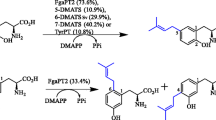
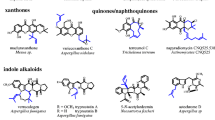
Abstract
A series of putative indole prenyltransferase genes could be identified in the genome sequences of different fungal strains including Aspergillus fumigatus and Neosartorya fischeri. The gene products show significant sequence similarities to dimethylallyltryptophan synthases from various fungi. These genes belong to different gene clusters and are involved in the biosynthesis of secondary metabolites. Ten of them were cloned and overexpressed in Escherichia coli and Saccharomyces cerevisiae and proven to be soluble proteins. They catalyse different prenyl transfer reactions onto indole moieties of various substrates and do not require divalent metal ions for their prenyl transfer reactions. These enzymes showed broad substrate specificities towards their aromatic substrates. Diverse simple tryptophan derivatives and tryptophan-containing cyclic dipeptides were accepted by several prenyltransferases as substrates and converted to prenylated derivatives. This feature of substrate flexibility was successfully used for regiospecific and stereospecific synthesis of different indole derivatives.







Similar content being viewed by others
References
Abe K, Gomi K, Hasegawa F, Machida M (2006) Impact of Aspergillus oryza genomics on industrial production of metabolites. Mycopathologia 162:143–153
Bok JW, Hoffmeister D, Maggio-Hall LA, Murillo R, Glasner JD, Keller NP (2006) Genomic mining for Aspergillus natural products. Chem Biol 13:31–37
Botta B, Monache GD, Menendez P, Boffi A (2005) Novel prenyltransferase enzymes as a tool for flavonoid prenylation. Trends Pharmacol Sci 26:606–608
Chong YS, Su LL, Arulkumaran S (2004) Current strategies for the prevention of postpartum haemorrhage in the third stage of labour. Curr Opin Obstet Gynecol 16:143–150
Cole RJ, Kirksey JW, Dorner JW, Wilson DM, Johnson JC Jr, Johnson AN, Bedell DM, Springer JP, Chexal KK, Clardy JC et al (1977) Mycotoxins produced by Aspergillus fumigatus species isolated from molded silage. J Agric Food Chem 25:826–830
Cui CB, Kakeya H, Okada G, Onose R, Ubukata M, Takahashi I, Isono K, Osada H (1995) Tryprostatins A and B, novel mammalian cell cycle inhibitors produced by Aspergillus fumigatus. J Antibiot 48:1382–1384
Cui CB, Kakeya H, Osada H (1996) Novel mammalian cell cycle inhibitors, tryprostatins A, B and other diketopiperazines produced by Aspergillus fumigatus. II. Physico-chemical properties and structures. J Antibiot 49:534–540
Ding Y, Williams RM, Sherman DH (2008) Molecular analysis of a 4-dimethylallyltryptophan synthase from Malbranchea aurantiaca. J Biol Chem 283:16068–16076
Gebler JC, Poulter CD (1992) Purification and characterization of dimethylallyl tryptophan synthase from Claviceps purpurea. Arch Biochem Biophys 296:308–313
Goadsby PJ (2003) Migraine: diagnosis and management. Intern Med J 33:436–442
Grundmann A, Li S-M (2005) Overproduction, purification and characterization of FtmPT1, a brevianamide F prenyltransferase from Aspergillus fumigatus. Microbiology 151:2199–2207
Grundmann A, Kuznetsova T, Afiyatullov SS, Li S-M (2008) FtmPT2, an N-prenyltransferase from Aspergillus fumigatus, catalyses the last step in the biosynthesis of fumitremorgin B. Chembiochem 9:2059–2063
Heide L (2009) Prenyl transfer to aromatic substrates: genetics and enzymology. Curr Opin Chem Biol 13:171–179
Heinstein PF, Lee SI, Floss HG (1971) Isolation of dimethylallylpyrophosphate: tryptophan dimethylallyl transferase from the ergot fungus (Claviceps spec.). Biochem Biophys Res Commun 44:1244–1251
