Abstract
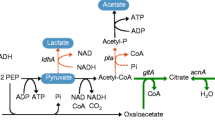
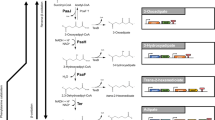
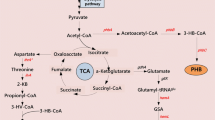
The linear C6 dicarboxylic acid adipic acid is an important bulk chemical in the petrochemical industry as precursor of the polymer nylon-6,6-polyamide. In recent years, efforts were made towards the biotechnological production of adipate from renewable carbon sources using microbial cells. One strategy is to produce adipate via a reversed β-oxidation pathway. Hitherto, the adipate titers were very low due to limiting enzyme activities for this pathway. In most cases, the CoA intermediates are non-natural substrates for the tested enzymes and were therefore barely converted. We here tested heterologous enzymes in Escherichia coli to overcome these limitations and to improve the production of adipate via a reverse β-oxidation pathway. We tested in vitro selected enzymes for the efficient reduction of the enoyl-CoA and in the final reaction for the thioester cleavage. The genes encoding the enzymes which showed in vitro the highest activity were then used to construct an expression plasmid for a synthetic adipate pathway. Expression of paaJ, paaH, paaF, dcaA, and tesB in E. coli BL21(DE3) resulted in the production of up to 36 mg/L of adipate after 30 h of cultivation. Beside the activities of the pathway enzymes, the availability of metabolic precursors may limit the synthesis of adipate, providing another key target for further strain engineering towards high-yield production of adipate with E. coli.





Similar content being viewed by others
References
Babu T, Yun EJ, Kim S, Kim DH, Liu KH, Kim SR, Kim KH (2015) Engineering Escherichia coli for the production of adipic acid through the reversed β-oxidation pathway. Process Biochem 50(12):2066–2071
Bart JCJ, Cavallaro S (2015a) Transiting from adipic acid to bioadipic acid. 1, petroleum-based processes. Ind Eng Chem Res 54(1):1–46. doi:10.1021/ie5020734
Bart JCJ, Cavallaro S (2015b) Transiting from adipic acid to bioadipic acid. Part II Biosynthetic pathways Ind Eng Chem Res 54(2):567–576. doi:10.1021/ie502074d
Becker DF, Fuchs JA, Stankovich MT (1994) Product binding modulates the thermodynamic properties of a Megasphaera elsdenii short-chain acyl-CoA dehydrogenase active-site mutant. Biochemistry 33(23):7082–7087. doi:10.1021/bi00189a010
Binieda A, Fuhrmann M, Lehner B, Rey-Berthod C, Frutiger-Hughes S, Hughes G (1999) Purification, characterization, DNA sequence and cloning of a pimeloyl-CoA synthetase from Pseudomonas mendocina 35. Biochem J 340(3):793–801
Blattner FR, Plunkett G 3rd, Bloch CA, Perna NT, Burland V, Riley M, Collado-Vides J, Glasner JD, Rode CK, Mayhew GF, Gregor J, Davis NW, Kirkpatrick HA, Goeden MA, Rose DJ, Mau B, Shao Y (1997) The complete genome sequence of Escherichia coli K-12. Science (New York, NY) 277(5331):1453–1462
Bugg TD, Rahmanpour R (2015) Enzymatic conversion of lignin into renewable chemicals. Curr Opin Chem Biol 29:10–17. doi:10.1016/j.cbpa.2015.06.009
Chen H, Bjerknes M, Kumar R, Jay E (1994) Determination of the optimal aligned spacing between the Shine–Dalgarno sequence and the translation initiation codon of Escherichia coli mRNAs. Nucleic Acids Res 22(23):4953–4957. doi:10.1093/nar/22.23.4953
Cheong S, Clomburg JM, Gonzalez R (2016) Energy- and carbon-efficient synthesis of functionalized small molecules in bacteria using non-decarboxylative Claisen condensation reactions. Nature Biotechnol 34(5):556–561. doi:10.1038/nbt.3505
Clomburg JM, Vick JE, Blankschien MD, Rodriguez-Moya M, Gonzalez R (2012) A synthetic biology approach to engineer a functional reversal of the beta-oxidation cycle. ACS Synth Biol 1(11):541–554. doi:10.1021/sb3000782
Clomburg JM, Blankschien MD, Vick JE, Chou A, Kim S, Gonzalez R (2015) Integrated engineering of β-oxidation reversal and ω-oxidation pathways for the synthesis of medium chain ω-functionalized carboxylic acids. Metab Eng 28:202–212
Cummings JG, Lau SM, Powell PJ, Thorpe C (1992) Reductive half-reaction in medium-chain acyl-CoA dehydrogenase: modulation of internal equilibrium by carboxymethylation of a specific methionine residue. Biochemistry 31(36):8523–8529
