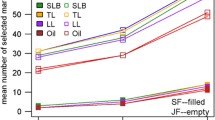
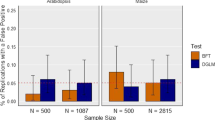
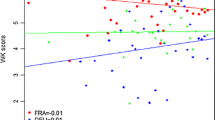
Abstract
Pleiotropy has played an important role in understanding quantitative traits. However, the extensiveness of this effect in the genome and its consequences for plant improvement have not been fully elucidated. The aim of this study was to identify pleiotropic quantitative trait loci (QTLs) in maize using Bayesian multiple interval mapping. Additionally, we sought to obtain a better understanding of the inheritance, extent and distribution of pleiotropic effects of several components in maize production. The design III procedure was used from a population derived from the cross of the inbred lines L-14-04B and L-08-05F. Two hundred and fifty plants were genotyped with 177 microsatellite markers and backcrossed to both parents giving rise to 500 backcrossed progenies, which were evaluated in six environments for grain yield and its components. The results of this study suggest that mapping isolated traits limits our understanding of the genetic architecture of quantitative traits. This architecture can be better understood by using pleiotropic networks that facilitate the visualization of the complexity of quantitative inheritance, and this characterization will help to develop new selection strategies. It was also possible to confront the idea that it is feasible to identify QTLs for complex traits such as grain yield, as pleiotropy acts prominently on its subtraits and as this “trait” can be broken down and predicted almost completely by the QTLs of its components. Additionally, pleiotropic QTLs do not necessarily signify pleiotropy of allelic interactions, and this indicates that the pervasive pleiotropy does not limit the genetic adaptability of plants.





Similar content being viewed by others
References
Banerjee S, Yandell BS, Yi N (2008) Bayesian quantitative trait loci mapping for multiple traits. Genetics 179:2275–2289
Buchanan AV, Sholtis S, Richtsmeier J, Weiss KM (2009) What are genes “for” or where are traits “from”? What is the question? Bioessays 31:198–208
Buckler ES, Holland JB, Bradbury PJ et al (2009) The genetic architecture of maize flowering time. Science 325:714–718
Carbone MA, Jordan KW, Lyman RF et al (2006) Phenotypic variation and natural selection at Catsup, a pleiotropic quantitative trait gene in Drosophila. Curr Biol 16:912–919
Cheverud JT, Ehrich H, Vaughn TT et al (2004) Pleiotropic effects on mandibular morphology II: differential epistasis and genetic variation in morphological integration. J Exp Zool 302:424–435
Flint J, Mackay TFC (2009) Genetic architecture of quantitative traits in mice, flies, and humans. Genome Res 19:723–773
Frascaroli E, Canè MA, Landi PG et al (2007) Classical genetic and quantitative trait loci analyses of heterosis in a maize hybrid between two elite inbred lines. Genetics 176:625–644
Garcia AA, Wang S, Melchinger AE, Zeng ZB (2008) Quantitative trait mapping and the genetic basis of heterosis in maize and rice. Genetics 180:1707–1724
George EI, McCulloch RE (1993) Variable selection via Gibbs Sampling. J Am Stat Assoc 88:881–889
Gilbert H, Le Roy P (2003) Comparison of three multitrait methods for QTL detection. Genet Sel Evol 35:281–304
Gore MA, Chia JM, Elshire R et al (2009) A first-generation haplotype map of maize. Science 326:1115–1117
Hastings WK (1970) Monte Carlo sampling methods using Markov chains and their applications. Biometrika 57:97–109
Jiang C, Zeng Z (1995) Multiple trait analysis of genetic mapping for quantitative trait loci. Genetics 140:1111–1127
Joehanes R, Nelson JC (2008) QGene 4.0, an extensible Java QTL-analysis platform. Bioinformatics 24:2788–2789
Lincoln SE, Daly MJ, Lander ES (1992) Constructing genetic maps with Mapmaker Exp 3.0, 3rd edn. Whitehead Institute for Biometrical Research, Cambridge
Liu J, Liu Y, Liu X, Deng HW (2007) Bayesian mapping of quantitative trait loci for multiple complex traits with the use of variance components. Am J Hum Genet 81:304–320
Lu H, Romero-Severson J, Bernardo R (2003) Genetics basis of heterosis explored by simple sequence repeat markers in a random-mated maize population. Theor Appl Genet 107:494–502
Mackay TFC (2001) The genetic architecture of quantitative traits. Annu Rev Genet 35:336–339
Mackay TFC, Stone EA, Ayroles JF (2009) The genetics of quantitative traits: challenges and prospects. Nat Rev Genet 10:565–577
Malosetti M, Ribaut JM, Vargas M, Crossa J, Van Eeuwijk FAA (2008) Multi-trait multi-environment QTL mixed model with an application to drought and nitrogen stress trials in maize (Zea mays L.). Euphytica 161:241–257
Melchinger AE, Utz HF, Piepho HP, Zeng ZB, Schön CC (2007) The role of epistasis in the manifestation of heterosis: a systems-oriented approach. Genetics 177:1815–1825
Metropolis N, Rosemblut AW, Rosemblut MN, Teller AH, Teller E (1953) Equations of state calculations by fast computing machines. J Chem Phys 21:1087–1092
