Abstract
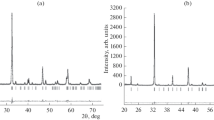
Chemical and structural properties of the mixed metal oxides (1−x)Fe2O3+xCr2O3 were studied by different techniques. X-ray powder diffraction showed the existence of solid solutions, (Fe1−x Cr x )2O3, over the whole concentration region, 0⩽x⩽1. The gradual replacement of Fe3+ with Cr3+ ions in samples prepared at 900°C caused changes in unit-cell parameters; most of these changes took place in the region fromx≈0.3–0.9. The samples having the fraction of Cr2O3 in the region from ∼0.7–0.8, contained two closely related phases, with slightly different compositions. After an additional heat treatment at 1100°C, these samples contained only one phase.57Fe Mössbauer spectroscopy showed a gradual decrease of hyperfine magnetic field with increasing Cr2O3 content. The sample having the fraction of Cr2O3 of 0.7, and prepared at 900°C, exhibited two separated sextets at room temperature, in comparison with other compositions showing one sextet. It was shown that Fourier transform infrared (FT-IR) spectroscopy is a powerful method for the investigation of structural changes in these solid solutions. The increase in the Cr2O3 content resulted in shifts of the corresponding infrared bands. In addition, a gradual transition of the spectrum typical for α-Fe2O3 to the spectrum typical for Cr2O3 was shown. The transition effects observed in the FT-IR spectra were correlated with the X-ray powder diffraction and57Fe Mössbauer spectroscopic results.
Similar content being viewed by others
References
S. Musić, S. Popović andM. Ristić,J. Mater. Sci. 24 (1989) 2722.
M. Ristić, S. Popović, M. Tonković andS. Musić ibid. 26 (1991) 4225.
S. Musić, S. Popović, M. Metikoš-Huković andV. Gvozdić,J. Mater. Sci. Lett. 10 (1991) 197.
M. Ristić, S. Popović andS. Musić,ibid. 9 (1900) 872.
S. Musić, V. Ilakovac, M. Ristić andS. Popović,J. Mater. Sci. 27 (1992) 1011.
International Centre for Diffraction Data, Joint Committee on Powder Diffraction Standards, Powder Diffraction File, 1601 Park Lane, Swarthmore, PA, 19081, USA.
J. K. Srivastava andR. P. Sharma,J. Physique 35 (1974) C6–663.
J. K. Srivastava andS. Muralidhara Rao,Phys. Status Solidi (b)90 (1978) KI75.
G. Doppler, A. X. Trautwein, H. M. Ziethen, E. Ambach, R. Lehnert andM. J. Sprague,Appl. Catal. 40 (1989) 119.
D. G. Rethwisch, J. Phillips, Y. Chen, T. F. Hayden andJ. A. Dumesic,J. Catal. 91 (1985) 167.
C. J. Serna, J. L. Rendon andJ. E. Iglesias,Spectrochim. Acta 38A (1982) 797.
J. E. Iglesias, M. Ocana andC. J. Serna,Appl Spectros. 44 (1990) 418.
D. R. Renneke andD. W. Lynch,Phys. Rev. 138 (1965) A530.
D. K. Ottesen,J. Electrochem. Soc. 132 (1985) 2250.
M. Lenglet, R. Guillamet, J. Lopitaux andB. Hannoyer,Mater. Res. Bull. 25 (1990) 575.
B. Gillot, F. Bouton, F. Chassagneux andA. Rousset,J. Solid State Chem. 33 (1980) 245.
B. Gillot, F. Bouton, J. F. Ferriot, F. Chassagneux andA. Rousset,ibid. 21 (1977) 375.
B. Gillot, F. Jemmali andA. Rousset,ibid. 50 (1983) 138.
F. Chassagneux, A. Rousset andJ. P. Redoules,ibid. 56 (1985) 74.
B. Gillot, F. Jemmali, M. Laarj, F. Chassagneux andA. Rousset,J. Mater. Sci. 23 (1988) 872.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Musić, S., Popović, S. & Ristić, M. Chemical and structural properties of the system Fe2O3-Cr2O3 . J Mater Sci 28, 632–638 (1993). https://doi.org/10.1007/BF01151237
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01151237