Dekishima Y, Lan EI, Shen CR, Cho KM, Liao JC (2011) Extending carbon chain length of 1-butanol pathway for 1-hexanol synthesis from glucose by engineered Escherichia coli. J Am Chem Soc 133(30):11399–11401
Dellomonaco C, Clomburg JM, Miller EN, Gonzalez R (2011) Engineered reversal of the β-oxidation cycle for the synthesis of fuels and chemicals. Nature 476(7360):355–359
Deng Y, Mao Y (2015) Production of adipic acid by the native-occurring pathway in Thermobifida fusca B6. J Appl Microbiol 119(4):1057–1063
Engler C, Gruetzner R, Kandzia R, Marillonnet S (2009) Golden gate shuffling: a one-pot DNA shuffling method based on type IIs restriction enzymes. PLoS One 4(5):e5553
Giacomini A, Ollero FJ, Squartini A, Nuti MP (1994) Construction of multipurpose gene cartridges based on a novel synthetic promoter for high-level gene expression in Gram-negative bacteria. Gene 144(1):17–24
Haapalainen AM, Merilainen G, Wierenga RK (2006) The thiolase superfamily: condensing enzymes with diverse reaction specificities. Trends Biochem Sci 31(1):64–71. doi:10.1016/j.tibs.2005.11.011
Hoffmeister M, Piotrowski M, Nowitzki U, Martin W (2005) Mitochondrial trans-2-enoyl-CoA reductase of wax ester fermentation from Euglena gracilis defines a new family of enzymes involved in lipid synthesis. J Biol Chem 280(6):4329–4338
Karl RM, Hupperich M, Thomer A, Eggerer H (1995) Inhibition of 3-hydroxy-3-methylglutaryl-CoA reductase and citrate synthase by sulfoxides and sulfones of substrate-analogue CoA-thioether derivatives. Eur J Biochem 227(1–2):292–295
Kaschabek SR, Kuhn B, Müller D, Schmidt E, Reineke W (2002) Degradation of aromatics and chloroaromatics by Pseudomonas sp. strain B13: purification and characterization of 3-oxoadipate: succinyl-coenzyme A (CoA) transferase and 3-oxoadipyl-CoA thiolase. J Bacteriol 184(1):207–215
Küberl A, Fränzel B, Eggeling L, Polen T, Wolters DA, Bott M (2014) Pupylated proteins in Corynebacterium glutamicum revealed by MudPIT analysis. Proteomics 14(12):1531–1542. doi:10.1002/pmic.201300531
Lehman TC, Hale DE, Bhala A, Thorpe C (1990) An acyl-CoA dehydrogenase assay utilizing the ferrocenium ion. Anal Biochem 186:280–284
Lin H, Castro NM, Bennett GN, San K-Y (2006) Acetyl-CoA synthetase overexpression in Escherichia coli demonstrates more efficient acetate assimilation and lower acetate accumulation: a potential tool in metabolic engineering. Appl Microbiol Biotechnol 71(6):870–874
Modis Y, Wierenga RK (1999) A biosynthetic thiolase in complex with a reaction intermediate: the crystal structure provides new insights into the catalytic mechanism. Structure 7(10):1279–1290
Musser MT (2005) Adipic acid. ULLMANN’S Encyclopedia of Industrial Chemistry (Volume 1). Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, pp 537–48
Nie L, Ren Y, Janakiraman A, Smith S, Schulz H (2008) A novel paradigm of fatty acid β-oxidation exemplified by the thioesterase-dependent partial degradation of conjugated linoleic acid that fully supports growth of Escherichia coli. Biochemistry 47(36):9618–9626
Niu W, Draths KM, Frost JW (2002) Benzene-free synthesis of adipic acid. Biotechnol Prog 18(2):201–211
Nolte JC, Schürmann M, Schepers C-L, Vogel E, Wübbeler JH, Steinbüchel A (2014) Novel characteristics of succinate coenzyme A (succinate-CoA) ligases: conversion of malate to malyl-CoA and CoA-thioester formation of succinate analogues in vitro. Appl Environ Microb 80(1):166–176
Paczia N, Nilgen A, Lehmann T, Gatgens J, Wiechert W, Noack S (2012) Extensive exometabolome analysis reveals extended overflow metabolism in various microorganisms. Microb Cell Factories 11:122. doi:10.1186/1475-2859-11-122
Parke D, Garcia MA, Ornston LN (2001) Cloning and genetic characterization of dca genes required for beta-oxidation of straight-chain dicarboxylic acids in Acinetobacter sp. strain ADP1. Appl Environ Microb 67(10):4817–4827
Parthasarathy A, Pierik AJ, Kahnt J, Zelder O, Buckel W (2011) Substrate specificity of 2-hydroxyglutaryl-CoA dehydratase from Clostridium symbiosum: toward a bio-based production of adipic acid. Biochemistry 50(17):3540–3550
Polen T, Spelberg M, Bott M (2013) Toward biotechnological production of adipic acid and precursors from biorenewables. J Biotechnol 167(2):75–84
Grand View Research (2014) Adipic acid market analysis and segment forecasts to 2020.
