Abstract
Key message
Using association and linkage mapping, two SNP markers closely linked to the SBWMV resistance gene on chromosome 5D were identified and can be used to select the gene in breeding.
Abstract
Soil-borne wheat mosaic virus (SBWMV) disease is a serious viral disease of winter wheat growing areas worldwide. SBWMV infection can significantly reduce grain yield up to 80 %. Developing resistant wheat cultivars is the only feasible strategy to reduce the losses. In this study, wheat Infinium iSelect Beadchips with 9 K wheat SNPs were used to genotype an association mapping population of 205 wheat accessions. Six new SNPs from two genes were identified to be significantly associated with the gene for SBWMV resistance on chromosome 5D. The SNPs and Xgwm469, an SSR marker that has been reported to be associated with the gene, were mapped close to the gene using F6-derived recombinant inbred lines from the cross between a resistant parent ‘Heyne’ and a susceptible parent ‘Trego’. Two representative SNPs, wsnp_CAP11_c209_198467 and wsnp_JD_c4438_5568170, from the two linked genes in wheat were converted into KBioscience Competitive Allele-Specific Polymerase assays and can be easily used in marker-assisted selection to improve wheat resistance to SBWMV in breeding.



Similar content being viewed by others
References
Allen AM, Barker GL, Berry ST, Coghill JA, Gwilliam R et al (2011) Transcriptspecific, single nucleotide polymorphism discovery and linkage analysis in hexaploid bread wheat (Triticum aestivum L.). Plant Biotechnol J 9:1086–1099
Atwell S, Huang YS, Vilhjalmsson BJ, Willems G, Horton M et al (2010) Genome-wide association study of 107 phenotypes in Arabidopsis thaliana inbred lines. Nature 465:627–631
Barbosa M, Goulart L, Prestes A, Juliatti F (2001) Genetic control of resistance to Soil-borne wheat mosaic virus in Brazilian cultivars of Triticum aestivum L. Thell Euphytica 122:417–422
Bass C, Hendley R, Adams MJ, Hammond-Kosack KE, Kanyuka K (2006) The Sbm1 locus conferring resistance to Soil-borne cereal mosaic virus maps to a gene-rich region on 5DL in wheat. Genome 49:1140–1148
Bever WM, Pendleton JW (1954) The effect of soil-borne wheat mosaic on yield of winter wheat. Plant Dis Rep 38:266–267
Bradbury PJ, Zhang Z, Kroon DE, Casstevens TM, Ramdoss Y, Buckler ES (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23:2633–2635
Brenchley R, Spannagl M, Pfeifer M, Barker GLA, D’Amore R, Allen AM et al (2012) Analysis of the bread wheat genome using whole-genome shotgun sequencing. Nature 491:705–710
Byers RL, Harker DB, Yourstone SM, Maughan PJ, Udall JA (2012) Development and mapping of SNP assays in allotetraploid cotton. Theor Appl Genet 124:1201–1214
Cadle-Davidson L, Sorrells ME, Gray SM, Bergstrom GC (2006) Identification of small grains genotypes resistant to Soil-borne mosaic virus. Plant Dis 90:1039–1044
Cavanagh CR, Chao S, Wang S, Huang BE, Stephen S et al (2013) Genome-wide comparative diversity uncovers multiple targets of selection for improvement in hexaploid wheat landraces and cultivars. PNAS 110:8057–8062
Diao A, Chen J, Gitton F, Antoniw JF, Mullins J, Hall AM, Adams MJ (1999) Sequences of European Wheat mosaic virus and Oat golden stripe virus and genome analysis of the genus Furovirus. Virology 261:331–339
Driskel BA, Hunger RM, Payton ME, Verchot-Lubicz J (2002) Response of hard red winter wheat to Soil-borne wheat mosaic virus using novel inoculation methods. Phytopathology 92:347–354
Hall MD, Brown-Guedira G, Klatt A, Fritz AK (2009) Genetic analysis of resistance to Soil-borne wheat mosaic virus derived from Aegilops tauschii. Euphytica 169:169–176
Hao Y, Wang Y, Chen Z, Bland D, Li S, Brown-Guedira G, Johnson J (2012) A conserved locus conditioning Soilborne wheat mosaic virus resistance on the long arm of chromosome 5D in common wheat. Mol Breed 30:1453–1464
Hariri D, Courtillot M, Zaoui P, Lapierre H (1987) Multiplication of Soil-borne wheat mosaic virus (SBWMV) in wheat roots infected by a soil carrying SBWMV and wheat yellow mosaic virus (WYMV). Agronomie 7:789–796
