Abstract
Objective
Although the effects of tacrolimus on T cells are well-known, direct effects on rheumatoid synovial fibroblasts (RSF) remain unclear. We studied the effects of tacrolimus on RSF by a DNA microarray analysis.
Materials and methods
Tacrolimus and interleukin (IL)-1β were added to cultured RSF. Total RNA was prepared from the cells and the gene expression profile was analyzed by a DNA microarray screening system. mRNA expressions influenced by tacrolimus in the screening system were confirmed by real-time PCR. The effects of tacrolimus on nuclear translocation of nuclear factor-κB (NF-κB) were also examined.
Results
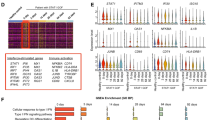
The mRNA expressions of CCL3, CCL4, and CXCL8 were up-regulated by IL-1β and down-regulated by tacrolimus. The levels of these IL-1β-induced chemokines in culture supernatant were decreased by a therapeutic concentration of tacrolimus. Tumor necrosis factor-α as well as IL-1β induced these chemokines, while tacrolimus inhibited their production and mRNA expression. Chemotaxis of polymorphonuclear cells in response to IL-1β was also inhibited by tacrolimus. Nuclear translocation of p50 and p65 NF-κB in response to IL-1β was decreased by tacrolimus.
Conclusion
IL-1β-induced chemokine expressions were down-regulated by tacrolimus, suggesting that tacrolimus exerts its anti-inflammatory effect partly through inhibiting chemokine production by RSF.







Similar content being viewed by others
References
Kino T, Hatanaka H, Miyata S, Inamura N, Nishiyama M, Yajima T, et al. FK-506, a novel immunosuppressant isolated from a Streptomyces. II. Immunosuppressive effect of FK-506 in vitro. J Antibiot. 1987;40(9):1256–65.
Kelly PA, Burckart GJ, Venkataramanan R. Tacrolimus: a new immunosuppressive agent. Am J Health Syst Pharm. 1995;52(14):1521–35.
Knoll G. Trends in kidney transplantation over the past decade. Drugs. 2008;68 (Suppl 1):3–10.
Kawai S, Hashimoto H, Kondo H, Murayama T, Kiuchi T, Abe T. Comparison of tacrolimus and mizoribine in a randomized, double-blind controlled study in patients with rheumatoid arthritis. J Rheumatol. 2006;33(11):2153–61.
Kondo H, Abe T, Hashimoto H, Uchida S, Irimajiri S, Hara M, et al. Efficacy and safety of tacrolimus (FK506) in treatment of rheumatoid arthritis: a randomized, double blind, placebo controlled dose-finding study. J Rheumatol. 2004;31(2):243–51.
Kitahara K, Kawai S. Cyclosporine and tacrolimus for the treatment of rheumatoid arthritis. Curr Opin Rheumatol. 2007;19(3):238–45.
Kawai S. Current drug therapy for rheumatoid arthritis. J Orthop Sci. 2003;8:259–63.
Koch AE. Chemokines and their receptors in rheumatoid arthritis: future targets? Arthritis Rheum. 2005;52(3):710–21.
Kitahara K, Kusunoki N, Kakiuchi T, Suguro T, Kawai S. Adiponectin stimulates IL-8 production by rheumatoid synovial fibroblasts. Biochem Biophys Res Commun. 2009;378(2):218–23.
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31(3):315–24.
Frevert CW, Wong VA, Goodman RB, Goodwin R, Martin TR. Rapid fluorescence-based measurement of neutrophil migration in vitro. J Immunol Methods. 1998;213(1):41–52.
Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65(1–2):55–63.
Baksh S, Burakoff SJ. The role of calcineurin in lymphocyte activation. Semin Immunol. 2000;12(4):405–15.
Wolpe SD, Davatelis G, Sherry B, Beutler B, Hesse DG, Nguyen HT, et al. Macrophages secrete a novel heparin-binding protein with inflammatory and neutrophil chemokinetic properties. J Exp Med. 1988;167(2):570–81.
Szekanecz Z, Strieter RM, Kunkel SL, Koch AE. Chemokines in rheumatoid arthritis. Springer Semin Immunopathol. 1998;20(1–2):115–32.
Koch AE, Kunkel SL, Harlow LA, Mazarakis DD, Haines GK, Burdick MD, et al. Macrophage inflammatory protein-1 alpha. A novel chemotactic cytokine for macrophages in rheumatoid arthritis. J Clin Invest. 1994;93(3):921–8.
Chintalacharuvu SR, Wang JX, Giaconia JM, Venkataraman C. An essential role for CCL3 in the development of collagen antibody-induced arthritis. Immunol Lett. 2005;100(2):202–4.
Patel DD, Zachariah JP, Whichard LP. CXCR3 and CCR5 ligands in rheumatoid arthritis synovium. Clin Immunol 2001;98(1):39–45.