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd ed Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Shikata K, Fukui T, Atomi H, Imanaka T (2007) A novel ADP-forming succinyl-CoA synthetase in Thermococcus kodakaraensis structurally related to the archaeal nucleoside diphosphate-forming acetyl-CoA synthetases. J Biol Chem 282(37):26963–26970. doi:10.1074/jbc.M702694200
Suastegui M, Matthiesen JE, Carraher JM, Hernandez N, Rodriguez Quiroz N, Okerlund A, Cochran EW, Shao Z, Tessonnier JP (2016) Combining metabolic engineering and electrocatalysis: application to the production of polyamides from sugar. Angew Chem 55(7):2368–2373. doi:10.1002/anie.201509653
Teufel R, Mascaraque V, Ismail W, Voss M, Perera J, Eisenreich W, Haehnel W, Fuchs G (2010) Bacterial phenylalanine and phenylacetate catabolic pathway revealed. Proc Natl Acad Sci 107(32):14390–14395. doi:10.1073/pnas.1005399107
Thakker C, Martínez I, San KY, Bennett GN (2012) Succinate production in Escherichia coli. Biotechnol J 7(2):213–224
Thomas JM, Raja R, Johnson BF, O’Connell TJ, Sankar G, Khimyak T (2003) Bimetallic nanocatalysts for the conversion of muconic acid to adipic acid. Chem Commun (Camb) 21(10):1126–1127
Thorpe C, Kim J (1995) Structure and mechanism of action of the acyl-CoA dehydrogenases. FASEB J 9(9):718–725
van Duuren JB, Wijte D, Leprince A, Karge B, Puchalka J, Wery J, Dos Santos VA, Eggink G, Mars AE (2011) Generation of a catR deficient mutant of P. putida KT2440 that produces cis, cis-muconate from benzoate at high rate and yield. J Biotechnol 156(3):163–172
Villemin D, Labiad B, Loupy A (1993) Clay catalysis: a convenient and rapid formation of anhydride from carboxylic acid and isopropenyl acetate under microwave irradiation. Synth Commun 23(4):419–424. doi:10.1080/00397919308009796
Wells T, Ragauskas AJ (2012) Biotechnological opportunities with the β-ketoadipate pathway. Trends Biotechnol 30(12):627–637
Yu JL, Xia XX, Zhong JJ, Qian ZG (2014) Direct biosynthesis of adipic acid from a synthetic pathway in recombinant Escherichia coli. Biotechnol Bioeng 111(12):2580–2586
Zhang H, Li Z, Pereira B, Stephanopoulos G (2015) Engineering E. coli–E. coli cocultures for production of muconic acid from glycerol. Microb Cell Factories 14(1):1
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
.
ESM 1
(PDF 327 kb)
Rights and permissions
About this article
Cite this article
Kallscheuer, N., Gätgens, J., Lübcke, M. et al. Improved production of adipate with Escherichia coli by reversal of β-oxidation. Appl Microbiol Biotechnol 101, 2371–2382 (2017). https://doi.org/10.1007/s00253-016-8033-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-8033-3