Pavlicev M, Kenney-Hunt JP, Norgard EA et al (2008) Genetic variation in pleiotropy: differential epistasis as a source of variation in the allometric relationship between long bone lengths and body weight. Evolution 62:199–213
Raftery AE, Newton MA, Satagopan J, Krivitsky P (2007) Estimating the integrated likelihood via posterior simulation using the harmonic mean identity (with discussion). Bayesian Stat 8:1–45
Schnell FW, Cockerham CC (1992) Multiplicative vs. arbitrary gene action in heterosis. Genetics 131:461–469
Schön CC, Dhillon BS, Utz HF, Melchinger AE (2010) High congruency of QTL positions for heterosis of grain yield in three crosses of maize. Theor Appl Genet 120:321–332
Shannon P, Markiel A, Ozier O et al (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504
Sibov ST, Souza-Júnior CL, Garcia AAF et al (2003) Molecular mapping in tropical maize (Zea mays L.) using microsatellite markers. 1. Map construction and localization of loci showing distorted segregation. Hereditas 139:96–106
Stearns FW (2010) One hundred years of pleiotropy: a retrospective. Genetics 186:767–773
Su Z, Zeng Y, Gu X (2010) A preliminary analysis of gene pleiotropy estimated from protein sequences. J Exp Zool 314:115–122
Van Eeuwijk FA, Boer M, Totir RL et al (2010) Mixed model approaches for the identification of QTLs within a maize hybrid breeding program. Theor Appl Genet 120:429–440
Varona L, Gomez-Raya L, Rauw WM, Clop A, Ovilo C, Noguera JL (2004) Derivation of a Bayes factor to distinguish between linked or pleiotropic quantitative trait loci. Genetics 166:1025–1035
Wagner GP, Zhang J (2011) The pleiotropic structure of the genotype–phenotype map: the evolvability of complex organisms. Nat Rev Genet 12:204–213
Wagner GP, Kenney-Hunt JP, Pavlicev M et al (2008) Pleiotropic scaling of gene effects and the ‘cost of complexity’. Nature 27:470–472
Wang H, Zhang YM, Li X et al (2005) Bayesian shrinkage estimation of quantitative trait loci parameters. Genetics 170:465–480
Wang Z, Liao BY, Zhang J (2010) Genomic patterns of pleiotropy and the evolution of complexity. Proc Natl Acad Sci 107:18034–18039
Weller JI, Wiggans GR, Vanraden PM, Ron M (1996) Application of a canonical transformation to detection of quantitative trait loci with the aid of genetic markers in a multitrait experiment. Theor Appl Genet 92:998–1002
Wolf JB, Leamy LL, Routman EJ, Cheverud JM (2005) Epistatic pleiotropy and the genetic architecture of covariation within early and late-developing skull trait complexes in mice. Genetics 171:1–12
Wolf JB, Pomp D, Eisen EJ, Cheverud JM, Leamy LL (2006) The contribution of epistatic pleiotropy to the genetic architecture of covariation among polygenic traits in mice. Evol Dev 8:468–476
Wright S (1968) Evolution and the genetics of populations. University of Chicago Press, Chicago
Xu S (2003) Estimating polygenic effects using markers of the entire genome. Genetics 163:789–801
Xu C, Wang X, Li Z, Xu S (2009) Mapping QTL for multiple traits using Bayesian statistics. Genet Res 91:23–37
Yang RC (2004) Epistasis of quantitative trait loci under different gene action models. Genetics 167:1493–1505
Yang R, Xu S (2007) Bayesian shrinkage analysis of quantitative trait loci for dynamic traits. Genetics 176:1169–1175
Zeng ZB, Wang T, Zou W (2005) Modeling quantitative trait loci and interpretation of models. Genetics 169:1711–1725
Acknowledgments
Authors would like to thank Professor Anete Pereira de Souza of the Department of Biology and Evolution of the State University of Campinas for genotyping the population. Criticisms of both referees were very insightful and made this a better paper. Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) has supported this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Carbonell.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supporting Fig 1 Behavior of Bayes Factor in selection of Pleiotropic or Linkage model (BMP 1739 kb)
122_2012_1847_MOESM2_ESM.bmp
Supporting Fig 2 QTL location (blue arrows) for pleiotropic model (A) and linkage model (B). One pleiotropic QTL (28 cM) and two linked QTLs (70 and 78 cM) acting on two simulated traits (trait 1 and trait 2) (BMP 3165 kb)
122_2012_1847_MOESM3_ESM.bmp
Supporting Fig 3 Estimates QTL intensity location for the linkage model (A) and the pleiotropic model using Jiang and Zeng (1995) method (B). One pleiotropic QTL (28 cM) and two linked QTLs (70 and 78 cM) acting on two simulated trait (trait 1 and trait 2) (BMP 806 kb)
Rights and permissions
About this article
Cite this article
Balestre, M., Von Pinho, R.G., de Souza Junior, C.L. et al. Bayesian mapping of multiple traits in maize: the importance of pleiotropic effects in studying the inheritance of quantitative traits. Theor Appl Genet 125, 479–493 (2012). https://doi.org/10.1007/s00122-012-1847-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-012-1847-1