Huang X, Wei X, Sang T, Zhao Q, Feng Q et al (2010) Genome-wide association studies of 14 agronomic traits in rice landraces. Nat Genet 42:961–967
Johnson F (1942) Heat inactivation of wheat mosaic virus in soils. Science 95:610
Kapooria RG, Ndunguru J, Clover GRG (2000) First reports of Soilborne wheat mosaic virus and Wheat spindle streak mosaic virus in Africa. Plant Dis 84:921
Koehler B, Bever WM, Bonnett OT (1952) Soilborne wheat mosaic. University of Illinois Agricultural Experiment Station in cooperation with U.S. Dept. of Agric Bull 556:565–599
Koenig R, Huth W (2000) Soilborne rye mosaic and European wheat mosaic virus: two names for a furovirus with variable genome properties which are widely distributed in several cereal crops in Europe. Arch Virol 145:689–697
Koenig R, Huth W (2003) Natural infection of wheat by the type strain of Soilborne wheat mosaic virus in New Zealand. Australas Plant Pathol 38:85–90
Kosambi DD (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Krill AM, Kirst M, Kochian LV, Buckler ES, Hoekenga OA (2010) Association and linkage analysis of aluminum tolerance genes in maize. PLoS One 5:e9958
Kulwal P, Ishikawa G, Benscher D, Feng Z, Yu L, Jadhav A, Mehetre S, Sorrells ME (2012) Association mapping for pre-harvest sprouting resistance in white winter wheat. Theor Appl Genet 125:793–805
Kump KL, Bradbury PJ, Wisser RJ, Buckler ES, Belcher AR et al (2011) Genome-wide association study of quantitative resistance to southern leaf blight in the maize nested association mapping population. Nat Genet 43:163–168
Lebas BSM, Ochoa-Corona FM, Elliott DR, Tang J, Blouin AG, Timudo OE, Ganev S, Alexander BJR (2009) Investigation of an outbreak of Soil-borne wheat mosaic virus in New Zealand. Australas Plant Pathol 38:85–90
Letta T, Maccaferri M, Badebo A, Ammar K, Ricci A, Crossa J, Tuberosa R (2013) Searching for novel sources of field resistance to Ug99 and Ethiopian stem rust races in durum wheat via association mapping. Theor Appl Genet 126:1237–1256
Liu S, Cai S, Graybosch R, Chen C, Bai G (2008) Quantitative trait loci for resistance to pre-harvest sprouting in US hard white winter wheat Rio Blanco. Theor Appl Genet 117:691–699
Maccaferri M, Ratti C, Rubies-Autonell C, Vallega V, Demontis A et al (2011) Resistance to Soil-borne cereal mosaic virus in durum wheat is controlled by a major QTL on chromosome arm 2BS and minor loci. Theor Appl Genet 123:527–544
Malosetti M, van der Linden CG, Vosman B, van Eeuwijk FA (2007) A mixed-model approach to association mapping using pedigree information with an illustration of resistance to Phytophthora infestans in potato. Genetics 175:879–889
McKinney HH (1923) Investigations on the rosette disease of wheat and its control. J Agric Res 23:771–800
Merkle OG, Smith EL (1983) Inheritance of resistance to soilborne mosaic in wheat. Crop Sci 23:1075–1076
Miyake M (1938) Mendelian inheritance of resistance to mosaic disease in wheat varieties. Jpn J Genet 14:232–239
Modawi RS, Heyne EG, Brunetta D, Willis WG (1982) Genetic studies of field reaction to wheat Soilborne mosaic virus. Plant Dis 66:1183–1184
Myers LD, Sherwood JL, Siegerist WC, Hunger RM (1993) Temperature-influenced virus movement in expression of resistance to Soilborne wheat mosaic virus in hard red winter wheat (Triticum aestivum). Phytopathology 83:548–551
Myles S, Peiffer J, Brown PJ, Ersoz ES, Zhang Z et al (2009) Association mapping: critical considerations shift from genotyping to experimental design. Plant Cell 21:2194–2202
Narasimhamoorthy B, Gill BS, Fritz AK, Nelson JC, Brown-Guedira GL (2006) Advanced backcross QTL analysis of a hard winter wheat × synthetic wheat population. Theor Appl Genet 112:787–796
Nijman IJ, Kuipers S, Verheul M, Guryev V, Cuppen E (2008) A genome-wide SNP panel for mapping and association studies in the rat. BMC Genom 9:95
Perovic D, Förster J, Devaux P, Hariri D, Guilleroux M et al (2009) Mapping and diagnostic marker development for Soil-borne cereal mosaic virus resistance in bread wheat. Mol Breed 23:641–653