Janardhanan KK, Sattar A, Husain A (1984) Production of fumigaclavine A by Aspergillus tamarii Kita. Can J Microbiol 30:247–250
Jones MG (2007) The first filamentous fungal genome sequences: Aspergillus leads the way for essential everyday resources or dusty museum specimens? Microbiology 153:1–6
Keller NP, Turner G, Bennett JW (2005) Fungal secondary metabolism—from biochemistry to genomics. Nat Rev Microbiol 3:937–947
Kharel Y, Takahashi S, Yamashita S, Koyama T (2006) Manipulation of prenyl chain length determination mechanism of cis-prenyltransferases. FEBS J 273:647–657
Kondoh M, Usui T, Mayumi T, Osada H (1998) Effects of tryprostatin derivatives on microtubule assembly in vitro and in situ. J Antibiot 51:801–804
Kremer A, Li S-M (2008) Potential of a 7-dimethylallyltryptophan synthase as a tool for production of prenylated indole derivatives. Appl Microbiol Biotechnol 79:951–961
Kremer A, Westrich L, Li S-M (2007) A 7-dimethylallyltryptophan synthase from Aspergillus fumigatus: overproduction, purification and biochemical characterization. Microbiology 153:3409–3416
Lee SL, Floss HG, Heinstein P (1976) Purification and properties of dimethylallylpyrophosphate: tryptophan dimethylallyl transferase, the first enzyme of ergot alkaloid biosynthesis in Claviceps sp. SD 58. Arch Biochem Biophys 177:84–94
Li S-M (2009) Evolution of aromatic prenyltransferases in the biosynthesis of indole derivatives. Phytochemistry (in press)
Liang PH, Ko TP, Wang AH (2002) Structure, mechanism and function of prenyltransferases. Eur J Biochem 269:3339–3354
Lu YP, Liu HG, Liang PH (2009) Different reaction mechanisms for cis- and trans-prenyltransferases. Biochem Biophys Res Commun 379:351–355
Ma HY, Song YC, Mao YY, Jiang JH, Tan RX, Luo L (2006) Endophytic fungal metabolite fumigaclavine C causes relaxation of isolated rat aortic rings. Planta Med 72:387–392
Perez-Sala D (2007) Protein isoprenylation in biology and disease: general overview and perspectives from studies with genetically engineered animals. Front Biosci 12:4456–4472
Rabindran SK, He H, Greenberger LM (2002) Reversal of multidrug resistance in human colon carcinoma cells using fumitremorgins and diketopiperazines. US patent no. 6372775
Raskin DM, Seshadri R, Pukatzki SU, Mekalanos JJ (2006) Bacterial genomics and pathogen evolution. Cell 124:703–714
Ruan H-L, Yin W-B, Wu J-Z, Li S-M (2008) Reinvestigation of a cyclic dipeptide N-prenyltransferase reveals rearrangement of N-1 prenylated indole derivatives. Chembiochem 9:1044–1047
Sasaki K, Mito K, Ohara K, Yamamoto H, Yazaki K (2008) Cloning and characterization of naringenin 8-prenyltransferase, a flavonoid-specific prenyltransferase of Sophora flavescens. Plant Physiol 146:1075–1084
Schardl CL, Panaccione DG, Tudzynski P (2006) Ergot alkaloids—biology and molecular biology. In: Cordell GA (ed) The alkaloids: chemistry and biology, vol 63. Academic, Amsterdam
Schneider P, Weber M, Hoffmeister D (2008) The Aspergillus nidulans enzyme TdiB catalyzes prenyltransfer to the precursor of bioactive asterriquinones. Fungal Genet Biol 45:302–309
Steffan N, Li S-M (2009) Increasing structure diversity of prenylated diketopiperazine derivatives by using a 4-dimethylallyltryptophan synthase. Arch Microbiol 191:461–466
Steffan N, Unsöld IA, Li S-M (2007) Chemoenzymatic synthesis of prenylated indole derivatives by using a 4-dimethylallyltryptophan synthase from Aspergillus fumigatus. Chembiochem 8:1298–1307
Steffan N, Grundmann A, Yin W-B, Kremer A, Li S-M (2009) Indole prenyltransferases from fungi: a new enzyme group with high potential for the production of prenylated indole derivatives. Curr Med Chem 16:218–231