Pokkali S, Das SD. Augmented chemokine levels and chemokine receptor expression on immune cells during pulmonary tuberculosis. Hum Immunol. 2009;70(2):110–5.
Ribeiro S, Horuk R. The clinical potential of chemokine receptor antagonists. Pharmacol Ther. 2005;107(1):44–58.
Vierboom MP, Zavodny PJ, Chou CC, Tagat JR, Pugliese-Sivo C, Strizki J, et al. Inhibition of the development of collagen-induced arthritis in rhesus monkeys by a small molecular weight antagonist of CCR5. Arthritis Rheum. 2005;52(2):627–36.
Szekanecz Z, Kim J, Koch AE. Chemokines and chemokine receptors in rheumatoid arthritis. Semin Immunol. 2003;15(1):15–21.
Patterson AM, Schmutz C, Davis S, Gardner L, Ashton BA, Middleton J. Differential binding of chemokines to macrophages and neutrophils in the human inflamed synovium. Arthritis Res. 2002;4(3):209–14.
Hayashida K, Nanki T, Girschick H, Yavuz S, Ochi T, Lipsky PE. Synovial stromal cells from rheumatoid arthritis patients attract monocytes by producing MCP-1 and IL-8. Arthritis Res. 2001;3(2):118–26.
Hogan M, Sherry B, Ritchlin C, Fabre M, Winchester R, Cerami A, et al. Differential expression of the small inducible cytokines GRO alpha and GRO beta by synovial fibroblasts in chronic arthritis: possible role in growth regulation. Cytokine. 1994;6(1):61–9.
Seitz M, Dewald B, Ceska M, Gerber N, Baggiolini M. Interleukin-8 in inflammatory rheumatic diseases: synovial fluid levels, relation to rheumatoid factors, production by mononuclear cells, and effects of gold sodium thiomalate and methotrexate. Rheumatol Int. 1992;12(4):159–64.
Koch AE, Kunkel SL, Burrows JC, Evanoff HL, Haines GK, Pope RM, et al. Synovial tissue macrophage as a source of the chemotactic cytokine IL-8. J Immunol. 1991;147(7):2187–95.
Endo H, Akahoshi T, Takagishi K, Kashiwazaki S, Matsushima K. Elevation of interleukin-8 (IL-8) levels in joint fluids of patients with rheumatoid arthritis and the induction by IL-8 of leukocyte infiltration and synovitis in rabbit joints. Lymphokine Cytokine Res. 1991;10(4):245–52.
Harada A, Sekido N, Akahoshi T, Wada T, Mukaida N, Matsushima K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. J Leukoc Biol. 1994;56(5):559–64.
Koch AE, Polverini PJ, Kunkel SL, Harlow LA, DiPietro LA, Elner VM et al. Interleukin-8 as a macrophage-derived mediator of angiogenesis. Science. 1992;258(5089):1798–801.
Yoo SA, Park BH, Park GS, Koh HS, Lee MS, Ryu SH, et al. Calcineurin is expressed and plays a critical role in inflammatory arthritis. J Immunol. 2006;177(4):2681–90.
Okamoto S, Mukaida N, Yasumoto K, Rice N, Ishikawa Y, Horiguchi H, et al. The interleukin-8 AP-1 and kB-like sites are genetic end targets of FK506-sensitive pathway accompanied by calcium mobilization. J Biol Chem. 1994;269:8582–9.
Simmonds RE, Foxwell BM. Signalling, inflammation and arthritis: NF-kappaB and its relevance to arthritis and inflammation. Rheumatology. 2008;47(5):584–90.
Du S, Hiramatsu N, Hayakawa K, Kasai A, Okamura M, Huang T, et al. Suppression of NF-kappaB by cyclosporin a and tacrolimus (FK506) via induction of the C/EBP family: implication for unfolded protein response. J Immunol. 2009;182(11):7201–11.
Acknowledgments
This work was supported in part by a Project Research Grant (20-12) and Dr. Takeshi Yanase’s grant from Toho University School of Medicine to K.K.; Heisei 20 and 23 Strategic Research Foundation Grants from the Japanese Ministry of Education, Culture, Sports, Science & Technology to Toho University; Grants from Japan Society for the Promotion of Science (20591177, 23591449) to S.K.; and Grants from Nakatomi Foundation and Takeda Science Foundation to N.K. We wish to thank Ms. Sonoko Sakurai for secretarial assistance.
Conflict of interest
S.K. has received honoraria for lectures and research grants from Astellas Pharma Inc. (Tokyo, Japan), the manufacturer of tacrolimus. The other authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Ikuo Morita.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kitahara, K., Kusunoki, N., Takahashi, H. et al. Tacrolimus down-regulates chemokine expressions on rheumatoid synovial fibroblasts: screening by a DNA microarray. Inflamm. Res. 61, 1385–1394 (2012). https://doi.org/10.1007/s00011-012-0541-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-012-0541-8