Pritchard JK, Stephens M, Donnelly PJ (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Reif JC, Gowda M, Maurer HP, Longin CFH (2011) Korzun K (2011) Association mapping for quality traits in soft winter wheat. Theor Appl Genet 122:961–970
Sawada E (1927) Control of Wheat yellow mosaic virus. J Plant Prot 14:444–449
Semagn K, Babu R, Hearne S, Olsen M (2013) Single nucleotide polymorphism genotyping using Kompetitive Allele Specific PCR (KASP): overview of the technology and its application in crop improvement. Mol Breed. doi:10.1007/s11032-013-9917-x
Shaalan MI, Heyne EG, Sill WH (1966) Breeding wheat for resistance to Soil-borne wheat mosaic virus, Wheat streak-mosaic virus, leaf rust, stem rust, and bunt. Phytopathology 56:664–669
Shirako Y, Suzuki N, French RC (2000) Similarity and divergence among viruses in the genus Furovirus. Virology 270:201–207
Somers DJ, Isaac P, Edwards K (2004) A high-density wheat microsatellite consensus map for bread wheat (Triticum aestivum L.). Theor Appl Genet 109:1105–1114
Terracciano I, Maccaferri M, Bassi F, Mantovani P, Sanguineti MC et al (2013) Development of COS-SNP and HRM markers for high throughput and reliable haplotype-based detection of Lr14a in durum wheat (Triticum durum Desf.). Theor Appl Genet 126:1077–1101
Toojinda T, Baird E, Booth A, Broers L, Hayes P (1998) Introgression of quantitative trait loci (QTLs) determining stripe rust resistance in barley: an example of marker-assisted line development. Theor Appl Genet 96:123–131
Van Ooijen JW, Voorrips RE (2001) JionMap version 3.0: Software for the calculation of genetic linkage maps
Zhang D, Bai G, Zhu C, Yu J, Carver BF (2010) Genetic diversity, population structure, and linkage disequilibrium in US elite winter wheat. Plant Gen 3:117–127
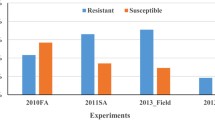
Zhang D, Bai G, Hunger RM, Bockus WW, Yu J, Carver BF, Brown-Guedira G (2011) Association study of resistance to Soilborne wheat mosaic virus in US winter wheat. Phytopathology 101:1322–1329
Zhang XH, Pan HY, Bai GH (2012) Quantitative trait loci for Fusarium head blight resistance in US hard winter wheat cultivar ‘Heyne’. Crop Sci 52:1187–1194
Zhu C, Gore M, Buckler E, Yu J (2008) Status and prospects of association mapping in plants. Plant Gen 1:5–20
Acknowledgments
This is contribution number 14-056-J from the Kansas Agricultural Experiment Station, Manhattan, KS, USA. This project is partly funded by the National Research Initiative Competitive Grants CAP project 2011- 68002-30029 from the USDA National Institute of Food and Agriculture. The mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. E. Sorrells.
Electronic supplementary material
Below is the link to the electronic supplementary material.
122_2014_2277_MOESM1_ESM.docx
Supplemental Fig. 1 Linkage disequilibrium analysis indicated the six SNPs were within a strong linkage block with the marker Xgwm469 on wheat chromosome 5DL. The linkage map from Somers et al. (2004) was used as a reference map to indicate that only Xgwm469 had LD with the six SNPs, not other two adjacent markers (Xcfd10 and Xgwm565). Supplemental Fig. 2 KASP assay profiling of SNP wsnp_CAP11_c209_198467 (A) and SNP wsnp_JD_c4438_5568170 (B) in 205 wheat accessions. (A). Allele X (KASPFAM, blue color) represents the A nucleotide, Allele Y (KASPHEX, green color) represents the G nucleotide. (B). Allele X (KASPFAM, blue color) represents the A nucleotide, Allele Y (KASPHEX, green color) represents the G nucleotide. The black dots and crosses in the circle represent water controls and missing data. Supplementary material 1 (DOCX 287 kb)
Rights and permissions
About this article
Cite this article
Liu, S., Yang, X., Zhang, D. et al. Genome-wide association analysis identified SNPs closely linked to a gene resistant to Soil-borne wheat mosaic virus. Theor Appl Genet 127, 1039–1047 (2014). https://doi.org/10.1007/s00122-014-2277-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-014-2277-z