Takahashi S, Koyama T (2006) Structure and function of cis-prenyl chain elongating enzymes. Chem Rec 6:194–205
Tsai HF, Wang H, Gebler JC, Poulter CD, Schardl CL (1995) The Claviceps purpurea gene encoding dimethylallyltryptophan synthase, the committed step for ergot alkaloid biosynthesis. Biochem Biophys Res Commun 216:119–125
Tudzynski P, Holter K, Correia T, Arntz C, Grammel N, Keller U (1999) Evidence for an ergot alkaloid gene cluster in Claviceps purpurea. Mol Gen Genet 261:133–141
Tudzynski P, Correia T, Keller U (2001) Biotechnology and genetics of ergot alkaloids. Appl Microbiol Biotechnol 57:593–605
Unsöld IA, Li S-M (2005) Overproduction, purification and characterization of FgaPT2, a dimethylallyltryptophan synthase from Aspergillus fumigatus. Microbiology 151:1499–1505
Unsöld IA, Li S-M (2006) Reverse prenyltransferase in the biosynthesis of fumigaclavine C in Aspergillus fumigatus: gene expression, purification and characterization of fumigaclavine C synthase FgaPT1. Chembiochem 7:158–164
Usui T, Kondoh M, Cui CB, Mayumi T, Osada H (1998) Tryprostatin A, a specific and novel inhibitor of microtubule assembly. Biochem J 333:543–548
Vinokurova NG, Ozerskaya SM, Baskunov BP, Arinbasarov MU (2003) The Penicillium commune Thom and Penicillium clavigerum demelius fungi producing fumigaclavines A and B. Microbiology (Moscow) 72:149–151
Wang J, Machado C, Panaccione DG, Tsai HF, Schardl CL (2004) The determinant step in ergot alkaloid biosynthesis by an endophyte of perennial ryegrass. Fungal Genet Biol 41:189–198
Williams RM, Stocking EM, Sanz-Cervera JF (2000) Biosynthesis of prenylated alkaloids derived from tryptophan. Topics Curr Chem 209:97–173
Wu X-F, Fei M-J, Shu R-G, Tan R-X, Xu Q (2005) Fumigaclavine C, an fungal metabolite, improves experimental colitis in mice via downregulating Th1 cytokine production and matrix metalloproteinase activity. Int Immunopharmacol 5:1543–1553
Yin W-B, Ruan H-L, Westrich L, Grundmann A, Li S-M (2007) CdpNPT, an N-prenyltransferase from Aspergillus fumigatus: overproduction, purification and biochemical characterisation. Chembiochem 8:1154–1161
Yin W-B, Cheng J, Li S-M (2009a) Stereospecific synthesis of aszonalenins by using two recombinant prenyltransferases. Org Biomol Chem 7:2202–2207
Yin W-B, Grundmann A, Cheng J, Li S-M (2009b) Acetylaszonalenin biosynthesis in Neosartorya fischeri: identification of the biosynthetic gene cluster by genomic mining and functional proof of the genes by biochemical investigation. J Biol Chem 284:100–109
Zhao S, Smith KS, Deveau AM, Dieckhaus CM, Johnson MA, Macdonald TL, Cook JM (2002) Biological activity of the tryprostatins and their diastereomers on human carcinoma cell lines. J Med Chem 45:1559–1562
Zhao Y, Liu J, Wang J, Wang L, Yin H, Tan R, Xu Q (2004) Fumigaclavine C improves concanavalin A-induced liver injury in mice mainly via inhibiting TNF-alpha production and lymphocyte adhesion to extracellular matrices. J Pharm Pharmacol 56:775–782
Zou H, Zheng X, Li S-M (2009) Substrate promiscuity of the cyclic dipeptide prenyltransferases from Aspergillus fumigatus. J Nat Prod 72:44–52
Acknowledgement
The works in the author's laboratory were supported by the Deutsche Forschungsgemeinschaft (SPP1152).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, SM. Applications of dimethylallyltryptophan synthases and other indole prenyltransferases for structural modification of natural products. Appl Microbiol Biotechnol 84, 631–639 (2009). https://doi.org/10.1007/s00253-009-2128-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2128-z