohistochemistry
- IL:
-
Interleukin
- LAK:
-
Lymphokine-activated killer cells
- NK:
-
Natural killer cells
- TCR:
-
T cell receptor
- TMZ:
-
Temozolomide
- TNFα:
-
Tumor necrosis factor α
- UAB:
-
University of Alabama at Birmingham
- ZOL:
-
Zoledronic acid
References
Ishikawa E, Takano S, Ohno T, Tsuboi K (2012) Adoptive cell transfer therapy for malignant gliomas. Adv Exp Med Biol 746:109–120
Bielamowicz K, Khawja S, Ahmed N (2013) Adoptive cell therapies for glioblastoma. Front Oncol 3:275
Lamb LS (2009) Gammadelta T cells as immune effectors against high-grade gliomas. Immunol Res 45:85–95
Vantourout P, Hayday A (2013) Six-of-the-best: unique contributions of γδ T cells to immunology. Nat Rev Immunol 13:88–100
Beetz S, Marischen L, Kabelitz D, Wesch D (2007) Human gamma delta T cells: candidates for the development of immunotherapeutic strategies. Immunol Res 37:97–111
Kabelitz D, Wesch D, He W (2007) Perspectives of gammadelta T cells in tumor immunology. Cancer Res 67:5–8
Preusser M, de Ribaupierre S, Wöhrer A, Erridge SC, Hegi M et al (2011) Current concepts and management of glioblastoma. Ann Neurol 70:9–21
Mukherjee D, Coates PJ, Lorimore SA, Wright EG (2014) Responses to ionizing radiation mediated by inflammatory mechanisms. J Pathol 232:289–299
Zhao W, Robbins ME (2009) Inflammation and chronic oxidative stress in radiation-induced late normal tissue injury: therapeutic implications. Curr Med Chem 16:130–143
Friedman EJ (2002) Immune modulation by ionizing radiation and its implications for cancer immunotherapy. Curr Pharm Des 8:1765–1780
Rödel F, Frey B, Gaipl U, Keilholz L, Fournier C et al (2012) Modulation of inflammatory immune reactions by low-dose ionizing radiation: molecular mechanisms and clinical application. Curr Med Chem 19:1741–1750
Rödel F, Frey B, Multhoff G, Gaipl U (2015) Contribution of the immune system to bystander and non-targeted effects of ionizing radiation. Cancer Lett 356:105–113
Bryant NL, Gillespie GY, Lopez RD, Markert JM, Cloud GA et al (2011) Preclinical evaluation of ex vivo expanded/activated γδ T cells for immunotherapy of glioblastoma multiforme. J Neurooncol 101:179–188
Wu KL, Tu B, Li YQ, Wong CS (2010) Role of intercellular adhesion molecule-1 in radiation-induced brain injury. Int J Radiat Oncol Biol Phys 76:220–228
Spear P, Wu MR, Sentman ML, Sentman CL (2013) NKG2D ligands as therapeutic targets. Cancer Immun 13:8
Raulet DH, Gasser S, Gowen BG, Deng W, Jung H (2013) Regulation of ligands for the NKG2D activating receptor. Annu Rev Immunol 31:413–441
Kim SJ, Kim JS, Park ES, Lee JS, Lin Q et al (2011) Astrocytes upregulate survival genes in tumor cells and induce protection from chemotherapy. Neoplasia 13:286–298
Wang L, Cossette SM, Rarick KR, Gershan J, Dwinell MB et al (2013) Astrocytes directly influence tumor cell invasion and metastasis in vivo. PLoS One 8:e80933
Moore ED, Kooshki M, Metheny-Barlow LJ, Gallagher PE, Robbins ME (2013) Angiotensin-(1–7) prevents radiation-induced inflammation in rat primary astrocytes through regulation of MAP kinase signaling. Free Radic Biol Med 65:1060–1068
Lamb LS, Bowersock J, Dasgupta A, Gillespie GY, Su Y et al (2013) Engineered drug resistant γδ T cells kill glioblastoma cell lines during a chemotherapy challenge: a strategy for combining chemo- and immunotherapy. PLoS One 8:e51805
Couzin-Frankel J (2013) Breakthrough of the year 2013. Cancer immunotherapy. Science 342:1432–1433
Morgan RA, Yang JC, Kitano M, Dudley ME, Laurencot CM et al (2010) Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther 18:843–851
Nicol AJ, Tokuyama H, Mattarollo SR, Hagi T, Suzuki K et al (2011) Clinical evaluation of autologous gamma delta T cell-based immunotherapy for metastatic solid tumours. Br J Cancer 105:778–786
Gomes AQ, Martins DS, Silva-Santos B (2010) Targeting γδ T lymphocytes for cancer immunotherapy: from novel mechanistic insight to clinical application. Cancer Res 70:10024–10027
Bryant NL, Suarez-Cuervo C, Gillespie GY, Markert JM, Nabors LB et al (2009) Characterization and immunotherapeutic potential of gammadelta T-cells in patients with glioblastoma. Neuro Oncol 11:357–367
Coudert JD, Held W (2006) The role of the NKG2D receptor for tumor immunity. Semin Cancer Biol 16:333–343
