Abstract
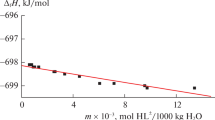
Integral enthalpies of solution of some sugars and polyols in water at low concentrations have been determined calorimetrically at 25 and 35°C. These data have been used to derive heat capacities of solution ΔC°p at 30°C. Partial molal heat capacities C°p,2 have been obtained by combining ΔC°p with C *p,2 , the heat capacity of pure solid compounds. Apparent molal volumes have been obtained from density data. The sugars as well as polyols show significantly high positive ΔC°p and C°p,2 values. The results have been explained in terms of a specific hydration model. The effect of substitution of-OH by glycosidic-OCH3 and of-CHOH by deoxy-CH2 are also discussed.
Similar content being viewed by others
References
B. Philipp, W. Bock, and F. Schierbaum,J. Polymer Sci. Polymer Symposium,66, 83 (1979).
D. A. Rees,Int. Rev. of Science, Biochemistry Series 5, 1 (1975).
A. Suggett,J. Solution Chem. 5, 33 (1976) and references therein.
F. Franks, J. R. Ravenhill, and D. S. Reid,J. Solution Chem. 1, 3 (1972).
T. S. Sarma, R. K. Mohanty, and J. C. Ahluwalia,Trans. Faraday Soc. 65, 2333 (1969).
V. B. Parker,Thermal Properties of Aqueous Un-univalent Electrolytes, NSRDS. NBS-2, Washington D.C. (1965).
S. Gunn,J. Phys. Chem. 69, 2902 (1965).
G. S. Kell,J. Chem. Eng. Data 15, 119 (1970).
H. S. Isbell and W. Pigmann,Adv. in Carbohydrate Chem. 24, 14 (1969);23, 11 (1968).
J. M. Sturtevant,J. Phys. Chem. 45, 127 (1941).
M. A. Kabayama, D. Patterson, and L. Piche,Can. J. Chem. 36, 557, 563 (1958).
J. B. Taylor and J. S. Rowlinson,Trans. Faraday Soc. 51, 1183 (1955).
G. S. Parks and K. E. Manchester,J. Am. Chem. Soc. 74, 3435 (1952).
Int. Critical Tables of Numerical Data, Phys., Chem., and Technology, Vol. V, E. W. Washburn, ed. (Mcgraw-Hill, New York, 1929), p. 102.
J. Timmerman,Physico-Chemical Constants of the Pure Organic Compounds, (Elsevier, 1950).
CRC Handbook of Chemistry and Physics, 59th edition, R. C. Weast, ed. (Chemical Rubber Company, Cleveland, 1978–79).
F. Kawaizumi, N. Nishio, H. Nomura, and Y. Miyahara,J. Chem. Thermodyn. 13, 89 (1981).
G. Dipaola and B. Belleau,Can. J. Chem. 55, 3825 (1977).
O. D. Bonner and P. J. Cerutti,J. Chem. Thermodyn. 8, 105 (1976).
C. Jolicoeur and G. Lacroix,Can. J. Chem. 54, 624 (1976).
J. T. Edsall,J. Am. Chem. Soc. 57, 1506 (1935).
F. Shahidi, P. G. Farrell, and J. T. Edwards,J. Solution Chem. 5, 807 (1976).
L. G. Longsworth,Electrochemistry in Biology and Medicine, T. Shedlovsky, ed. (Wiley, New York, 1955), chap. 12.
J. L. Neal and D. A. I. Goring,Can. J. Chem. 48, 3745 (1970).
H. Hoiland and E. Vikingstad,Acta. Chemica. Scand. A 30, 182 (1976).
J. T. Edward, P. G. Farrell, and F. Shahidi,J. Chem. Soc. Faraday 1,73, 705 (1977).
F. Franks, M. A. J. Quickenden, D. S. Reid, and B. Watson,Trans. Faraday. Soc. 66, 582 (1970).
N. Nichols, R. Skold, C. Spink, J. Suurkuusk, and I. Wadso,J. Chem. Thermodyn. 8, 1081 (1976).
J. P. Guthrie,Can. J. Chem. 55, 3700 (1977).
J. Konicek and I. Wadso,Acta. Chem. Scand. 25, 1571 (1971).
F. Franks, D. S. Reid, and A. Suggett,J. Solution Chem. 2, 99 (1973).
K. P. Prasad and J. C. Ahluwalia,J. Solution Chem. 5, 491 (1976).
M. C. R. Symon, J. A. Benbow, and J. M. Harvey,Carbohydr. Res. 83, 9 (1980).
M. J. Tait, A. Suggett, F. Franks, S. Ablett, and P. A. Quickenden,J. Solution Chem. 1, 131 (1972).
D. T. Warner,Nature 196, 1055 (1962).
A. H. Narten, M. D. Danford, and H. A. Levy,Disc. Faraday Soc. 43, 97 (1967).
K. Kusano, J. Suurkuusk, and I. Wadso,J. Chem. Thermodyn. 5, 757 (1973).
S. A. Angyl and R. Lefur,Carbohydr. Res. 84, 201 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jasra, R.V., Ahluwalia, J.C. Enthalpies of solution, partial molal heat capacities and apparent molal volumes of sugars and polyols in water. J Solution Chem 11, 325–338 (1982). https://doi.org/10.1007/BF00649291
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00649291