Schmudde M, Braun A, Pende D, Sonnemann J, Klier U et al (2008) Histone deacetylase inhibitors sensitize tumour cells for cytotoxic effects of natural killer cells. Cancer Lett 272:110–121
Rohner A, Langenkamp U, Siegler U, Kalberer CP, Wodnar-Filipowicz A (2007) Differentiation-promoting drugs up-regulate NKG2D ligand expression and enhance the susceptibility of acute myeloid leukemia cells to natural killer cell-mediated lysis. Leuk Res 31:1393–1402
Poggi A, Catellani S, Garuti A, Pierri I, Gobbi M et al (2009) Effective in vivo induction of NKG2D ligands in acute myeloid leukaemias by all-trans-retinoic acid or sodium valproate. Leukemia 23:641–648
Rosental B, Appel MY, Yossef R, Hadad U, Brusilovsky M et al (2012) The effect of chemotherapy/radiotherapy on cancerous pattern recognition by NK cells. Curr Med Chem 19:1780–1791
Kim JY, Son YO, Park SW, Bae JH, Chung JS et al (2006) Increase of NKG2D ligands and sensitivity to NK cell-mediated cytotoxicity of tumor cells by heat shock and ionizing radiation. Exp Mol Med 38:474–484
Bedel R, Thiery-Vuillemin A, Grandclement C, Balland J, Remy-Martin JP et al (2011) Novel role for STAT3 in transcriptional regulation of NK immune cell targeting receptor MICA on cancer cells. Cancer Res 71:1615–1626
Xu X, Rao GS, Groh V, Spies T, Gattuso P et al (2011) Major histocompatibility complex class I-related chain A/B (MICA/B) expression in tumor tissue and serum of pancreatic cancer: role of uric acid accumulation in gemcitabine-induced MICA/B expression. BMC Cancer 11:194
Butler JE, Moore MB, Presnell SR, Chan HW, Chalupny NJ et al (2009) Proteasome regulation of ULBP1 transcription. J Immunol 182:6600–6609
Riederer I, Sievert W, Eissner G, Molls M, Multhoff G (2010) Irradiation-induced up-regulation of HLA-E on macrovascular endothelial cells confers protection against killing by activated natural killer cells. PLoS One 5:e15339
Gasser S, Orsulic S, Brown EJ, Raulet DH (2005) The DNA damage pathway regulates innate immune system ligands of the NKG2D receptor. Nature 436:1186–1190
Hayday AC (2009) Gammadelta T cells and the lymphoid stress-surveillance response. Immunity 31:184–196
Schwacha MG (2009) Gammadelta T-cells: potential regulators of the post-burn inflammatory response. Burns 35:318–326
Matsushima A, Ogura H, Fujita K, Koh T, Tanaka H et al (2004) Early activation of gammadelta T lymphocytes in patients with severe systemic inflammatory response syndrome. Shock 22:11–15
Williams J, Chen Y, Rubin P, Finkelstein J, Okunieff P (2003) The biological basis of a comprehensive grading system for the adverse effects of cancer treatment. Semin Radiat Oncol 13:182–188
Lee WH, Sonntag WE, Mitschelen M, Yan H, Lee YW (2010) Irradiation induces regionally specific alterations in pro-inflammatory environments in rat brain. Int J Radiat Biol 86:132–144
Rola R, Sarkissian V, Obenaus A, Nelson GA, Otsuka S et al (2005) High-LET radiation induces inflammation and persistent changes in markers of hippocampal neurogenesis. Radiat Res 164:556–560
Schindler MK, Forbes ME, Robbins ME, Riddle DR (2008) Aging-dependent changes in the radiation response of the adult rat brain. Int J Radiat Oncol Biol Phys 70:826–834
Greene-Schloesser D, Moore E, Robbins ME (2013) Molecular pathways: radiation-induced cognitive impairment. Clin Cancer Res 19:2294–2300
Moravan MJ, Olschowka JA, Williams JP, O’Banion MK (2011) Cranial irradiation leads to acute and persistent neuroinflammation with delayed increases in T-cell infiltration and CD11c expression in C57BL/6 mouse brain. Radiat Res 176:459–473
Acknowledgments
We thank Dr. Mitchell S. Berger, Department of Neurological Surgery, University of California, San Francisco for making available FFPE samples of human brain tumors. Support from Elsa U Pardee Foundation (Lawrence S. Lamb) is acknowledged and appreciated.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pereboeva, L., Harkins, L., Wong, S. et al. The safety of allogeneic innate lymphocyte therapy for glioma patients with prior cranial irradiation. Cancer Immunol Immunother 64, 551–562 (2015). https://doi.org/10.1007/s00262-015-1662-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-015-1662-z